Abstract
While coronavirus disease 2019 (COVID-19) predominantly involves the respiratory system, it is increasingly recognized that the infection is not restricted to pulmonary system only but other organs are also affected to varying degrees of severity. Gastrointestinal, neurological and cardiac manifestations are reported in addition to dysfunction of hepatic, renal, hematological systems. The wide range of clinical presentations involving multiple organ systems necessitate investigating the causal and association factors of COVID-19 infection. Prompt recognition of the symptoms is necessary for selection of appropriate diagnostic methods and confirmatory tests to reduce delay in initiating the therapy. This manuscript aims to review the spectrum of clinical and imaging aspects of gastrointestinal and thrombo embolic manifestations of COVID-19.
Keywords: coronavirus; COVID-19; SARS-CoV-2; gastrointestinal manifestations; thromboembolism
Full Text
Key message
A broad spectrum of clinical presentations of COVID-19 patients is due to involvement of gastrointestinal system, central nervous system, hepato-renal and musculoskeletal systems in addition to the more extensively known pulmonary infection by the virus. The radiological imaging features indicate the etiopathogenesis of COVID-19 infection. Abdominal cross sectional imaging of intensive care patients often reveals bowel ischemia, gall bladder pathology and vascular occlusions. Pulmonary embolism is another fatal hypercoagulable condition requiring immediate recognition. In addition, cardiac arrhythmia, myocarditis, acute coronary syndromes, hepatocellular injury, hyperglycemia and ketosis are also reported. Isolated non pulmonary clinical presentation should be considered in the diagnostic work up of these patients. Abdominal imaging for other indications should include the lower chest also to look for evidence of COVID-19 infection.
Introduction
The Ministry of Health and Family Welfare, Government of India confirmed a total of 849,553 cases, 534,620 recoveries and 22,674 deaths in the country due to corona virus disease 2019 (COVID-19) pandemic at the time of submission of this manuscript. The corona virus 2 (novel corona virus-2019; COVID-19; SARS-CoV-2) is a genetic consequence of the original SARS-CoV which spread to 29 countries in the years 2002—2004 with an epidemic leaving more than 700 dead [1, 2]. The highly contagious COVID-19 virus was first reported in China spreading rapidly to more than 160 countries. It is increasingly recognised that the respiratory system is not the only target to the virus, but it is a multi system disease because of its affinity for angiotensin converting enzyme 2 (ACE2) receptors resulting in cytokine-mediated immune response and inflammation. The receptors are present in the gastrointestinal system, hepatocytes, vascular endothelial cells, gall bladder, brain, heart and kidneys increasing their vulnerability to infection. Single cell RNA-sequencing demonstrated the organs at risk, such as lung, heart, esophagus, kidney, bladder, and ileum [3]. Decreased immune status because of underlying comorbid factors such as chronic pulmonary disease, diabetes, hypertension, altered renal function triggers the disease manifesting in non pulmonary symptoms of COVID-19 [4, 5].
Extra pulmonary symptoms include headaches, syncope, seizures, confusion, fatigue, myalgia, diarrhea, vomiting, chest pain, acute cardiovascular events, abdominal pain, elevated liver enzymes and acute kidney injury [6]. Hematological involvement manifests as thromboembolic phenomena. These COVID-19 clinical presentations with variable radiologic findings need a broad range of radiologic imaging strategies and this article attempts to present an overview of the atypical imaging spectrum in gastrointestinal and thrombo embolic manifestations of COVID-19.
Gastrointestinal manifestations & imaging
Some patients with COVID-19 infection may experience gastrointestinal symptoms such as abdominal pain, vomiting and diarrhea and a few showing severe acute respiratory syndrome coronavirus (SARS-CoV-2) RNA in the fecal matter. Systematic review and meta-analysis of the current research demonstrated that digestive tract is definitely involved in a significant percentage of patients with COVID-19. Of the 2256 studies from USA, South Korea, Singapore and China, initially generated in the literature search, 37 studies were eligible for inclusion in the meta-analysis. The prevalence rates of diarrhea, nausea/vomiting, and abdominal discomfort/pain were ranging from 6.6 to 9 % with almost similar prevalence in children. SARS-CoV-2 RNA was detected in feces by reverse-transcriptase polymerase-chain-reaction (RT-PCR) in 30 % of these patients though positive RT-PCR in the stool specimen was detected in some asymptomatic patients. The meta-analysis revealed higher prevalence of gastrointestinal (GI) symptoms in patients with severe disease than in those with less severe disease indicating the need for closer monitoring [7, 8].
GI tract involvement as a potential route of transmission is explained by the expression of large amount of ACE2 surface expression in the enterocytes of small and large intestines and the receptor-binding domain on COVID-2 binding to ACE2 with strong affinity [9-11]. Fever, cough and breathlessness are the most recognised and expected symptoms of COVID-19 patients. Those presenting with atypical symptoms may go untested since they do not have the well known respiratory symptoms. It has been also reported that patients undergoing imaging for some other reason may demonstrate lung changes on CT scanning as the first indicators of COVID-19, preceding the illness. Timely recognition of imaging features and treatment of patients with atypical clinical presentations prevents the spread [12, 13]. There is very little published literature on abdominal imaging in COVID-19. In a single centre study of largest number of 224 patients who tested positive for severe COVID-19 disease, abdominal imaging studies were performed (Ultrasound=44, CT=42, MRI=1, radiography=137). Most of these patients who had cross sectional imaging presented with abdominal pain (33%) and sepsis (29%). Also, Nausea / Vomiting, diarrhea, GI bleed, suspected intestinal ischemia were observed as presenting features. Right upper quadrant ultrasound was most commonly performed and majority were admitted in ICU (84%). Elevated LFT, acute kidney injury and source of infection as evidenced by laboratory findings were considered indications for ultrasonography in these patients. Source of infection was noted in 29 % [14].
In this study frank bowel infarction and ischemic mucosal necrosis were observed at laparotomy in two patients each (Table 1). Direct viral infection, small vessel thrombosis, or nonocclusive mesenteric ischemia are possible explanations for the bowel involvement. Systemic coagulopathy is common in critically ill patients which supports the mechanism of complement associated microvascular injury and fibrin thrombi causing ischemic necrosis of the bowel. It has been reported that vascular endothelium may be directly affected by inflammation [15]. Submucosal edema, pneumatosis and arterioles with fibrin thrombi beneath the damaged mucosa were noted at histopathology [16]. Four patients, all in the ICU were detected to have cholestasis on ultrasonography which revealed negative bacterial culture. Interestingly radiograph or ultrasound in this study recognized three of the four patients with pneumatosis or portal venous gas though sensitivity on radiography or ultrasonography is low. The study did not identify portal venous thrombosis despite the hypercoagulable state of these patients (Figures 1-5).
Table 1: CT (plain and contrast enhanced) features of abdomen in COVID-19 in 42 patients.
|
Bowel wall thickening
Colon / rectal thickening
Small bowel wall thickening
Pneumatosis or portal venous gas
Pneumatosis cystoides intestinalis
Perforation of small bowel
|
12
7
5
4
1
1
|

Figure 1: (a) Non-contrast CT brain in a 57-year-old male with COVID-19 shows hypoattenuation in the left MCA territory. (b) CT angiography in the same patient demonstrates occlusion of the left M2 branch (arrow). There is no distal reformation.

Figure 2: (a) Unenhanced CT scan of head demonstrates symmetric low attenuation within the bilateral medial thalami (arrows). (b) Axial CT venogram demonstrates patency of the cerebral venous vasculature, including the internal cerebral veins (arrows).
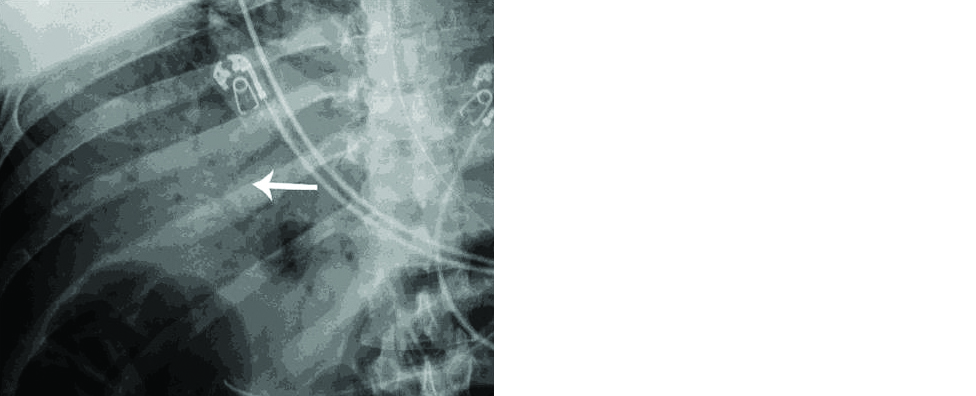
Figure 3: Contrast-enhanced cranial 1.5T MRI examination of a 59-year old intubated male patient with altered mental status despite tapering of sedoanalgesia. Axial FLAIR images at level of midbrain (a) and centrum semiovale (b) demonstrate prominent symmetric white matter hyperintensity and right frontal cortical hyperintensity. There is also prominent linear hyperintensity within frontal sulci. Axial b2000 DWI (c) shows frontal increased signal with corresponding low ADC (images not provided). Axial T1WI (d) shows right frontal sulcal effacement. Post-contrast T1WI (e) shows mild pial-subarachnoid enhancement. Axial SWI at the level of corona radiata (f) and centrum semiovale (g) demonstrates blooming artifact in the frontal sulci. Post-contrast FLAIR (h) depicts the bilateral leptomeningeal enhancement. ADC apparent diffusion coefficient; FLAIR fluid-attenuated inversion recovery; SWI susceptibility weighted image.

Figure 4: Axial FLAIR in four different COVID-19 patients. (a) 58-year old man with impaired consciousness: FLAIR hyperintensities located in the left medial temporal lobe. (b) 66-year old man with impaired consciousness: FLAIR ovoid hyperintense lesion located in the central part of the splenium of the corpus callosum. (c) 71-year old woman with pathological wakefulness after sedation: extensive and confluent supratentorial white matter FLAIR hyperintensities (arrows). Association with leptomeningeal enhancement (stars), (d) 61-year old man with confusion: hyperintense lesions involving both middle cerebellar peduncles.

Figure 5: 65-year-old man with pathological wakefulness after sedation. Non-confluent multifocal white matter hyperintense lesions on FLAIR and diffusion, with variable enhancement, and hemorrhagic lesions. Axial Diffusion (a, b), Apparent Diffusion Coefficient (ADC) map (c), axial FLAIR (d, e), sagittal FLAIR (f), axial Susceptibility weighted imaging (SWI) (g), and postcontrast T1 weighted MR images (h). Multiple nodular hyperintense Diffusion and FLAIR lesions localized in the white matter including the corpus callosum (f). Some of them (white arrow) are associated with reduced ADC corresponding to cytotoxic edema (c). Other lesions are located next to the lenticular nucleus (cross) (e, g, h), with hemorrhagic changes (g), and enhancement after contrast administration.
Pulmonary embolism
Recent literature indicates significant prevalence of pulmonary embolism in COVID-19 patients compromising the pulmonary function which is already diminished significantly. Many of these patients are severely ill and have acute kidney injury and multi-organ failure. Clinical reports and recent European cohort studies are published on pulmonary embolism correlating the increased serum proteins such as D-dimer and increased mortality. D-dimer values showed a statistically significant difference between CT pulmonary angiography (CTPA) positive and CTPA negative subgroups indicating the severity of pulmonary obstruction. In a cohort study, CTPA is indicated for the patients who are in respiratory distress, tachycardia, elevated D-Dimer, lower extremity swelling, hypoxia, chest pain and unspecified indications. The sensitivity and specificity of CTPA positive result depend on the threshold value of the D-dimer, a clear indicator of hypercoagulable state. Other laboratory values of platelet counts, serum ferritin, C-reactive protein, fibrinogen did not correlate with CTPA results statistically [17-20]. Recent studies confirmed the association of COVID-19 and pulmonary embolism, with a high prevalence of peripheral venous thrombosis in 56% and pulmonary embolism in 23% of ICU patients, in spite of prophylaxis and therapeutic anticoagulation compared with normal population. A large single centre study concluded that 37.1% of COVID positive patients were diagnosed pulmonary embolism on CTPA and D-dimer can be used to stratify patients regarding PE risk and severity [21-24]. On CTPA, it is difficult to distinguish thrombus from embolism and whether the central CTPA intravascular defects migrate distally into subsegmental vessels (Figures 6-9). Thrombosis secondary to vascular injury and hypercoagulable state is a proposed mechanism in these patients. The higher rate of DVT (53%) compared to 29% in the pre-COVID cohort, suggests a thromboembolic component for the intravascular defects on CTPA. Vascular enlargement within the areas of lung opacity are reported as a feature of COVID-19 on high resolution CT examinations of chest. This may be appreciated in milder and earlier presentations of pulmonary COVID-19 when HRCT is performed as a routine diagnostic test [25, 26]. A subsegmental vessel greater than 3 mm in diameter is considered as definite vascular enlargement. The enlargement may be related to regional hyperemia and inflammation. In addition, reverse halo sign is noted as a combination of consolidation and ground-glass opacity [27-29].
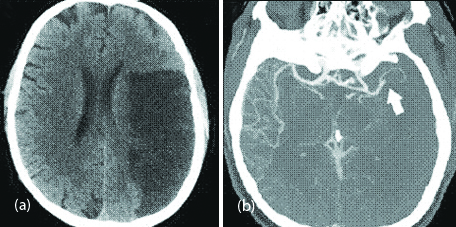
Figure 6: 57-year-old man with pathological wakefulness after sedation. Extensive and isolated white matter microhemorrhages. Axial Susceptibility weighted imaging (SWI) (a-d): Multiple microhemorrhages mainly affecting the subcortical white matter, corpus callosum, internal capsule, and cerebellar peduncles.

Figure 7: 54-year-old man with pathological wakefulness after sedation. Non-confluent multifocal white matter hyperintense lesions on FLAIR and diffusion, with variable enhancement. Axial Diffusion (a, b), Apparent Diffusion Coefficient (ADC) map (c, d), axial postcontrast FLAIR (e, f), and postcontrast T1 weighted MR images (g, h). Multiple nodular hyperintense Diffusion and FLAIR subcortical and corticospinal tracts lesions, with very mild mass effect on adjacent structures. The lesions present a center with an elevation of ADC corresponding to vasogenic edema and a peripheral ring of reduced ADC corresponding to cytotoxic edema (c, d). After contrast administration, small areas of very mild enhancement are detected (g, h).
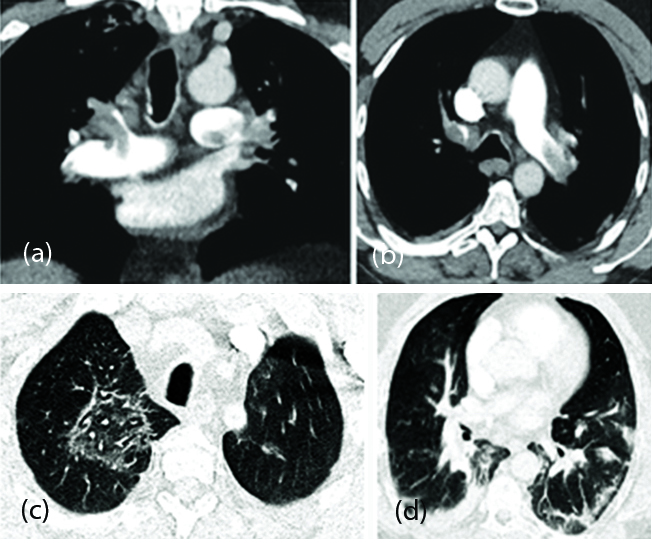
Figure 8: (a, b, e, f) T2-weighted fluid-attenuated inversion recovery MRI scans demonstrate hyperintensity within the bilateral medial temporal lobes and thalami (arrows), with evidence of hemorrhage indicated by, (c, g) hypointense signal (arrows) on susceptibility-weighted images and, (d, h) rim enhancement (arrows) on contrast material–enhanced images.
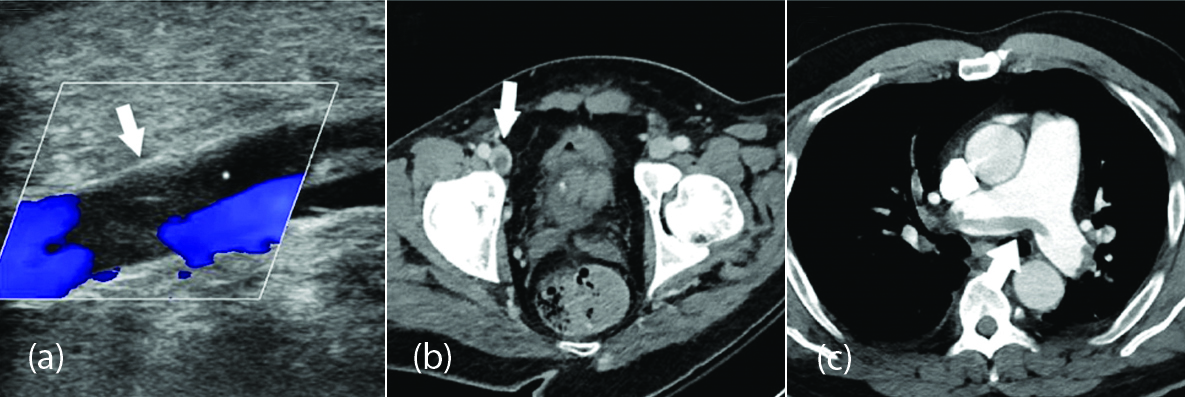
Figure 9: Axial (a) and coronal (b) CT of the abdomen and pelvis with IV contrast in a 57-year-old man with a high clinical suspicion for bowel ischemia. There was generalized small bowel distension and segmental thickening (arrows), with adjacent mesenteric congestion (thin arrow in b), and a small volume of ascites (* in B). Findings are nonspecific but suggestive of early ischemia or infection.
Mastora obstruction scoring system helps grading the degree of obstruction of pulmonary vessels (Grade 1 to 5 : 25% - 100%) main pulmonary artery (MPA), right pulmonary artery (RPA), left pulmonary artery (LPA), both interlobar arteries, lobar, and segmental pulmonary arteries. Positive correlation between the obstruction scores (ObstCenRatio and ObstTotRatio) and D-dimer levels was noted in severe pulmonary embolism [30]. It should be noted though CT pulmonary angiography (CTPA) is the mainstay to diagnose pulmonary embolism there is a potential risk of nephrotoxicity due to contrast administration particularly in the acutely-ill and multi-organ failure patients with possible acute kidney injury.
Conclusion
COVID-19 is a progressive multisystem disease manifesting widely in different organs. Mechanism of dissemination of the virus into extrapulmonary systems causing multiorgan injury is not yet clear. Although widespread expression of ACE2, receptor for SARS-CoV-2 is established, the causal effect needs further studies. Understanding of the imaging features in relevant extrapulmonary organ systems is essential for diagnosis, treatment and prevention of complications.
Acknowledgements
Acknowledgement to the source of the figures: Fig. 1 (Manna et al., https://pubs.rsna.org/doi/10.1148/ryct.2020200210); Fig. 2&8 (Poyiadji et al., https://pubs.rsna.org/doi/10.1148/radiol.2020201187); Fig. 3 (Kandemirli et al., https://pubs.rsna.org/doi/10.1148/radiol.2020201697); Fig. 4-7 (Kremer et al., https://pubs.rsna.org/doi/10.1148/radiol.2020202222); Fig. 9 (Bhayana et al., https://pubs.rsna.org/doi/10.1148/radiol.2020201908).
Conflicts of interest
Author declares no conflicts of interest.
References
[1] Ministry of Health and Family Welfare. Accessed on 12 July 2020 from: https://www.mohfw.gov.in/
[2] WHO. "China's latest SARS outbreak has been contained, but biosafety concerns remain – Update 7". World Health Organization. Archived from the original on 12 February 2020. Retrieved 14 February 2020. Accessed on 18 May 2004.
[3] Zou X, Chen K, Zou J, Hao J, Han Z. Single-cell RNA-seq data analysis on the receptor ACE2 expression reveals the potential risk of different human organs vulnerable to 2019-nCoV infection. Front Med. 2020; 14:185–192.
[4] Hosseiny M, Kooraki S, Gholamrezanezhad A, Reddy S, Myers L. Radiology perspective of coronavirus disease 2019 (COVID-19): lessons from severe acute respiratory syndrome and Middle East respiratory syndrome. Am J Roentgenol. 2020; 214(5):1–5.
[5] Kooraki S, Hosseiny M, Myers L, Gholamrezanezhad A. Coronavirus outbreak: what the Department of Radiology should know. J Am Coll Radiol. 2020; 17(4):447–451.
[6] Rodriguez-Morales AJ, Cardona-Ospinaa JA, Gutierrez-Ocampoa E, Villamizar-Peñaa R, Holguin-Rivera Y, et al. Clinical, laboratory and imaging features of COVID-19: A systematic review and meta-analysis. Travel Med Infect Dis. 2020; 34:101623.
[7] Rokkas T. Gastrointestinal involvement in COVID-19: a systematic review and meta-analysis. Annals of Gastroenterology. 2020; 33:1–11.
[8] Tang A, Tong Z, Wang H, Dai YX, Li KF, et al. Detection of novel coronavirus by RT-PCR in stool specimen from asymptomatic child, China. Emerg Infect Dis. 2020; 26(6):1337–1339.
[9] Wu Y, Guo C, Tang L, Hong Z, Zhou J, et al. Prolonged presence of SARS-CoV-2 viral RNA in faecal samples. The Lancet Gastroenterology & Hepatology. 2020; 5(5):434–435.
[10] Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020; 181(2):271–280.e8.
[11] Walls AC, Park YJ, Tortorici MA, Wall A, McGuire AT, et al. Structure, function, and antigenicity of the SARS-CoV-2 spike glycoprotein. Cell. 2020; 181(2):281–292.e6.
[12] Siegel A, Chang PJ, Jarou ZJ, Paushter DM. Lung Base Findings of Coronavirus Disease (COVID-19) on Abdominal CT in Patients With Predominant Gastrointestinal Symptoms. American Journal of Roentgenology. 2020; 215:607–609.
[13] Lin C, Ding Y, Xie B, Sun Z, Li X, et al. Asymptomatic novel coronavirus pneumonia patient outside Wuhan: the value of CT images in the course of the disease. Clin Imaging. 2020; 63:7-9.
[14] Bhayana R, Som A, Li MD, Carey DE, Anderson MA, et al. Abdominal Imaging Findings in COVID-19: Preliminary Observations. Radiology. 2020; 297(1):E207–E215.
[15] Caruso D, Zerunian M, Polici M, Pucciarelli F, Polidori T, et al. Chest CT Features of COVID-19 in Rome, Italy. Radiology. 2020; 296(2):E79–E85.
[16] Magro C, Mulvey JJ, Berlin D, Nuovo G, Salvatore S, et al. Complement associated microvascular injury and thrombosis in the pathogenesis of Severe COVID-19 infection: A report of five cases. Transl Res. 2020; 220:1–13.
[17] Kaminetzky M, Moore W, Fansiwala K, Babb JS, Kaminetzky D, et al. Pulmonary Embolism on CTPA in COVID-19 Patients. Radiology: Cardiothoracic Imaging. 2020; 2(4). Available from: https://pubs.rsna.org/doi/pdf/10.1148/ryct.2020200308
[18] Zhou F, Yu T, Du R, Fan G, Liu Y, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020; 395(10229):1054–1062.
[19] Llitjos JF, Leclerc M, Chochois C, Monsallier JM, Ramakers M, et al. High incidence of venous thromboembolic events in anticoagulated severe COVID-19 patients. J Thromb Haemost. 2020; 18(7):1743–1746.
[20] Leonard-Lorant I, Delabranche X, Severac F, Helms J, Pauzet C. Acute Pulmonary Embolism in COVID-19 Patients on CT Angiography and Relationship to D-Dimer Levels. Radiology. 2020; 296(3):E189–E191.
[21] Cheng Y, Luo R, Wang K, Zhang M, Wang Z, et al. Kidney disease is associated with in-hospital death of patients with COVID-19. Kidney Int. 2020; 97(5):829–838.
[22] Simpson S, Kay FU, Abbara S, Bhalla S, Chung JH, et al. Radiological Society of North America Expert Consensus Statement on Reporting Chest CT Findings Related to COVID-19. Endorsed by the Society of Thoracic Radiology, the American College of Radiology, and RSNA. Radiology: Cardiothoracic Imaging 2020; 2(2):e200152.
[23] Zhao W, Zhong Z, Xie X, Yu Q, Liu J. Relation Between Chest CT Findings and Clinical Conditions of Coronavirus Disease (COVID-19) Pneumonia: A Multicenter Study. AJR Am J Roentgenol. 2020; 214(5):1072–1077.
[24] Li Y, Xia L. Coronavirus Disease 2019 (COVID-19): Role of Chest CT in Diagnosis and Management. AJR Am J Roentgenol. 2020; 214(6):1280–1286.
[25] Caruso D, Zerunian M, Polici M, Pucciarelli F, Polidori T, et al. Chest CT Features of COVID-19 in Rome, Italy. Radiology. 2020; 296(2):E79–E85.
[26] Mastora I, Remy-Jardin M, Masson P, Galland E, Delannoy V, et al. Severity of acute pulmonary embolism: evaluation of a new spiral CT angiographic score in correlation with echocardiographic data. Eur Radiol. 2003; 13(1):29–35.
[27] Zaim S, Chong JH, Sankaranarayanan V, Harky A. COVID-19 and multi-organ response. Curr Probl Cardiol. 2020; 45(8):100618.
[28] Petrilli CM, Jones SA, Rajagopalan H. Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: prospective cohort study. BMJ. 2020; 369:m1966.
[29] Cummings MJ, Baldwin MR, Abrams D, Jacobson SD, Meyer BJ, et al. Epidemiology, clinical course, and outcomes of critically ill adults with COVID-19 in New York City: a prospective cohort study. Lancet. 2020; 395(10239):1763–1770.
[30] Chu KH, Tsang WK, Tang CS, Lam MF, Lai FM, et al. Acute renal impairment in coronavirus-associated severe acute respiratory syndrome. Kidney Int. 2005; 67(2):698–705.