Full Text
Introduction
Since the initial spread of this new illness, known as coronavirus disease 2019 (COVID-19), many patients, have been hospitalized with respiratory symptoms [1]. The clinical spectrum is broad, including asymptomatic infection, mild upper respiratory tract disease and severe interstitial pneumonia with respiratory failure requiring oxygenation support or intubation [2, 3].
High resolution computed tomography (HRCT) is the most sensitive radiological technique for the diagnosis of COVID-19, showing diffuse lung alterations ranging from ground-glass opacities to parenchymal consolidations; several radiological patterns are observed at different times throughout the disease course [4, 5].
Pathophysiology
Structural and functional analysis showed that the spike for SARS-CoV-2 bound to ACE2. ACE2 expression was high in lung, heart, ileum, kidney and bladder. In lung, ACE2 was highly expressed on lung epithelial cells [6-8].
The symptom of patients infected with SARS-CoV-2 ranges from minimal symptoms to severe respiratory failure with multiple organ failure. On HRCT scan, the characteristic pulmonary ground glass opacification can be seen even in asymptomatic patients. Because ACE2 is highly expressed on the apical side of lung epithelial cells in the alveolar space, this virus can likely enter and destroy them. This matches with the fact that the early lung injury was often seen in the distal airway. Epithelial cells, alveolar macrophages and dendritic cells (DCs) are three main components for innate immunity in the airway. DCs reside underneath the epithelium. Macrophages are located at the apical side of the epithelium. DCs and macrophages serve as innate immune cells to fight against viruses till adaptive immunity is involved [8-12].
In addition to respiratory symptoms, thrombosis and pulmonary embolism have been observed in severe diseases. This is in line with the finding that elevated d-dimer and fibrinogen levels were observed in severe disease. The function of the endothelium includes promotion of vasodilation, fibrinolysis, and anti-aggregation. Because endothelium plays a significant role in thrombotic regulation, hypercoagulable profiles seen in severe disease likely indicate significant endothelial injury. Endothelial cells also express ACE2. Of note, the endothelial cells represent one third of lung cells. Microvascular permeability as a result of the endothelial injury can facilitate viral invasion [13-15].
Imaging evolution of COVID -19
Chest x-ray
Chest x-ray alterations were defined according to the Fleischner Society’s nomenclature, available in the Glossary of Terms for Thoracic Imaging [16]:
• Reticular alteration, as a collection of innumerable small linear opacities that, by summation, produce an appearance of a net;
• Consolidation, as a homogeneous increase in pulmonary parenchymal attenuation that obscures the margins of the vessels and airway walls (Figure 1);
• Ground-glass opacity (GGO), as an area of hazy, increased lung opacity, usually extensive, within which margins of pulmonary vessels may be indistinct (Figure 2a,b).
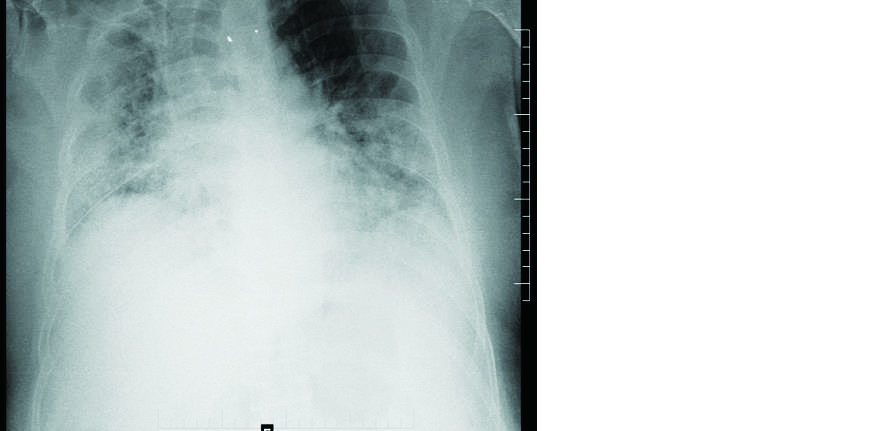
Figure 1: Chest x-ray showing consolidation bilaterally, predominantly in bilateral lower lobes.

Figure 2: (a) A 80-year-old patient with chief complaints of cough, First chest x-ray was done which shows subtle subpleural opacities bilaterally which on HRCT chest confirmed as subpleural GGO typical of COVID-19 (Figure 2b). (b) On HRCT chest confirmed as subpleural GGO typical of COVID-19.
Early changes of COVID-19 pneumonia may not be always evident on the chest x-ray. However in, moderate to severe disease, chest x-ray is helpful for follow-up to see the disease progression.
HRCT chest
The two main and common signs of COVID-19 on HRCT images are: (1) Ground glass opacities (GGO’s), (2) Consolidation, with or without vascular enlargement, interlobular septal thickening, air bronchogram sign, and air trapping, and the lesions are predominantly peripheral and subpleural.
In the first 5 days from the onset of the symptoms, reticular alteration is slightly more frequent than GGO, while after this period, GGOs become the predominant finding. Consolidation is constantly less frequent than the other two alterations, especially in the early phase of the disease. Early alterations are predominantly reticular, intermediate alterations are predominantly GGO with a period of overlap between these two, whereas consolidations increase in the late phase [17-19].
Consolidations are more common in patients with severe progressive disease. These CT findings may correspond to viscous secretions seeping through the pulmonary alveoli described in the autopsy reports [20] and suggest acute respiratory distress syndrome (ARDS).
ARDS characterized by an acute, diffuse, inflammatory lung injury, leading to an increased alveolar capillary permeability, is a strong predictor of mortality, reflecting the severity of respiratory failures [21].
Early changes in HRCT
The early CT findings (3 days after onset) are characterized as predominately subpleural GGO’s with posterior basal predominance for patients with different disease severity. There is a tendency of subpleural distribution for pulmonary opacities, consistent with the underlying pathogenic pattern that distal bronchioles as well as alveolar epithelial cells are impaired in viral pneumonia [17-19]. The posterior basal predominance shown in the heat map may due to the predilection of atelectasis posteriorly, corresponding to heterogeneous pulmonary opacity distribution in patients with acute respiratory distress syndrome [22]. Among all subpleural areas, the posteroinferior area of the lung shows leading frequency of opacity distribution which may due to the anatomical characteristics of the bronchus: more vertical for lower lobes compared with the other lobes. This makes the lower lobes more susceptible to pathogen attack.
Dynamic evolution of COVID-19 on chest computed tomography: experience from Jiangsu Province of China – Wang YC et al [23] (Figure 3).
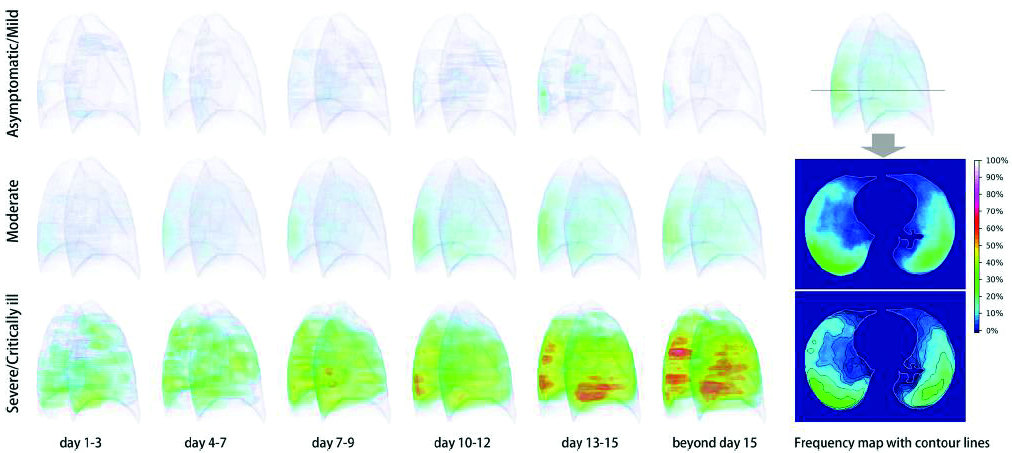
Figure 3: Three-dimensional heat maps show the frequency of location of pulmonary opacities in COVID-19 from the onset of symptoms (day 1) to beyond day 15. Asymptomatic/ mild patients have trace opacities, resolving partially after 15 days (top row). Moderately ill patients have more opacities and peaked on days 13–15, predominantly located in the posteroinferior subpleural area (middle row). Severe/ critically ill patients have the most prominent opacities and continue to progress beyond day 15 (bottom row). Axial frequency map shows a typical pattern of posterior subpleural distribution of the opacities with decreased frequency in the anterior and medial subpleural areas.
As the disease progresses, more inflammatory cells infiltrate the alveoli and interstitial space, followed by diffuse alveolar damage and hyaline membrane formation.
In the early stage of the disease, (2-5 days) HRCT images predominantly show unilateral or bilateral ground-glass opacities accompanied with enlarged small vessels. Sometimes, ground-glass with focal consolidation can be seen. In terms of distribution, the lower lobe and peripheral (i.e., sub-pleural area) are most commonly affected. The early involvement of the peripheral and sub pleural regions (Figure 4a, b) is related to the pathological mechanism of viral pneumonia, which causes early involvement of bronchioles and parenchyma around the bronchioles, and then involves the whole pulmonary lobule and causes diffuse alveolar damage [24, 25].

Figure 4: (a) HRCT chest showing subpleural GGO with mild reticulations in mild phase. (b) HRCT chest showing subpleural GGO with increased reticulation in initial phase.
Late changes
As the disease progresses, (5-9 days ) CT images show enlargement of opacities and an increased number of new ground-glass opacities affecting more and larger regions of multiple lung lobes (Figure 5a, b). Reticular interstitial thickening which is due to thickening of the pulmonary interstitium and the inter and intralobular septae may be seen. The prominently thickened interlobular septal lines superimposed on the GGO’s and /or consolidation give the appearance of irregularly paved stones termed as “Crazing paving” appearance. This pattern is commonly seen in subacute and chronic phase of infection.

Figure 5: (a) HRCT CHEST showing subpleural and peribronchial GGO with increased reticulations and thick atelectatic bands in early late phase. (b) HRCT chest shows subpleural and peribronchial ground glass densities with increased reticulations and consolidation in moderate disease.
Furthermore (9-15 and more) some of the original ground-glass opacities begin to consolidate. Thus, ground-glass opacities and consolidation are commonly seen. A reverse Halo sign or Atoll sign is described in this phase when there is central GGO surrounded by denser peripheral consolidation. Pathologically this correlates with central alveolar inflammation and debris with peripheral areas of organising inflammation in the interstitium and distal air space. This appearance may also be seen in the recovery phase of COVID-19 pneumonia [5]. Airway changes including bronchial wall thickening, bronchiectasis and brochiolectasis have been described in 10-20% of cases in few studies [26]. Development of pleural effusions and lymphadenopathy is rare and if present suggests worsening of disease or superimposed heart failure.
At this stage, if the patients does not receive effective treatment promptly or if the immunity is low, COVID-19 pneumonia may be life-threatening. HRCT images may show diffuse opacities in both lungs (Figure 6).

Figure 6a, b: A 48-year-old patient HRCT chest shows subpleural and peribronchial ground glass opacities with increased reticulations and consolidation suggestive of COVID-19 pneumonia- with moderate changes.
In severe cases so-called white lung manifestation (Figure 7) is seen. If the patient receives effective treatment or if immunity is enhanced, pneumonia will be gradually absorbed and the opacity diminishes. The disease will improve, possibly leaving the lung with fibrous stripes and thickened pleura [24, 27] (Figure 8).

Figure 7: Severe changes of COVID-19 pneumonia on HRCT chest also called white lungs.
On follow up after about 4 weeks HRCT chest shows resolving phase as there is decrease in the ground glass opacities and changes of fibrosis seen as subpleural bands are present.
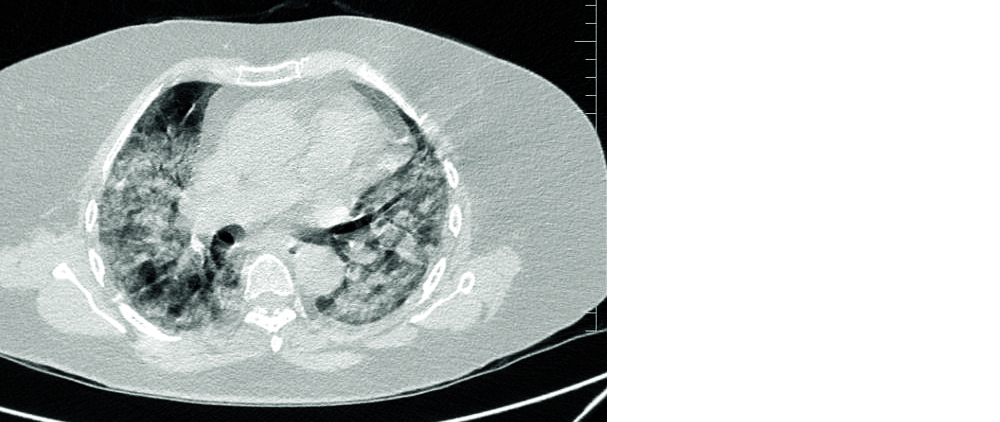
Figure 8: HRCT chest - Severe changes of COVID-19 pneumonia seen as extensive multifocal GGO’s and consolidation with air bronchogram in subpleural and peribronchial distribution in critically ill patient.
COVID-19 CO-RADS classification
In March 2020, the "COVID-19 standardized reporting working group" of the Dutch Association for Radiology (NVvR) proposed a CT scoring system for COVID-19 (Table 1). They called it CO-RADS (COVID-19 Reporting and Data System) to ensure CT reporting is uniform and replicable. This assigns a score of CO-RADS 1 to 5, dependent on the HRCT findings. In some cases a score of 0 or 6 may need to be assigned as an alternative. If the HRCT is uninterpretable then it is CO-RADS 0, and if there is a confirmed positive RT-PCR test then it is CO-RADS -6 [28, 29].
Table 1: CT scoring system - CO-RADS (COVID-19 reporting and data system).
|
|
Level of suspicion for pulmonary involvement of COVID-19
|
Summary
|
|
CO-RADS 0
|
Not interpretable
|
Scan technically insufficient for assigning a score
|
|
CO-RADS 1
|
Very low
|
Normal or Non-infectious
|
|
CO-RADS 2
|
Low
|
Typical for other infection but not COVID-19
|
|
CO-RADS 3
|
Equivocal/ unsure
|
Features compatible with COVID-19, but also other diseases
|
|
CO-RADS 4
|
High
|
Suspicious for COVID-19
|
|
CO-RADS 5
|
Very high
|
Typical for COVID-19
|
|
CO-RADS 6
|
Proven
|
RT-PCR positive for SARS-CoV-2
|
CT severity index [30]
A semi-quantitative CT severity scoring proposed by Pan et al [31] can be calculated per each of the 5 lobes considering the extent of anatomic involvement, as follows: 0, no involvement; 1, < 5% involvement; 2, 5–25% involvement; 3, 26–50% involvement; 4, 51–75% involvement; and 5, > 75% involvement.
The resulting global CT score is the sum of each individual lobar score (0 to 25). When present, related features such as fibrosis, subpleural lines, reversed “halo sign,” pleural effusion, and lymphadenopathy are also described.
A CT score ≥ 18 has shown to be highly predictive of patient’s mortality in short-term follow-up.
Differential diagnosis for ground glass and crazy paving [32, 33]
Pulmonary edema
It is the abnormal accumulation of fluid in the extravascular compartments of the lung, and, may be classified as increased hydrostatic pressure edema seen in heart failure, permeability edema with diffuse alveolar damage (DAD) as seen in ARDS, drug induced lung injury or ingestion of or mixed edema seen in patients with stroke, status epilepticus and subarachnoid hemorrhage (Figure 9). Findings of interstitial pulmonary edema are GGO, bronchovascular and interlobular septal thickening. Alveolar edema may manifest as airspace consolidation in addition to the above findings. Pleural effusions are a frequent accompanying finding in cardiogenic pulmonary edema, which is rarely seen in COVID-19 pneumonia.
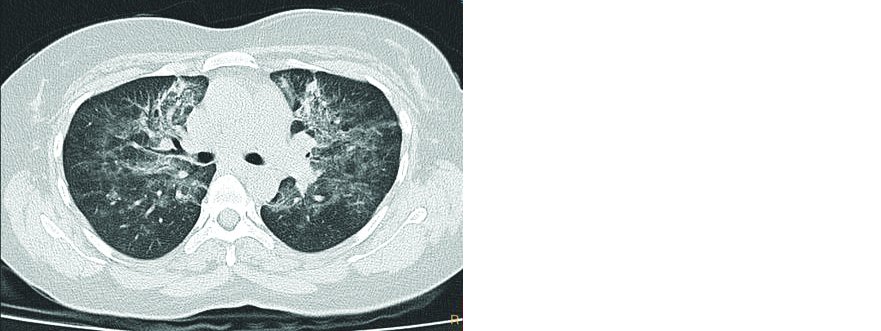
Figure 9: HRCT chest showing extensive ground-glass opacities predominantly in perihilar region suggestive of pulmonary edema.
Pulmonary hemorrhage syndromes
This includes a wide spectrum of diseases, including idiopathic pulmonary hemosiderosis, Wegener granulomatosis, Churg-Strauss syndrome, Goodpasture syndrome, collagen-vascular disease (systemic lupus erythematosus, rheumatoid arthritis, systemic sclerosis, polymyositis, and mixed connective-tissue disease. HRCT manifestations are symmetric acinar and ground-glass opacities or attenuation and the crazy-paving pattern (Figure 10).
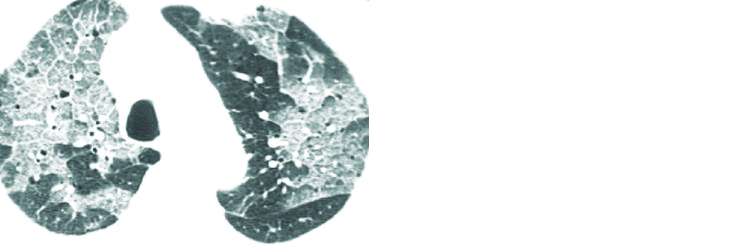
Figure 10: HRCT chest showing diffuse ground glass attenuation and crazy paving pattern bilaterally in a patient with Goodpasture syndrome.
Diffuse alveolar hemorrhage (DAH)
It occurs due to passage of blood into the alveoli.
• Imaging findings depend upon the chronicity of the process.
• Initially DAH may present with GGOs. After two to three days intralobular and smooth interlobular septal thickening superimpose on areas of GGOs and may sometimes give rise to a crazy-paving pattern (Figure 11).
• In the chronic stages, the GGOs typically recede and there maybe residual centrilobular nodules.

Figure 11: HRCT chest showing patchy ground glass densities with interlobular septal thickening in bilateral lungs secondary to diffuse alveolar haemorrhage.
Hypersensitivity pneumonitis (HP) or extrinsic allergic alveolitis (EAA)
In acute phase homogeneous ground-glass opacity: bilateral and symmetric but sometimes patchy and concentrated in the middle part and base of the lungs or having a bronchovascular distribution may be seen. Ground-glass opacity usually represents chronic interstitial inflammation but occasionally may be caused by fine fibrosis or organizing pneumonia.
In the fibrotic stage there is bilateral predominantly perihilar fibrosis with mid zone predominance. The distinct feature of HP is mosaic attenuation of the lungs. This pattern represents geographical areas of high attenuation interspersed with areas of low attenuation due to air trapping (Figure 12).
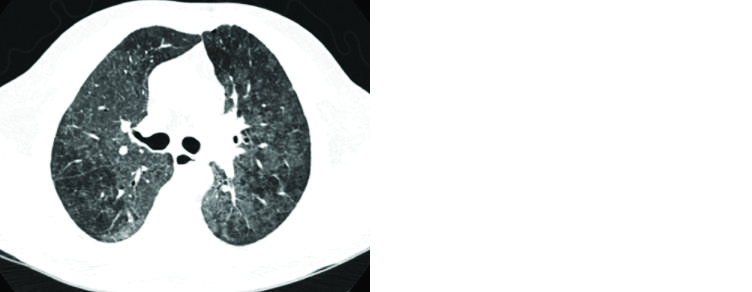
Figure 12: HRCT chest showing diffuse peribronchial ground glass opacities with perihilar fibrosis.
Nonspecific interstitial pneumonia (NSIP)
Bilateral pulmonary opacities, which are mainly localized in the middle and lower zones, are the predominant feature. The most common HRCT abnormality (80% of cases) is ground-glass attenuation. The ground-glass attenuation is usually bilateral and symmetric with a tendency to subpleural and basal predominance (Figure 13). Other findings include consolidation and irregular reticular lines that can be superimposed on a background of ground-glass attenuation. Honeycombing is typically absent.
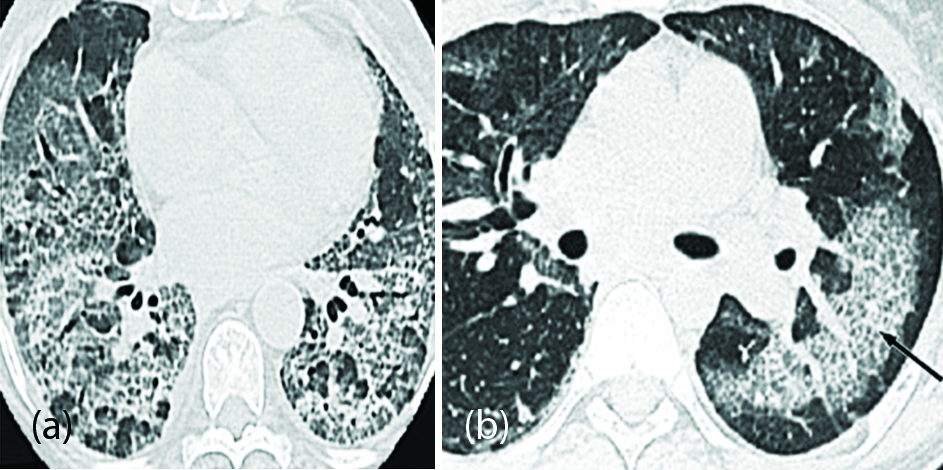
Figure 13a, b: HRCT Chest showing diffuse subpleural and peribronchial ground glass attenuation bilaterally with increased reticular bands in a case of NSIP.
Pneumocystis carinii pneumonia (PCP)
PCP is a common pulmonary infection in the severely immunocompromised patient. Symptoms include dry cough, dyspnea, and low-grade fever. CT usually reveals scattered ground-glass attenuation that can be associated with interlobular septal thickening (Figure 14).
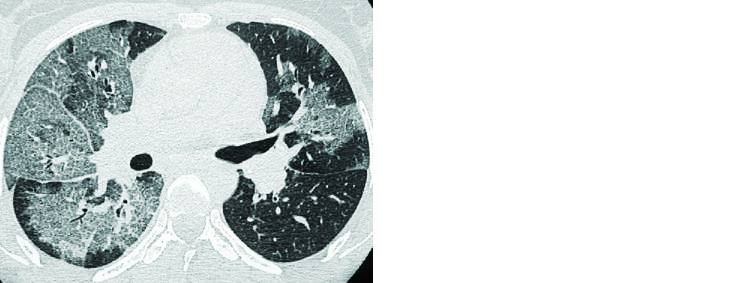
Figure 14: HRCT chest showing extensive ground glass attenuation with interlobular septal thickening predominantly in right lung in a post - renal transplant patient.
Protein alveolar proteinosis
Bilateral, symmetric alveolar consolidation or ground-glass opacity, particularly in a perihilar or hilar distribution resembling pulmonary edema. High-resolution CT typically shows diffuse ground-glass attenuation with superimposed intra- and interlobular septal thickening, often in polygonal shapes representing the secondary pulmonary lobule (Figure 15).

Figure 15: HRCT in a case of pulmonary alveolar proteinosis. Chest showing increased reticulations with interlobar septal thickening seen bilaterally.
Extrapulmonary manifestations of COVID-19 [34]
Blood hypercoagulability is a common finding among hospitalized patients with COVID-19. The effect of COVID-19 on pulmonary coagulation and the resulting development of PTE could be due to proinflammatory cytokines, endothelial dysfunction and hypoxia. Several papers report high incidence of PTE in COVID-19, ranging from 22% to 39%. Pulmonary thromboembolism (PTE) is frequently observed in COVID-19 patients, mainly involving the segmental (90%) and sub-segmental arteries (61%) of pulmonary segments affected by a consolidation pattern (67.6%) (Figure 16). Patients with more severe COVID-19 lung disease (higher CT lesion score, d-dimer, LDH and CRP) tend to be more affected by PTE [35].
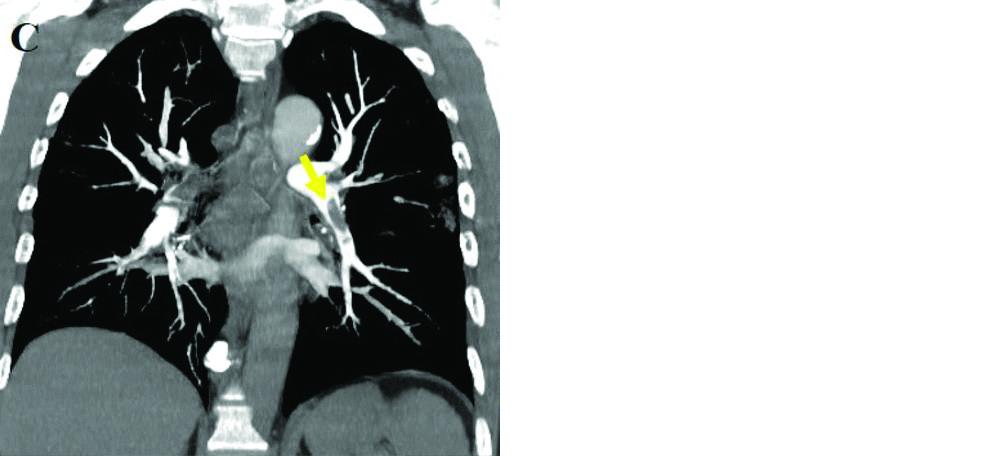
Figure 16: CT pulmonary angiogram showing thrombus in the left lower lobe artery extending into segmental and subsegmental arteries in COVID-19 positive patient.
Gastrointestinal complications [34]
Feco-oral transmission is an alternative route for COVID-19 spread. The radiologic manifestation of these findings can be diarrhea state and ileus pattern on abdominal radiographs and distended fluid filled small and large bowel loops with mural post-contrast enhancement and surrounding stranding on CT (Figure 17).
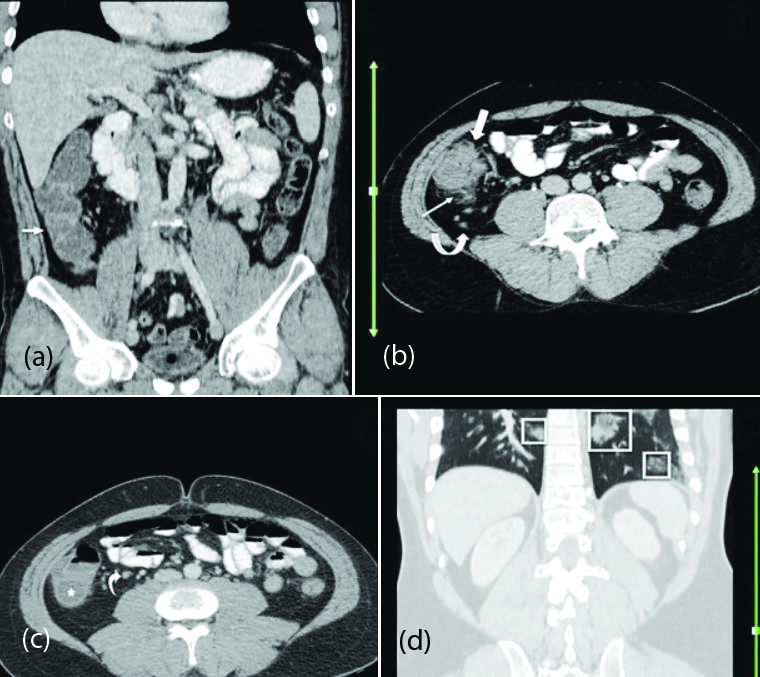
Figure 17: CECT abdomen (a-c) showing fluid distended bowel loops and wall thickening at ileocecal junction in COVID-19 positive patient. (d) Patchy areas of subpleural and peribronchial GGO’S in the posterior basal segment of both lungs.
Vascular abnormalities
Vascular enlargement on patients' chest HRCT is the major finding referring to luminal dilation/engorgement or mural thickening of pulmonary vessels.
Neurological findings
Coronaviruses may enter the CNS through the bloodstream or neuronal retrograde route. This may lead to meningitis and encephalitis with associated morbidity and risk of mortality. Although viral encephalitis may remain undiagnosed due to subtle or no symptoms. Due to hypercoagulable state incidence of stroke are reported in some cases, even in young population.
Cardiac manifestations [34]
ACE1 as a target receptor for SARS-CoV-2 is significantly expressed in the heart. This transmembrane aminopeptidase involved in the development of hypertension and heart function performs an extremely critical role in the cardiovascular system. Therefore, the possibility of cardiovascular injury or myocarditis should be considered in COVID-19 patients presenting with cardiovascular symptoms. Cardiac MRI is a helpful investigation to look for changes of myocarditis, which when present, are seen as patchy areas of mid myocardial enhancement on late gadolinium scans.
Conclusions
In conclusion, HRCT findings in patients with COVID-19 pneumonia are mixed and have a diverse pattern with both lung parenchymal and interstitial involvement. GGO’s with reticular pattern with bilateral, multilobar involvement and peripheral distribution and predilection for the posterior lobes are the hallmark of early disease. HRCT signs of aggravation and repair co-exist in patients with advanced disease. HRCT plays an important role in triage of patients with suspected COVID-19 pneumonia and also in determining the severity of disease. It helps in prediction of disease outcome and is a problem solver in patients with multiple comorbidities.
Conflicts of interest
Authors declare no conflicts of interest.
References
[1] McMichael TM, Currie DW, Clark S, Pogosjans S, Kay M, et al. Epidemiology of COVID-19 in a long-term care facility in King County, Washington. N Engl J Med. 2020; 382(21):2005–2011.
[2] Zhou F, Yu T, Du R, Fan G, Liu Y, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020; 395(10229):1054–1062.
[3] Poston JT, Patel BK, Davis AM. Management of critically ill adults with COVID-19. JAMA. 2020; 323(18):1839–1841.
[4] Shi H, Han X, Jiang N, Cao Y, Alwalid O, et al. Radiological findings from 81 patients with COVID-19 pneumonia in Wuhan, China: a descriptive study. Lancet Infect Dis. 2020; 20(4):425–434.
[5] Chung M, Bernheim A, Mei X, Zhang N, Huang M, et al. CT imaging features of2019 novel coronavirus (2019-nCoV). Radiology. 2020; 295(1):202–207.
[6] Chen Y, Guo Y, Pan Y, Zhao ZJ. Structure analysis of the receptor binding of 2019-nCoV. Biochem Biophys Res Commun. 2020; 525(1):135–140.
[7] Walls AC, Park YJ, Tortorici MA, Wall A, McGuire AT, et al. Structure, function, and antigenicity of the SARS-CoV-2 spike glycoprotein. Cell. 2020; 181(2):281–292.e6.
[8] Letko M, Marzi A, Munster V. Functional assessment of cell entry and receptor usage for SARS-CoV-2 and other lineage B betacoronaviruses. Nat Microbiol. 2020; 5(4):562–569.
[9] Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020; 382(18):1708–1720.
[10] Hamming I, Timens W, Bulthuis ML, Lely AT, Navis G, et al. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Pathol. 2004; 203(2):631–637.
[11] Jia HP, Look DC, Shi L, Hickey M, Pewe L, et al. ACE2 receptor expression and severe acute respiratory syndrome coronavirus infection depend on differentiation of human airway epithelia. J Virol. 2005; 79(23):14614–14621.
[12] Yoshikawa T, Hill T, Li K, Peters CJ, Tseng CT. Severe acute respiratory syndrome (SARS) coronavirus-induced lung epithelial cytokines exacerbate SARS pathogenesis by modulating intrinsic functions of monocyte-derived macrophages and dendritic cells. J Virol. 2009; 83(7):3039–3048.
[13] Wang M, Hao H, Leeper NJ, Zhu L, Career EC. Thrombotic regulation from the endothelial cell perspectives. Arterioscler Thromb Vasc Biol. 2018; 38(6):e90–e95.
[14] Lovren F, Pan Y, Quan A, Teoh H, Wang G, et al. Angiotensin converting enzyme-2 confers endothelial protection and attenuates atherosclerosis. Am J Physiol Heart Circ Physiol. 2008; 295(4):H1377–H1384.
[15] Sluimer JC, Gasc JM, Hamming I, van Goor H, Michaud A, et al. Angiotensin-converting enzyme 2 (ACE2) expression and activity in human carotid atherosclerotic lesions. J Pathol. 2008; 215(3):273–279.
[16] Lu X, Zhang L, Du H, Zhang J, Li YY, et al. SARS-CoV-2 infection in children. N Engl J Med. 2020; 382:1663–1665.
[17] Chen N, Zhou M, Dong X, Qu J, Gong F, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020; 395(10223):507–513.
[18] Wu Z, McGoogan JM. Characteristics of and important lessons from the Coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72314 cases from the Chinese Center for Disease Control and Prevention. JAMA. 2020; 323(13):1239–1242.
[19] Onder G, Rezza G, Brusaferro S. Case-fatality rate and characteristics of patients dying in relation to COVID-19 in Italy. JAMA. 2020; 323(18):1775–1776.
[20] Qiu H, Wu J, Hong L, Luo Y, Song Q, et al. Clinical and epidemiological features of 36 children with coronavirus disease 2019 (COVID-19) in Zhejiang, China: an observational cohort study. Lancet Infect Dis. 2020; 20(6):689–696.
[21] Dong Y, Mo X, Hu Y, Qi X, Jiang F, et al. Epidemiological characteristics of 2143 Pediatric patients with 2019 coronavirus disease in China. Pediatrics. 2020; 145(6):e20200702.
[22] Gautret P, Lagier JC, Parola P, Hoang VT, Meddeb L, et al. Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. Int J Antimicrob Agents. 2020; 56(1):105949.
[23] Wang, YC Luo H, Liu S, Huang S, Zhou Z, et al. Dynamic evolution of COVID-19 on chest computed tomography: experience from Jiangsu Province of China. European Radiology. 2020; 30:6194–6203.
[24] Gao Y, Li T, Han M, Li X, Wu D, et al. Diagnostic utility of clinical laboratory data determinations for patients with the severe COVID-19. J Med Virol. 2020; 92(7):791–796.
[25] Livingston E, Bucher K. Coronavirus disease 2019 (COVID-19) in Italy. JAMA. 2020; 323(14):1335.
[26] Li K, Wu J, Wu F, Guo D, Chen L, et al. The clinical and chest CT features associated with severe and critical COVID-19 pneumonia. Invest Radiol. 2020; 55(6):327–331.
[27] Du Y, Tu L, Zhu P, Mu M, Wang R, et al. Clinical features of 85 fatal cases of COVID-19 from Wuhan: a retrospective observational Study. Am J Respir Crit Care Med. 2020; 201(11):1372–1379.
[28] Dutch Association for Radiology (Nederlandse Vereniging voor Radiologie, NVvR). Accessed on 14 April 2020 from: https://www.radiologen.nl/secties/netwerk-covid-19/documenten/handreiking-standaardverslag-ct-thorax-covid-inclusief-co-rads
[29] Prokop M, van Everdingen W, van Rees Vellinga T, Quarles van Ufford J, Stöger L, et al. CO-RADS: A categorical CT assessment scheme for patients suspected of having COVID-19-Definition and Evaluation. Radiology. 2020; 296(2):E97–E104.
[30] Francone M, Iafrate F, Masci GM, Coco S, Cilia F, et al. Chest CT score in COVID-19 patients: correlation with disease severity and short-term prognosis. Eur Radiol. 2020; 30(12):6808–6817.
[31] Pan F, Ye T, Sun P, Gui S, Liang B, et al. Time course of lung changes on chest CT during recovery from 2019 novel coronavirus (COVID-19) pneumonia. Radiology. 2020; 295(3):715–721.
[32] Rossi SE, Erasmus JJ, Volpacchio M, Franquet T, Castiglioni T, et al. “Crazy-Paving” Pattern at Thin-Section CT of the Lungs: Radiologic-Pathologic Overview. Radiographics. 2003; 23(6):1509–1519.
[33] Parekh M, Donuru A, Balasubramanya R, Kapur S. Review of the chest CT differential diagnosis of ground-glass opacities in the COVID Era. Radiology. 2020; 297(3):E289–E302.
[34] Behzad S, Aghaghazvini L, Radmard AR, Gholamrezanezhad A. Extrapulmonary manifestations of COVID-19: Radiologic and clinical overview. Clin Imaging. 2020; 66:35–41.
[35] Cavagna E, Muratore F, Ferrari F. Pulmonary thromboembolism in COVID-19: Venous thromboembolism or arterial thrombosis? Radiology: Cardiothoracic Imaging. 2020; 2(4). Available from: https://pubs.rsna.org/doi/10.1148/ryct.2020200289