Full Text
Introduction
Neuroarthropathy also called neuropathic arthropathy or Charcot’s arthropathy refers to neuropathic joints from numerous etiologies. First officially recorded case was in 17th century. First description was by W. Musgrave in 1703. Jean Martin Charcot, the father of neurology described gait abnormalities with syphilis in 1868 and called it Tabes Dorsalis. Jordan in 1934 was the first to describe the relationship of diabetes to neuropathic arthropathy.
The following joints are involved, although most common site is the foot: i) Tarsometatarsal joints medial more than lateral – 60%, ii) Metatarsophalangeal joints – 30%, iii) Ankle joint – 10%, iv) Knee joint – rare, v) Shoulder joint in syringomyelia Hip joint, vi) Other joints are rarely involved. The following table (Table 1) presents the causative factors.
Table 1: Neuropathic arthropathy.
|
A. Congenital
- Spinal dysraphism
- Congenital indifference to pain (Asymbolia)
- Cerebral palsy
B. Acquired
· Central neuropathy
- Injury to brain/ spinal cord
- Syringomyelia
- Neurosyphilis
- Spinal cord tumors/ infection
- Poliomyelitis
· Peripheral neuropathy
- Diabetes mellitus
- Leprosy
- Peripheral nerve injury
- Alcoholic neuropathy
· Iaotrogenic – radiation, steroid therapy local/systemic etc.,
|
Pathology
The joint loses its ability to perceive proprioception, peripheral neuropathy and loss of pain sensation to trauma. In addition to micro trauma, there are neuropathy and vasculopathy. The sensation to pressure, puncture and other stress conditions is lost.
The regions of the foot affected include fore, mid and hind parts. Syringomyelia is the most common cause of neuropathic arthropathy of the shoulder and elbow. The clinical features include loss of function, edema, warmth, swelling and ulcers. Radiographic findings are mostly divided into two categories one is atrophic with osteolysis and two is hypertrophic with fractures and dislocations.
Radiographic features (6 Ds - Vochum and Rowe)
- Dense bones (subchondral sclerosis)
- Degeneration
- Destruction of articular cartilage
- Deformity (pencil-point deformity of metatarsal heads)
- Debris (loose bodies)
- Dislocation
The hypertrophic type includes mostly sclerotic bones with destruction of cartilage associated with subluxations. Fragmented bone (Debris) is noted in para articular soft tissues. Features of atrophic type include licked candy stick appearance, osteolysis and bone resorption. Neuroarthropathy is less frequently encountered in the axial skeleton as compared with peripheral skeleton, although manifestations are similar radiologically.
Congenital - asymbolia
Loss of the ability to comprehend by touch, the form and nature of an object. A form of aphasia in which the significance of signs cannot be comprehended, which is also called asemasia. Congenital insensitivity to pain with anhidrosis is an exceedingly rare disease. Only 31 cases have been reported. It is a severe autosomal recessive condition that leads to self-mutilation in the first months of life. Bone fractures, multiple scars, osteomyelitis, joint deformities, and limb amputation are present as the children grow older (Figures 1abcde). Mental retardation is common. The actual physiopathologic mechanism of this developmental disorder remains unknown.
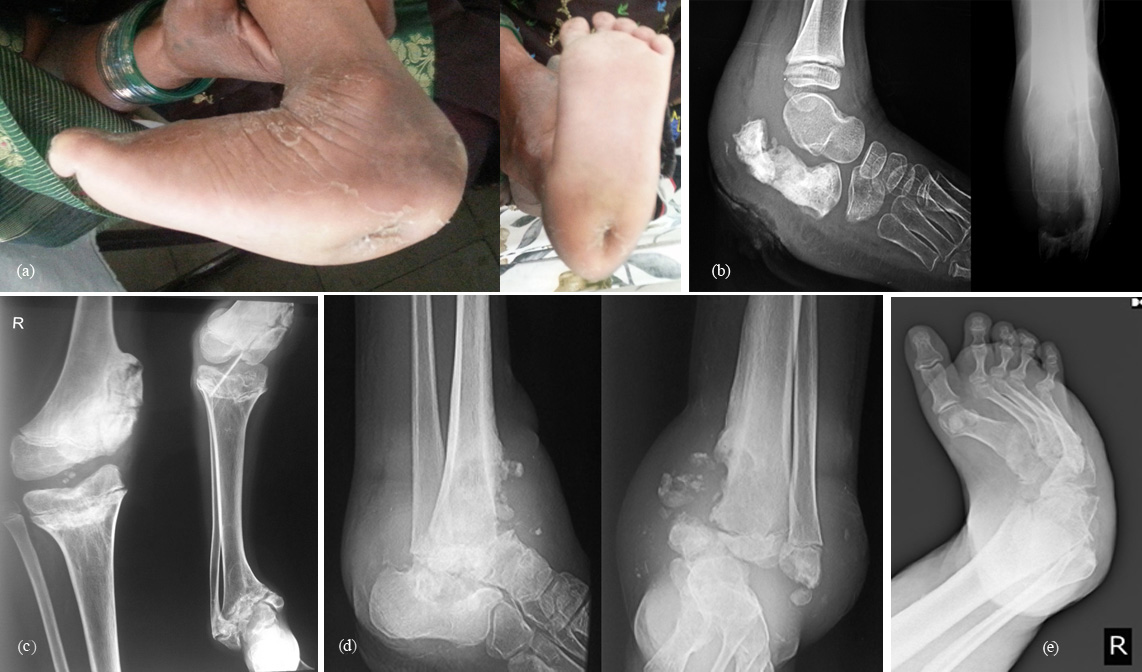
Figure 1: (a) Clinical photograph of the foot in congenital asymbolia (insensitivity to pain), (b) Neuroarthropathic joint in asymbolia. Note superimposed infection with osteomyelitis and pathological fracture of calcaneus, (c) Child with asymbolia – Bilateral neuroarthropathy involving right knee and ankle, (d) 14 year-male C/O painless swelling of left ankle joint with a discharging sinus since 4 months - Neuropathic arthropathy involving the ankle and hind foot in asymbolia, (e) Neuroarthropathic foot in cerebral palsy.
Amongst the acquired causes of neuropathic arthropathy is diabetes, particularly common in India. Foot is the most common site for this disorder. Vasculopathy, neuropathy, trauma and infection are the common factors in the manifestation of radiological findings. Articular injury with diminished sensation and ischemia result in aggressive appearing neuroarthropathy. Motor neuropathy can lead to muscular atrophy whereas autonomic neuropathy leads to soft tissue edema. There is associated tendinopathy with tendon dysfunction. It is more common in mid tarsal joints (Lisfranc’s and Chopart joints) but may involve other joints. Osteomyelitis is common in the calcaneus, metatarsal head and toes as they are weight bearing areas. Lisfranc’s joint disruption is common in diabetes. The Lisfranc’s joint involvement in diabetes is classified by Brodsky (Table 2).
Table 2: Brodsky classification 1992.
|
Type 1 – Lisfranc’s joint – 27-60%
Type 2 – Chopart’s joints and subtalar joints – 30-35%
Type 3A – Ankle joint – 9%
Type 3B – The posterior calcaneus
Type 4 – Multipel regions of the foot and / or ankle
Type 5 – The forefoot
|
The majority of Lisfranc’s dislocations involve the second tarsometatarsal joint. The second cuneiform bone is smaller than the first or the third. Hence, it is locked. The bases of lateral four metatarsals are united by strong interosseous ligaments so that the entire group is frequently displaced en masse (Figures 2abcd).
The natural history of the joint destruction process has a classification scheme of its own, offered by Eichenholtz decades ago:
Stage 0: Clinically, there is joint edema, but radiographs are negative. Note that a bone scan may be positive before a radiograph is, making it a sensitive but not very specific modality.
Stage 1: Osseous fragmentation with joint dislocation seen on radiograph ("acute Charcot").
Stage 2: Decreased local edema, with coalescence of fragments and absorption of fine bone debris
Stage 3: No local edema, with consolidation and remodeling (albeit deformed) of fracture fragments. The foot is now stable.
Destroyed Tarsometatarsal joints in the medial left foot, with fracture and dislocation of fragments; these are classic findings. Also note loss of the foot arch and acquired flat foot (pes planus) deformity.
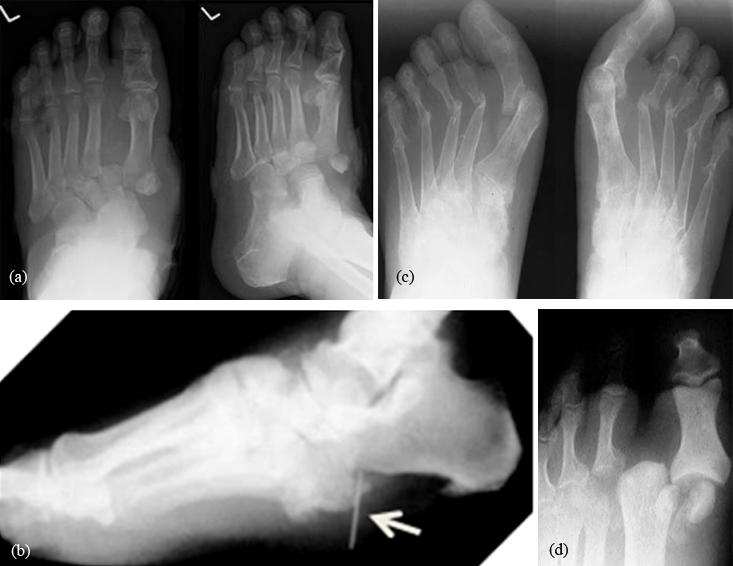
Figure 2: (a) Lisfranc’s joint in diabetic patient. Note divergent dislocation, (b) Neuroarthropathy in diabetic patient. Note the foreign body in the plantar aspect, (c) Neuropathic arthropathy–fore foot -atrophic type in diabetic patient. Note pencilling of the metatarsals, (d) diabetic foot with amputation of second toe.
Spinal cord lesions
Compressive myelopathy may be traumatic, infective or due to malignancy. It is mostly caused by tuberculosis of the spine in India. Pott’s spine destroys the disk and adjacent margins of the spine with a large paravertebral, prevertebral and post vertebral soft tissues swellings are noted (Figures 3ab). The posterior soft tissue swelling compresses the cord and produces neuropathic changes in the hips, knees, ankles and feet, depending upon the site involved. Traumatic fracture dislocations may produce neuroarthropathy in hips and lower limb joints (Figure 3c). Cervical spinal tuberculosis may produce neuroarthropathy of the shoulder (Figure 3d). But the most common cause of neuroarthropathy of the shoulder and elbow is syringomyelia (Figure 3e). In pre-antibiotic days syphilis was a common cause of neuroarthropathy (Figure 3f).
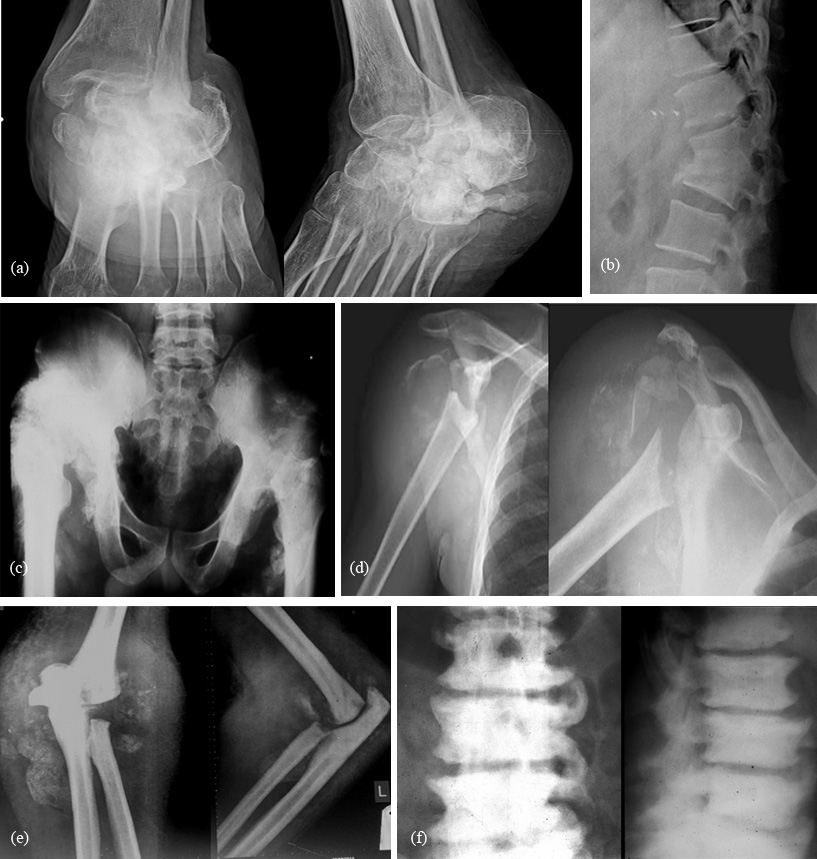
Figure 3: (a) 36 year-male – Neuroarthropathy of ankle and hind foot from TB spine, (b) Same patient - Healed tuberculosis of the spine involving L2, L3, (c) 45 year-male with traumatic paraplegia - Bilateral neuroarthropathy due to traumatic paraplegia. Note the ectopic ossifications around the joints, (d) Neuropathic arthropathy of right shoulder in syringomyelia, (e) Progressive neuroarthropathy of the elbows in syringomyelia, (f) Charoct’s spine in tabes dorsalis.
Leprosy
There are three types, one tuberculoid, two neural, three mixed. Peripheral nerves are mostly involved. Hence, hands and feet’s have ulcers, infection and neuropathic changes in the joints (Figure 3g).
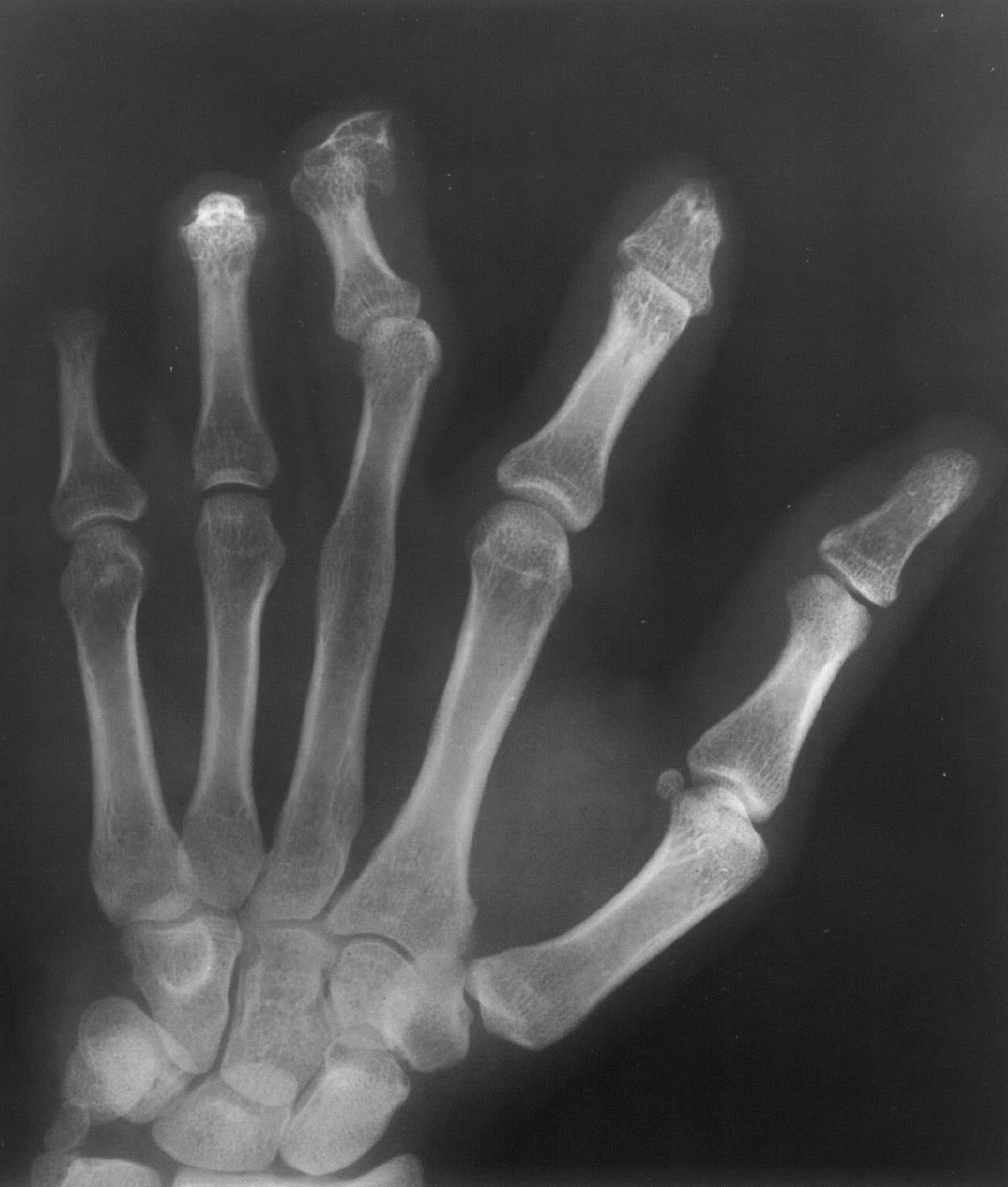
Figure 3g: Hand in a leprotic patient with autoamputation and neuroarthropathy.
Among the iatrogenic causes, steroid therapy particularly intraarticular injection is most common in producing neuroarthropathy (Figure 4a). Radiation therapy for carcinoma of breast has been a common modality of therapy. This may produce neuroarthropathy of the ipsilateral shoulder (Figure 4b). In addition to arthropathy, soft tissue atrophy and calcifications are common in the radiation field.
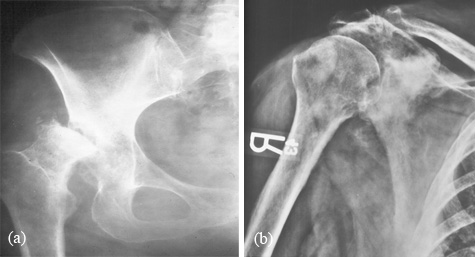
Figure 4: (a) Neuroarthropathy of the right hip due to treatment with steroids, (b) Neuroarthropathy of the shoulder due to radiation. Note sclerotic changes in the adjacent bones.
Primary or secondary amyloidosis in chronic rheumatoid joints is another cause of neuroarthropathy (Figure 5).
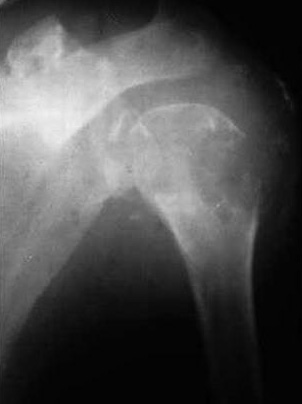
Figure 5: Neuropathic arthropathy in amyloidosis.
Summary
Neuroarthropathy (neuropathic arthropathy) is a common manifestation due to several causes. Whichever joint is involved the radiological manifestations are of two types. Of these, hypertrophic variety is common than atrophic. The radiological manifestations include joint destruction, debris, disorganization, increased density and dislocations. Diabetic foot has a special significance as besides neuroarthropathy, there are infection and trauma. The spectrum of radiological findings by conventional radiography is illustrated.
Acknowledgements
Acknowledgements are due to Department of Radiology and Imaging, NIMS and KIMS, Secunderabad, Telangana, India.
Conflict of interest
The author declares no conflict of interest.
References
1. Basu SP. Bone changes in leprosy. Ind J Radiol. 1972; 26:239-249.
2. Bhaskaran R, Suresh K, Iyer GV. Charcot’s elbow (a case report). J Postgrad Med. 1981; 27:194-196.
3. Bruckner FE, Howell A. Neuropathic joints. Semin Arthritis Rheum. 1972; 2:47-49
4. Delano PJ. The pathogenesis of Charcot’s joint. Am J Rheum. 1946; 56:189-197.
5. Feldman F, Johnson AM, Walter JF. Acute axial neuroarthropathy radiology 1974; 111:1-16:
6. Foster SC, Foster RR. Lisfranc’s Tarsometatarsal fracture dislocation Radiology. 1976; 120:70-83.
7. Gupta R. A short history of neuropathic arthropathy. Clin Ortho. 2001; 64.
8. Harrison M, Sachen M, et al. Spinal Charcot Arthropathy, Neuro Surg. 1991; 28: 273-277.
9. Stendler A. Tabetic arthropathies J Amer Med Assoc. 1931; 29:250-256.
10. Williams B. Syringomyelia. Neurosurg Clin North Am. 1990; 1:653-685.