Full Text
An Abdominal Aortic Aneurysm (AAA) is a relatively common and potentially life threatening condition. Approximately 90% of AAA involve the infra renal segment of the abdominal aorta. Untreated, an AAA eventually carries a mortality rate of 100%. Thirty to 75% of patients with a ruptured AAA die of sudden cardiovascular collapse before arriving at a hospital. The overall mortality rate in patients with a ruptured AAA ranges from 67% to 89% [1]. Therefore, detection of AAA before rupture and elective repair can prolong survival and decrease the complication rates. The problem with detecting an AAA lies in the fact that most often an AAA is asymptomatic or present with non-specific symptoms. A high index of suspicion is hence mandatory to identify these cases at an early stage.
Case summary
A 57-year-old male, hypertensive who is on irregular treatment, non-diabetic, chronic smoker (38 pack years), alcoholic, presented to the Gastroenterology OPD of a tertiary care hospital with complaints of abdominal discomfort on and off, associated with pain in the lower back since the last 6 months. On admission his vitals were stable and abdomen was soft to palpation with no other abnormalities detected on examination. Serum amylase and lipase were done which were within normal limits.
An ultrasound abdomen was performed. It revealed the presence of a saccular out-pouching of the infrarenal segment of the abdominal aorta measuring 4.0 cm in length and 3.8 cm in width with an eccentric thrombus measuring 2.1 cm. Patient was then referred to the cardiology department where he was evaluated further.
Examination of the cardiovascular system revealed normal peripheral pulses and normal findings on systemic examination. CT whole abdomen with contrast was done which revealed a saccular aneurysm measuring 5.0 cm in length & 4.7 cm in diameter arising from the anterior aspect of the infrarenal portion of the abdominal aorta with an eccentric mural thrombus. The aneurysm was located 25 mm caudal to the renal artery origin and 40 mm cranial to the aortic bifurcation (Figure 1).
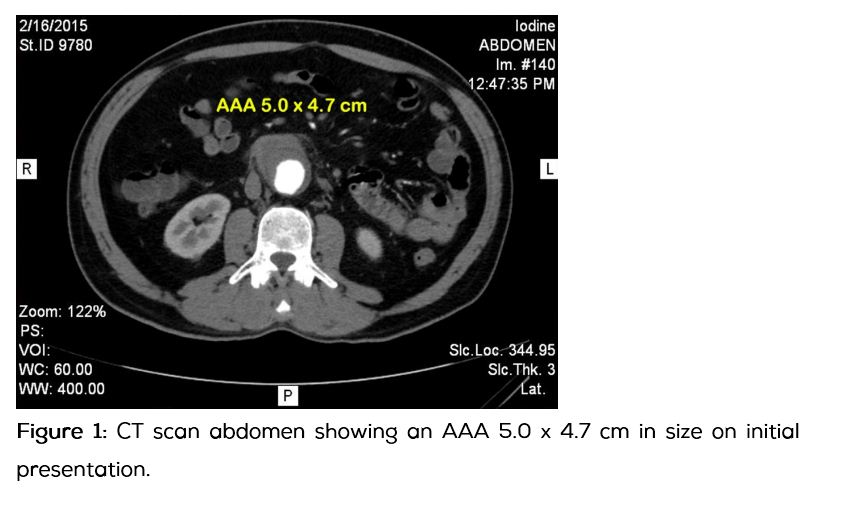
The patient was managed conservatively and was advised to come for review if pain worsens or persists. Fifteen days after discharge, he presented to the ER with severe low back pain which was relieved in sitting position. On admission to the ICCU, his blood pressure was 200/100 mm Hg in right & left upper limb. And it was 196/108 mm Hg in right & left lower limb. His pulse rate was 97 beats per minute, regular, normal volume, felt equally in all peripheries with no radioradial or radiofemoral delay. Cardiovascular and abdominal examination findings were normal.
He was haemodynamically stabilized and his blood pressure was controlled with IV labetolol and oral losartan. A repeat CT abdominal angiogram was done and it showed an increase in size of the saccular aneuruysm to 5.8 cm in diameter and 5.2 cm in length with a leak and impending rupture (Figure 2). 2D Echo, Chest X Ray and ECG were within normal limits.
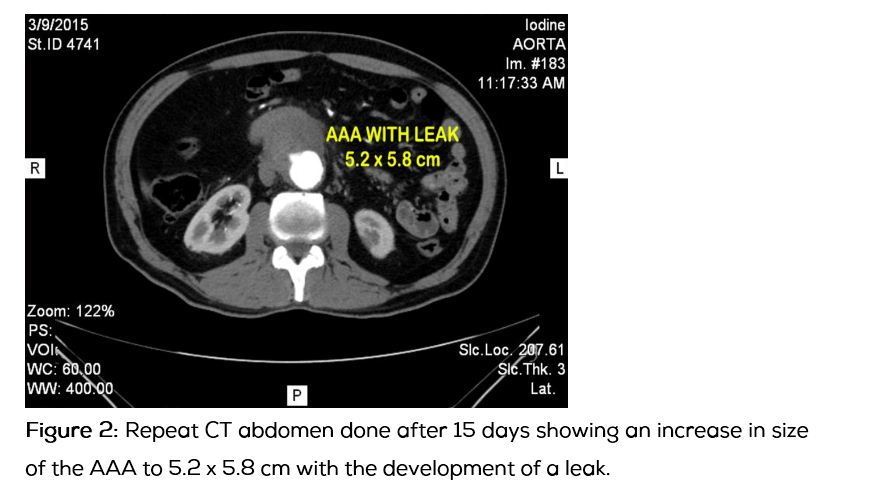
After explaining the nature of the underlying illness and course of treatment planned, an informed consent was taken and a standard Endovascular Aneurysm Repair (EVAR) procedure using a Medronic Y stent graft to the infra renal segment (Figures 3 to 5) was done via femoral approach.
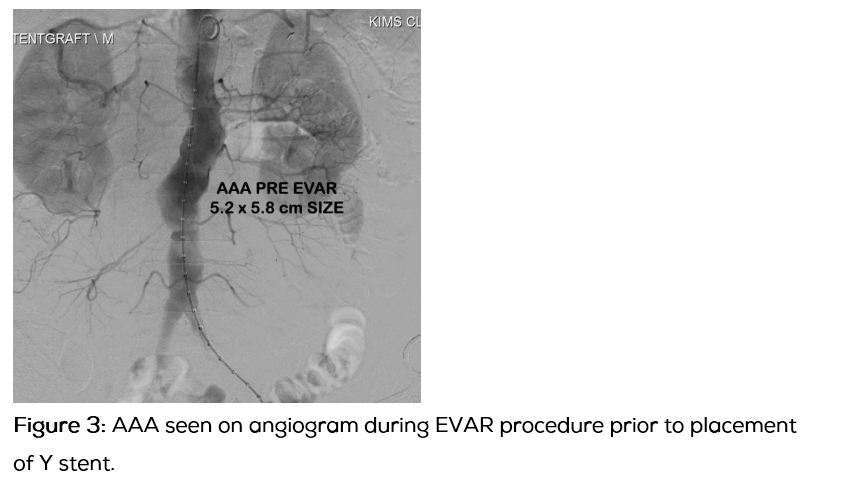
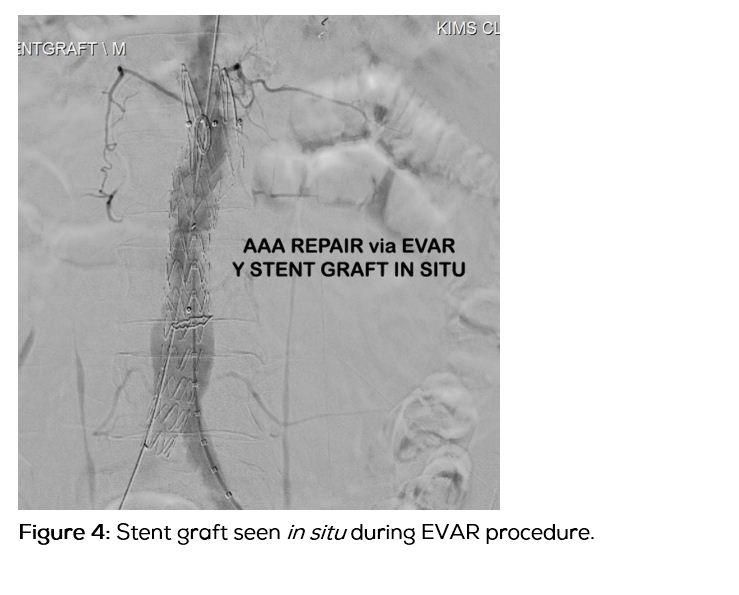
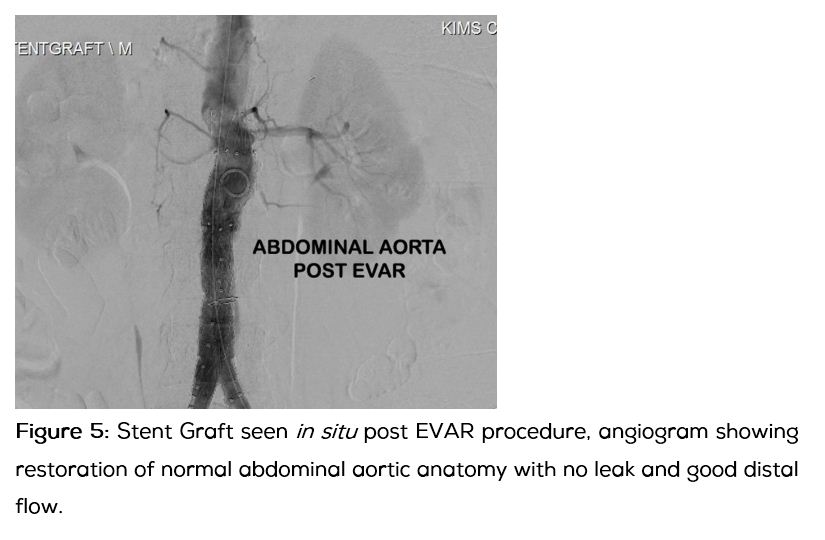
The procedure was uneventful and patient recovered well. On discharge, he was asymptomatic and hemodynamically stable. He is now on regular follow up.
Discussion
Abdominal aortic aneurysm (AAA) is defined as a permanent localized dilatation of the aorta that has at least a 50% increase in diameter as compared with the expected normal diameter of the aorta, which may vary according to age, sex and body size [1]. In individuals above 50 years of age, the normal diameter of the infrarenal aorta is 1.5 cm in men and 1.7 cm in women. An enlargement of the infrarenal aortic diameter to more than or equal to 3 cm is taken as an AAA.
Patients at increased risk of development of an AAA include males older than 65 years having peripheral vascular disease who have ever smoked. Hypertension, Chronic obstructive pulmonary disease (COPD), coronary artery disease, previous history of aneurysm repair or peripheral aneurysm (popliteal or femoral) are also the other known risk factors. Lesser common risk factors include Marfan syndrome, Ehlers-danlos syndrome and collagen vascular diseases. In less than 5% of cases, AAA is caused due to mycotic aneurysm of hematogenous origin most commonly due to gram positive organisms [2]. There has been a strong risk association amongst patients, with first degree relatives having AAA. The familial prevalence rate of AAA has been estimated as 15-25%. Tilson et al. described the potential for an autoimmune basis for the development of AAA involving the DRB1 major histocompatibility locus [3].
The most common pathogenesis involved is a proteolytic degradation in the media of the aortic wall. Most AAA occurs in individuals with advanced atherosclerosis. Atherosclerosis may induce AAA formation by causing mechanical weakening of aortic wall with loss of elastic recoil in the setting of underlying medial degeneration leading to ischemic changes via obstruction of the vasa vasorum [4]. Pathologically AAA are classified as fusiform and saccular, though clinically a spectrum of in between forms between these two extremes are also seen. Aneurysm diameter is an important risk factor for rupture (Table 1). In general, AAA gradually enlarge (0.2-0.8 mm/year) and eventually rupture [5].
Table 1: Abdominal aortic aneurysm size and estimated annual risk of rupture
|
AAA Diameter (cm)
|
Rupture Risk (%/y)
|
|
< 4
|
0
|
|
4-5
|
0.5-5
|
|
5-6
|
3-15
|
|
6-7
|
10-20
|
|
7-8
|
20-40
|
|
>8
|
30-50
|
| |
Abbreviations: AAA = abdominal aortic aneurysm.
Hemodynamic factors play an important role in determining the risk of rupture. Areas of high stress have been found in AAA and appear to correlate with the site of rupture. Computer-generated geometric models have demonstrated that aneurysm volume is a better predictor of areas of peak wall stress than aneurysm diameter. AAA rupture is believed to occur when the mechanical stress acting on the wall exceeds the strength of the wall tissue. The actual tension in the AAA wall appears to be a more sensitive predictor of rupture than diameter of aneurysm alone. Hence, control of acute blood pressure in patients with AAA an elevated blood pressure is paramount important to prevent rupture [6].
Most common presentation is an elderly white male, mostly above 50 years of age, with a history of chronic smoking, presenting with sudden onset of abdominal pain. The classical presentation of a pulsatile abdominal mass is seen in less than 50% of cases. Most patients with AAA are asymptomatic or present with vague abdominal, back or groin pains and are detected incidentally on diagnostic imaging for other reasons. More than 80% of patients with ruptured AAA present without a previous diagnosis of AAA, which contributes to an initial misdiagnosis rate of 24-42% [7].
Smoking is the most common risk factor associated with AAA. Smoking may augment the risk of rupture by 1.5 – 2.4 folds [1]. The US Preventive Services Task Force (USPSTF) recommends a one-time screening for AAA with ultrasonography (USG) in men who are 65-75 years of age and have a history of smoking (i.e., “ever smoker”: at least 100 cigarettes during lifetime). They also recommend selectively offering screening for mean age of 65-75 years based on other risk factors in men who do not have a smoking history [8].
In addition to the diameter of aneurysm, factors such as sex, aneurysm expansion rate, family history, and chronic obstructive pulmonary disease (COPD) also affect the risk of rupture (Table 2) [5].
Table 2: Factors affecting risk of abdominal aortic aneurysm rupture
| |
Low risk
|
Average risk
|
High risk
|
|
Diameter
|
< 5 cm
|
5-6 cm
|
>6 cm
|
|
Expansion
|
< 0.3 cm/y
|
0.3-0.6 cm/y
|
>0.6 cm/y
|
|
Smoking/COPD
|
None, mild
|
Moderate
|
Severe/steroids
|
|
Family history
|
No relatives
|
One relative
|
Numerous relatives
|
|
Hypertension
|
Normal blood pressure
|
Controlled
|
Poorly controlled
|
|
Shape
|
Fusiform
|
Saccular
|
Very eccentric
|
|
Wall stress
|
Low (35 N/cm2)
|
Medium (40 N/cm2)
|
High (45 N/cm2)
|
|
Sex
|
...
|
Male
|
Female (3 times higher risk)
|
| |
Abbreviations: COPD = chronic obstructive pulmonary disease.
USG is the standard imaging tool for AAA and should be done as an emergency bedside procedure when an AAA is suspected. USG has a sensitivity of nearly 100% and a specificity approaching 96% for the detection of infrarenal AAA. Ultrasonography can also detect free peritoneal blood indicating a leak or rupture [9]. In one study it was found that approximately 1/3rd of the abdominal aorta was not visualized in nonfasted patients without fasting on bedside USG [10]. Hence all patients with a high index of suspicion of AAA with a negative USG should undergo further evaluation through imaging.
CT has a sensitivity of nearly 100% in detecting AAA and has advantage over USG for definition of the aortic size, extent, involvement of visceral arteries, extension into the suprarenal aorta, as well as presence/absence of thrombus. CT is also the best modality for determining whether a patient is a candidate for Endovascular Aneurysm Repair (EVAR). CT also helps determine the condition of the iliac arteries and their dimensions, so as to plan for EVAR [11].
Management of an AAA depends on the type of AAA and integrity or increase in size of the AAA. All saccular aneurysms irrespective of size, symptoms or integrity should be treated. Asymptomatic patients with inflammatory AAA or an AAA that is associated with distal emboli, pain or bowel obstruction require emergency repair, regardless of the size, type or integrity of the aneurysm. It is not the intensity of pain but the presence of pain that should guide the need for intervention. Any patient who is a known case of an AAA who presents with back pain should undergo a repeat CT angiogram to look for change in size and rule out impending rupture of the AAA.
Presence of hypertension increases the aortic wall tension, which is a significant predictor of impending rupture. BP control is essential and the best drug is a beta blocker in this setting. Smoking cessation and beta blocker therapy should be instituted to lower the blood pressure and reduce stress on the arterial wall. These agents can be administered safely, unless the patient has a contraindication for their use (like COPD, allergy beta-blockers, bradycardia, or severe chronic heart failure).
Definitive treatment depends upon the integrity of the AAA. If ruptured, open surgical repair is the best treatment of choice. Unruptured aneurysms can undergo elective repair. Two primary methods of AAA repair are open surgical repair and EVAR.
Open repair is most established as a definitive treatment for many years and EVAR is generally advocated for patients at high risk for open repair. However in recent years EVAR has exceeded open repair as procedure of choice for AAA [12]. The perioperative mortality has been substantially reduced in EVAR as compared to open surgery. The absolute reduction in complications ranges from 30-70%, primarily in cardiac, pulmonary and gastrointestinal systems. One of the striking outcomes in patients undergoing EVAR is early recovery and reduced ICU/hospital stay compared to those undergoing open surgery [1].
Decision-making with regard to elective repair of abdominal aortic aneurysms (AAA) requires careful assessment of factors that influence risk of rupture, operative mortality, and life expectancy. Individualized consideration of these factors in each patient is essential, and the role of patient’s preference is of increasing importance. It is not possible or appropriate to recommend a single threshold diameter for intervention which can be generalized to all patients. Based upon the current evidence, 5.5 cm is the best threshold for repair in an “average” patient. However, subsets of younger, good-risk patients or aneurysms at higher risk of rupture may be identified in whom repair at smaller sizes is justified. Conversely, delay in repair until larger diameter may be best for older, higher-risk patients, especially if endovascular repair is not possible [5].
A study by Lederle et al. found that with AAA smaller than 5.5 cm, elective repair did not improve survival [13]. Prospective studies suggest that following aneurysms larger than 5.5 cm with serial ultrasonography or computed tomography (CT) is generally safe; this threshold may be lower for women. Intervention at diameter <5.5 cm appears indicated in women with AAA due to their inherently high risk of rupture compared to men [5].
If a patient has suitable anatomy, endovascular repair may be considered, and it is most advantageous for older, higher-risk patients or patients with a hostile abdomen or other technical factors that may complicate standard open repair. With endovascular repair, perioperative morbidity and recovery time are clearly reduced; however, there is a higher reintervention rate, increased surveillance burden, and a small but ongoing risk of AAA rupture. In the absence of large scale randomized control trial data, patient preference remains the strongest determinant in deciding between EVAR and open repair.
Thus, the decision to repair an AAA is a complex one in which the patient must play an important role. In many patients, the decision to operate is a balance between risks and benefits. In some very elderly patients or patients with limited life expectancy, aneurysm repair may not be appropriate. In these patients, the consequences of rupture should be frankly discussed.
The risk of in-hospital mortality after open and EVAR procedures can be assessed using the AAA SCORE developed by Ambler et al in 2014 [14]. The long term prognosis after successful AAA repair is related to associate comorbidities and survival is decreased with associated chronic heart failure and COPD. Overall, AAA is very durable with < 5% complication rate. Survival rate of patients with successful AAA repair is comparable to that of individuals in age matched general population who have never had an aneurysm [15].
An AAA is a smoldering disaster waiting to happen within the body of an individual. Untreated it carries a mortality of 100%. While back pain is one of the commonest complaints with which a patient seeks medical attention in the emergency room or outpatient department of any hospital, AAA remains the most misdiagnosed of the life threatening emergencies. The reason being that many a time it presents with vague symptoms and a nearly normal physical examination in its unruptured state.
The patient might very often report to a gastroenterologist or an orthopedician for a seemingly innocuous abdominal pain or lower back discomfort. It is hence prudent that the clinician should carry a high degree of suspicion especially in an elderly male more than 65 years of age with a history of smoking who complains of lower back pain. A simple bedside ultrasound while not 100% fool proof, will in many cases help to exclude an underlying AAA.
Once an AAA is diagnosed it is important to remember that it is not the severity of pain, but the mere presence of pain that should guide the need for intervention. As the very presence of pain is often a harbinger of an impending rupture. All saccular aneurysms irrespective of size should be treated as they are notorious to cause leaks and ruptures when compared to their fusiform counterparts. As an untreated ruptured AAA carries a 100% mortality rate while after its successful repair the patient has a survival rate comparable to the general population, timely repair is of utmost importance to reduce the disease burden in terms of mortality and morbidity.
Conclusion
A careful history and physical examination coupled with high degree of clinical suspicion and appropriate use of investigations can go a long way in early detection and timely management so as to reduce the complications arising from this deadly disease.
Key points: Any saccular aneurysm should be intervened irrespective of symptoms and size of the aneurysm due to their high risk of rupture. Severity of pain is not the criteria for a leaking aneurysm. Presence of pain however mild, indicates a leaking aneurysm. All smokers more than 65 years of age should have a screening USG abdomen to look for an AAA. Family history of AAA also is an important indication for USG screening. All EVAR patients should be followed up meticulously for endovascular leaks. Counselling for discontinuation of smoking is an important intervention in the management of AAA. Risk of paraparesis during endovascular procedures should be kept in mind and need for CSF drain should be planned if required.
Acknowledgements
Acknowledgements are due to the Krishna Institute of Medical Sciences (KIMS), Minister Road, Secunderabad – 500003, Telangana, India.
Conflict of Interest
The authors declare no conflict of interest.
References
1. Al-Omran M, Verma S, Lindsay TF, Weisel RD, Sternbach Y. Clinical decision making for endovascular repair of abdominal aortic aneurysm. Circulation. 2004; 110(23):e517–e523. DOI: 10.1161/01.CIR.0000148961.44397.C7
2. Blanchard JF, Armenian HK, Friesen PP. Risk Factors for abdominal aortic aneurysm: results of a case-control study. Am J Epidemiol. 2000; 151(6):575–583.
3. Tilson MD, Ozsvath KJ, Hirose H, Xia S. A genetic basis for autoimmune manifestations in the abdominal aortic aneurysm resides in the MHC class II locus DR-beta-1. Ann N Y acad Sci. 1996; 800:208–215.
4. Wassef M, Baxter BT, Chisholm RL, Dalman RL, Fillinger MF, et al. Pathogenesis of abdominal aortic aneurysms: a multidisciplinary research program supported by the National Heart, Lung, and Blood Institute. J Vasc Surg. 2001; 34(4):730–738.
5. Brewster DC, Cronenwett JL, Hallett JW Jr, Johnston KW, Krupski WC, et al. Guidelines for the treatment of abdominal aortic aneurysms. Report of a subcommittee of the Joint Council of the American Association for Vascular Surgery and Society for Vascular Surgery. J Vasc Surg. 2003; 37(5):1106–1117.
6. Fillinger MF, Raghavan ML, Marra SP, Cronenwett JL, Kennedy FE. In vivo analysis of mechanical wall stress and abdominal aortic aneurysm rupture risk. J Vasc Surg. 2002; 36(3):589–597.
7. Lerderle FA, Johnson GR, Wilson SE, Chute EP, Littooy FN, et al. Prevalence and associations of abdominal aortic aneurysm detected through screening. Aneurysm Detection and Management (ADAM) Veterans affairs Cooperative Study Group. Ann Intern Med. 1997; 126(6):441–449.
8. LeFevre ML. Screening for abdominal aortic aneurysm: U.S. Preventive Services Task Force. Screening for abdominal aortic aneurysm: recommendation statement. Ann Intern Med. 2014; 161(4):281–290.
9. Guirguis-Blake JM, Beil TL, Senger CA, Whitlock EP. Ultrasonography screening for abdominal aortic aneurysms: a systematic evidence review for the U.S. Preventive Services Task Force. Ann Intern Med. 2014; 160(5):321–329.
10. Blaivas M, Theodoro D. Frequency of incomplete abdominal aorta visualization by emergency department bedside ultrasound. Acad Emerg Med. 2004; 11(1):103–105.
11. Bobadilla JL, Suwanabol PA, Reeder SB, Pozniak MA, Bley TA, et al. Clinical implications of non-contrast-enhanced computed tomography for follow-up after endovascular abdominal aortic aneurysm repair. Ann Vasc Surgery. 2013; 27(8):1042–1048.
12. United Kingdom EVAR Trial Investigators. Greenhalgh RM, Brown LC, Powell JT, Thompson SG, et al. Endovascular versus open repair of abdominal aortic aneurysm. N Engl J Med. 2010; 362(20):1863–1871.
13. Lerderle FA, Wislon SE, Johnson GR, Reinke DB, Littooy FN, et al. Immediate repair compared with surveillance of small abdominal aortic aneurysms. N Engl J Med. 2002; 346(19):1437–1444.
14. Ambler GK, Gohel MS, Mitchell DC, Loftus IM, Boyle JR, et al. The Abdominal Aortic Aneurysm Statistically Corrected Operative Risk Evaluation (AAA SCORE) for predicting mortality after open and endovascular interventions. J Vasc Surg. 2015; 61(1):35–43.
15. Anjum A, Von allmen R, Greenhalgh R, Powell JT. Explaining the decrease in mortality from abdominal aortic aneurysm rupture. Br J Surg. 2012; 99(5):637–645.