Full Text
Introduction
Primary neoplasms of sphenoid sinus are rare in occurrence [1]. Maxillary sinus is most commonly involved followed by ethmoids [2]. It accounts to about 1-2%of all paranasal sinus tumors. They commonly occur in fifth decade. Typically tumor in the sphenoid sinus area occurs by local extension from surrounding structures. The most common neoplasms encountered were squamous cell carcinoma, adeno carcinoma, adenoid cystic carcinoma, lymphoma and neuro endocrine carcinoma etc. The common clinical presentations were Head ache, epistaxis, and abducens cranial nerve palsy [3]. Some patients may have trigeminal nerve palsy, partial third and fourth nerve palsies also. This is because of the close proximity of sphenoid sinus to cavernous sinus. CT or MRI should be performed immediately whenever encountering such less specific, neuro ophthalmic symptoms. The treatment modalities for this aggressive tumor need multimodality treatment. This includes, surgery which can be endonasal biopsy, tumor debulking if possible, followed by radiotherapy and chemotherapy. Proton therapy and stereotactic radio therapy may help to limit the morbidity. The presence of cranial nerve palsy suggests a poor outcome.
Case report
17-years-old male young boy presented with head ache, on and off for four months. Patient was also having epistaxis on and off. There was no history of fever or head injury. After twenty days patient noticed difficulty in moving both eyes (Figure 1). There was no history of visual impairment. There were no other nose, throat or ear complaints.

Figure 1a,b: Showing restricted ocular movements and proptosis.
On examination of nose, there was septal deviation towards left side. Middle meatus was clear, with no sinus tenderness.
Examination of eye revealed proptosis with defective bilateral abduction. Visual acuity is normal. Neck examination showed palpable lymph nodes bilaterally. In nasopharynx a suspicious mass was present (Figure 2).
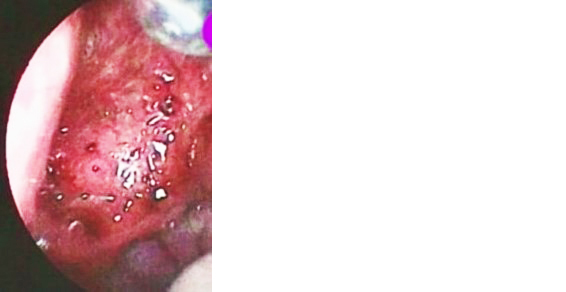
Figure 2: Nasal endoscopy showing mass in the nasopharynx, bleeding on touch.
Magnetic resonance contrast study showed contrast enhancing mass in the sphenoid sinus with extension into cavernous sinus and anteriorly into nasopharynx (Figure 3).
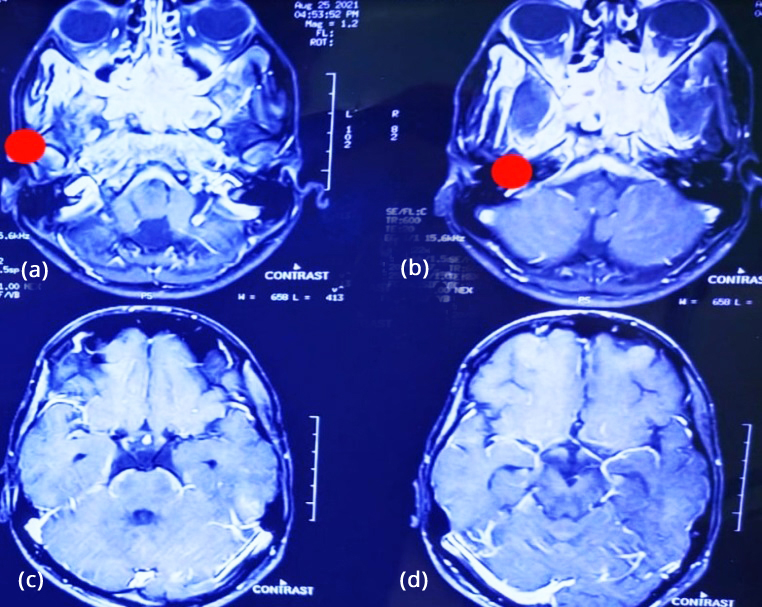
Figure 3a-d: Contrast MRI; showing mass in sphenoid sinus extending into cavernous sinus and nasopharynx.
Patient was taken up for endoscopic excision of the mass. Sphenoid sinus was opened. Pus from the sinus was aspirated and sent for gram stain and culture sensitivity. Debulking was done and tissue was sent for frozen biopsy and routine biopsy. During the procedure significant bleeding was encountered throughout. It was managed successfully. Basing on the age of the patient and clinical findings a clinical suspicion of fungal sinusitis or lymphoma was made.
Frozen biopsy revealed clusters of atypical cells and a possibility of malignancy. Histopathological examination of the tissue revealed nests of closely packed nests of spindle cells with elongated nuclei, nuclear hyperchromasia and frequent mitosis, suggestive of poorly differentiated squamous cell carcinoma (Figure 4).

Figure 4: Histopathology; spindle cells with elongated nuclei, hyperchromasia and mitosis.
Further Immunohistochemistry study with CK and P40 markers confirmed the diagnosis (Figure 5).

Figure 5: Immunohisto chemistry: (a) CK marker revealing epithelial origin of tumor, (b) P 40 marker confirming squamous cell origin of tumor.
During the course of management patient developed ptosis of right eye (Figure 6). Patient was referred to oncologist for further treatment.

Figure 6: Ptosis of right eye.
Whole body PET was done (Figure 7) to know the extent of the lesion and possible metastasis.18F-FDG-370MBq (10mci)/60 minute uptake period. The extent of study was from vertex of the skull to mid-thigh. IV contrast was given. The blood glucose level was 113mg/dl, serum creatinine was 0.60mg/dl.
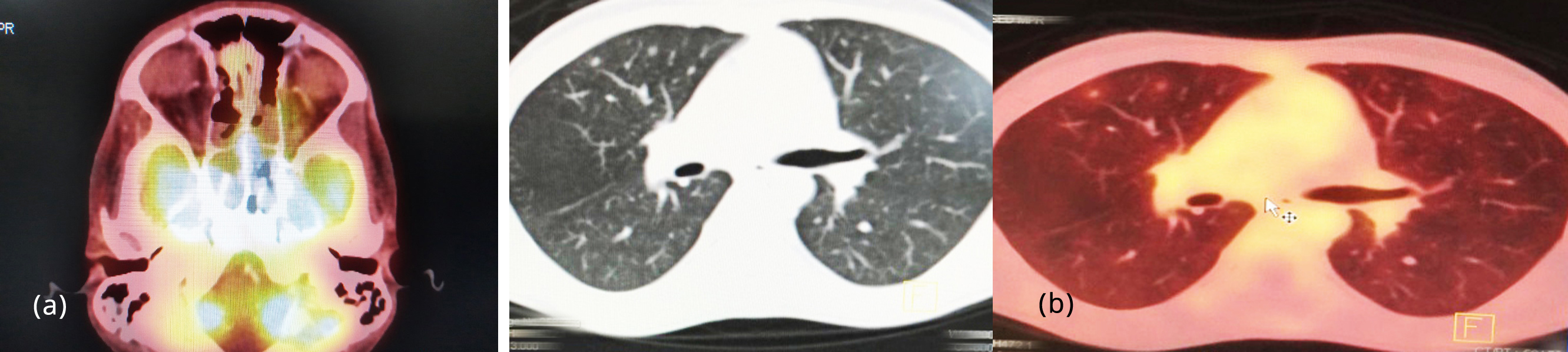
Figure 7: PET CT: (a) Ill-defined lytic lesions in basisphenoid, (b) Tiny lymph nodes in both lungs.
It revealed Ill-defined lytic lesions involving the basisphenoid with intense FDG activity (SUV max11). Multiple enlarged necrotic discrete and conglomerated bilateral level II lymph nodes with Intense FDG uptake (SUV max 12). Few mildly FDG avid tiny bilateral lung nodules.
In view of the extensiveness of the disease, patient was planned for neoadjuvant chemotherapy with Inj. Holoxan. Fluorouracil, cisplatin in cycles followed by concurrent chemo radiation. Poor prognosis explained to the attenders. Patient tolerated the chemotherapy well. After success full completion of chemotherapy PET CT was repeated (Figure 8). It showed reduced intake of uptake in delayed stage. Patient was started on radiotherapy. A dose of 70 grays in 35 fractions received by the patient. He was under observation after completion of chemo radiation.
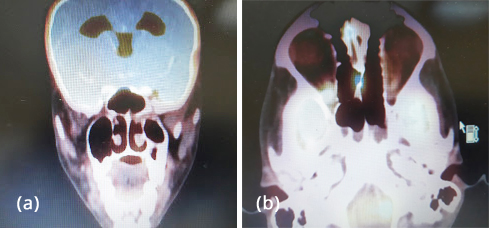
Figure 8a,b: PET CT after chemo showing reduced uptake intake.
Headache and bleeding from the nose were subsided, but the ptosis was persisting even after completion of chemo radiation (Figure 9).

Figure 9: Ptosis of Right eye after chemo radiation.
In due course patient developed loss of vision unfortunately. Magnetic resonance imaging (MRI) brain was done (Figure 10) which showed extensive progression involving cavernous sinus sella, supracellar area, pituitary stalk, orbital apex, orbits, optic nerves, optic chiasma, oculomotor nerve and clivus. Patient is now put on palliative care.
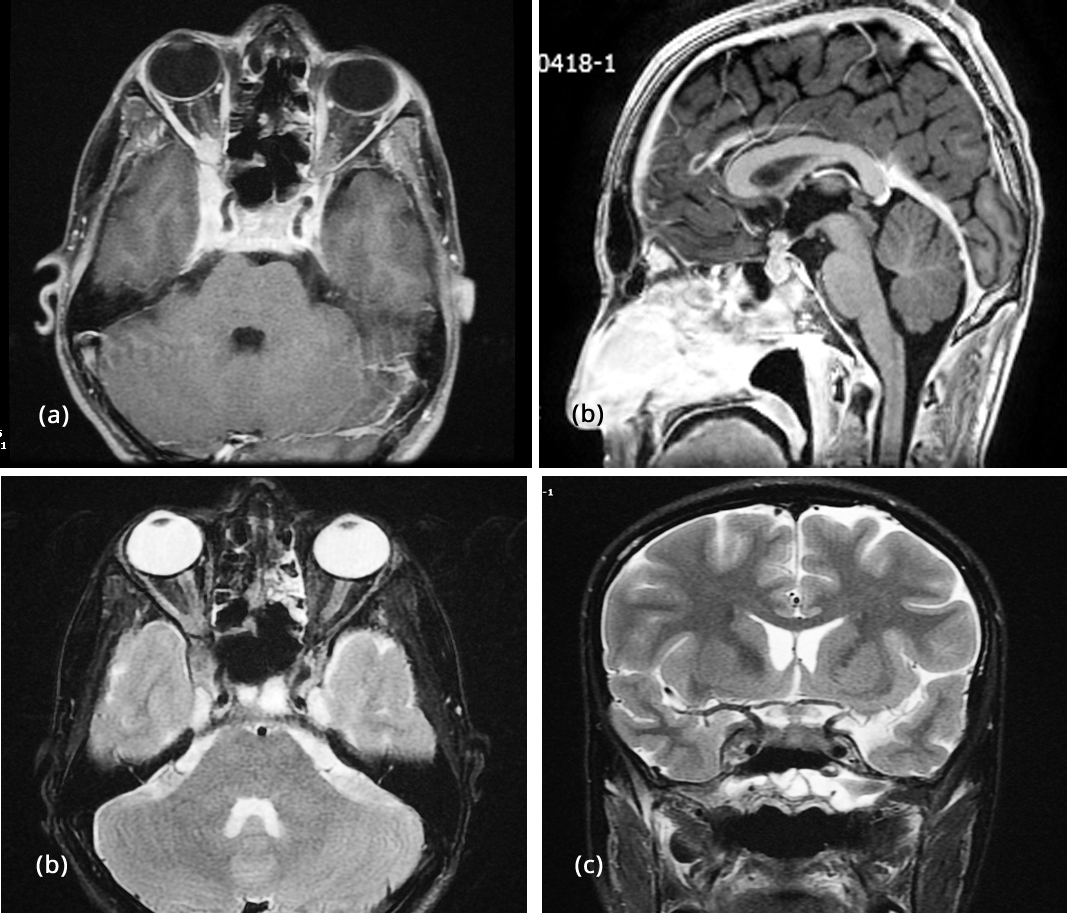
Figure 10: MRI after chemo radiation, (a) Post contrast T1FS Axial showing involvement of cavernous sinus, orbital apex, intra orbital extension, (b) T1 sagittal, (c) T2 FS coronals showing involvement of cavernous sinus, orbital apex, intra orbital extension, (d) T2 sagittal showing involvement of pituitary stalk.
Discussion
The sphenoid sinus also called as “silent sinus” is closely associated with important structures like carotid artery, optic nerve, cavernous sinus, brain stem, sella. Unlike the tumors of other para nasal sinuses which presents with rhinorrhea, nasal obstruction and epistaxis, the sphenoid sinus tumors presents with nonspecific headache, neuro ophthalmic symptoms. Typically the headache is ipsilateral, deep retro orbital. It probably occurs because of involvement of sphenopalatine nerve or ganglion. The ocular symptoms include diplopia, ptosis and sometimes visual loss. These symptoms are because of involvement of II III IV or VI nerves. Our patient however has good vision. Some patients may present with cranial neuropathies initially. This corresponds to advanced disease. To look for these features initial thorough clinical examination is mandatory to diagnose and to assess the extensiveness of the disease. Before subjecting the patient for any surgical intervention like biopsy or debulking CT, MRI is a must. This will help in diagnosing, staging, know the vascularity of the lesion, perineural or intracranial spread. Sometimes MRI may also indicate specific type of tumor type such as lymphoma [4, 5]. Angiography also may be needed in some cases.
Various endoscopic approaches are there for sphenoid sinus, like trans nasal, trans septal, trans ethmoidal [6]. The choice of approach depends on the surgeon’s familiarity. However if the tumor is big posterior ethmoidectomy, sphenoidotomy approaches are to be considered. Because of tumor extension complete surgical excision is not possible. The role of surgery is for biopsy and debulking. Sometimes with extensive disease teaming with neuro surgeon and head and neck surgeon may be needed.
The biopsy must be sufficient to get a reliable diagnosis. If the material is not sufficient arriving at an appropriate diagnosis may be difficult. In our case the material was sufficient for the pathologist to give frozen biopsy report and histopathological report also.
Conclusion
Primary tumors of the sphenoid sinus are rare. They present with headache, cranial neuropathies attributing to cavernous sinus pathology. Our patient also had the similar presentation. Patient presentation is more of neurological rather than rhino logical signs and symptoms. The best results are obtained with early diagnosis and early intervention with multidisciplinary approach which includes tumor debulking followed by concomitant chemo radiation rather than radiation alone. The presence of cranial neuropathy suggests a poor outcome.
Conflicts of interest
Authors declare no conflicts of interest.
References
[1] Osguthorpe JD. Sinus neoplasia. Arch Otolaryngol Head Neck Surg. 1994; 120:19–25.
[2] Dulguerov P, Jacobsen MS, Allal AS, Lehmann W, Calcaterra T. Nasal and paranasal sinus carcinoma: are we making progress? A series of 220 patients and a systematic review. Cancer. 2001; 92(12):3012–3029.
[3] DeMonte F, Ginsberg LE, Clayman G. Primary malignant tumors of the sphenoidal sinus. Neurosurgery. 2000; 46(5):1084–1091.
[4] Leong JL, Batra PS, Citardi MJ. Three-dimensional computed tomography angiography of the internal carotid artery for preoperative evaluation of sinonasal lesions and intraoperative surgical navigation. Laryngoscope. 2005; 115(9):1618–1623.
[5] Ginsberg LE. Neoplastic diseases affecting the central skull base: CT and MR imaging. AJR Am J Roentgenol. 1992; 159(3):581–589.
[6] Castelnuovo P, Pagella F, Semino L, De Bernardi F, DeluG. Endoscopic treatment of the isolated sphenoid sinus. lesions. Eur Arch Otorhinolaryngol. 2005; 262(2):142–147.