Full Text
Conn’s original description of primary aldosteronism (PA) was in 1954. Since then, improvements in qualitative laboratory based diagnostic methods have dramatically increased the diagnostic yield. Patients undergoing evaluation for hypertension are rarely screened for hyperaldosteronism and therefore is underdiagnosed. With the aid of plasma aldosterone concentration (PAC) to plasma renin activity (PRA) ratio), the screening and detection of PA has become more feasible and detection has increased [1].
Primary aldosteronism may be associated with resistant hypertension, which is defined as failure to achieve goal blood pressure despite adherence to an appropriate three-drug regimen including a diuretic. There are wide variations of estimate regarding the prevalence of PA, ranging from 5-13% [2, 3].
Excessive secretion of aldosterone is associated with an increased risk of cardiovascular disease and morbidity [4]. Risk is greater when compared to patients with other types of hypertension, including essential hypertension, pheochromocytoma, and Cushing's syndrome. The excess cardiovascular risk resolves after appropriate treatment of the mineralocorticoid excess.
Case report
A 48-year-old woman was admitted to KIMS hospital, Secunderabad, with symptoms of dizziness and vomitings of 2 days duration. She also had chest discomfort and palpitations. She had increased weakness and fatigue. She had history of hypertension (taking telmisartan) and was recently diagnosed to have diabetes mellitus (type 2). She had stenting done for right ureteral calculus 1 year ago.
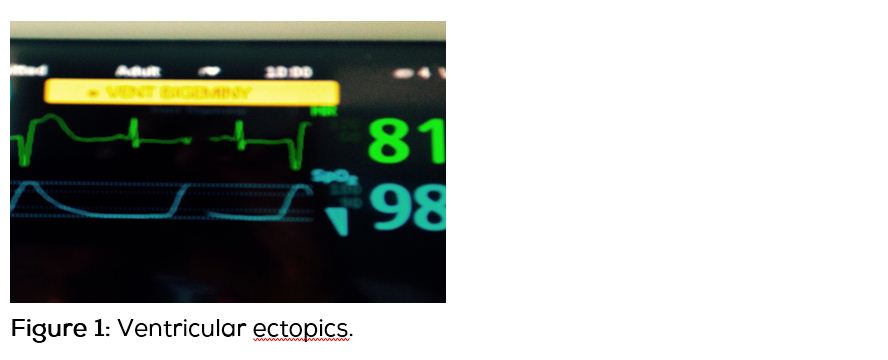
She had ventricular bigeminy on ECG. BP was 150/80, other vital signs were normal. ABG showed evidence of metabolic alkalosis. She had severe hypokalemia (K–2.4 mmol/L) and hypernatremia (Na–147 mmol/L). She received potassium chloride replacement for several days for persistent hypokalemia. Magnesium sulfate was also given because magnesium deficiency is frequently associated with hypokalemia. Concomitant magnesium deficiency aggravates hypokalemia and renders it refractory to treatment by potassium. Her Mg level even after supplementation was borderline low and was corrected again. Ventricular ectopics (Figure 1) resolved with correction of hypokalemia.
Transtubular potassium gradient (TTKG) was calculated. It was 7.64 (Value >3, in the setting of hypokalemia, is suggestive of renal loss of potassium). She was evaluated for suspected hyperaldosteronism. Aldosterone and plasma renin activity (PRA) were checked. Serum aldosterone while supine was 82.5 pg/mL (reference range: adults 10-105 supine and 34-273 upright). PRA was 0.06 ng/mL/hr (reference range: upright 1.90-6.00, supine 0.50-1.90). Aldosterone/PRA ratio was elevated (147); laboratory interpretation >30: suggestive of primary hyperaldosteronism and >50: diagnostic of primary hyperaldosteronism.
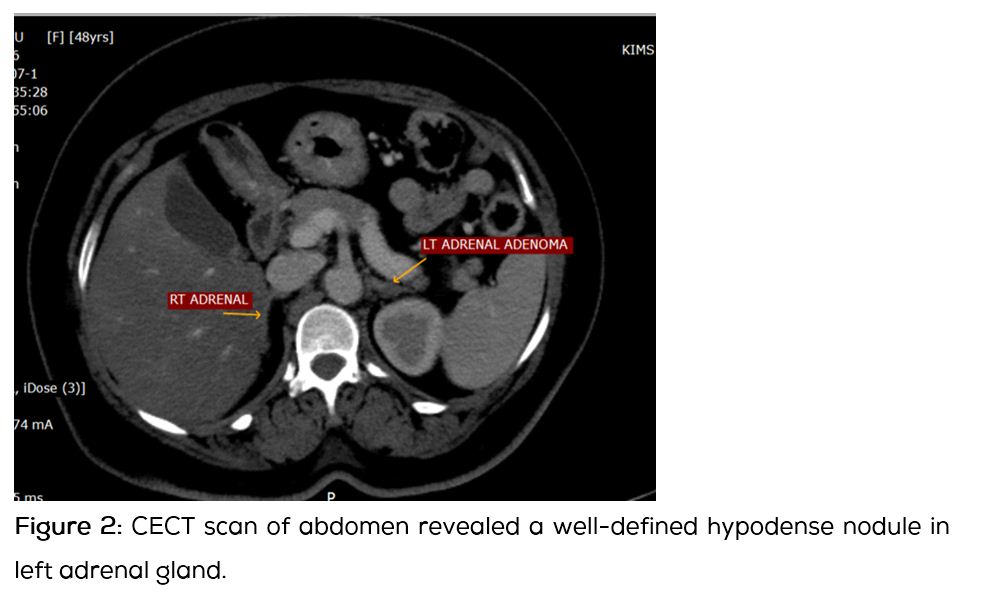
Dexamethasone suppression test and urinary catecholamines were both normal, ruling out Cushing’s syndrome and pheochromocytoma respectively. CT scan of abdomen (Figure 2) with contrast was done which revealed a well-defined hypodense nodule of size 7.5 × 5.7 mm in left adrenal gland, suggestive of adrenal adenoma.
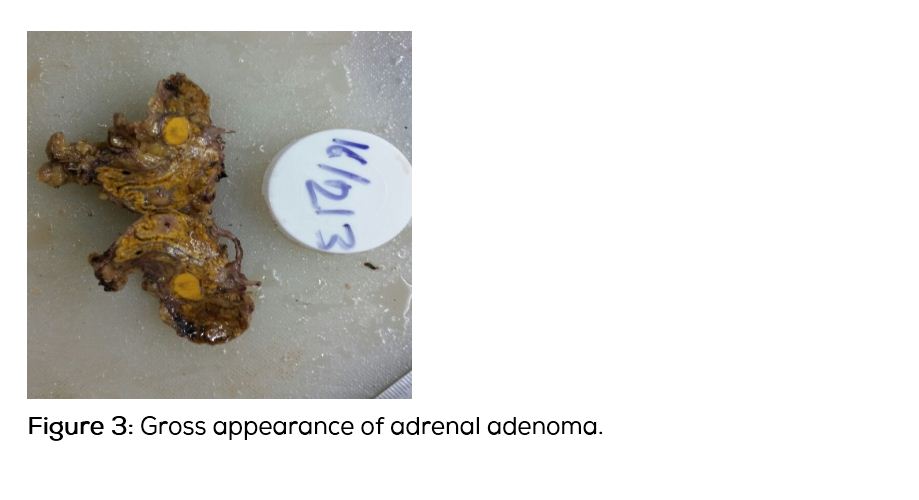
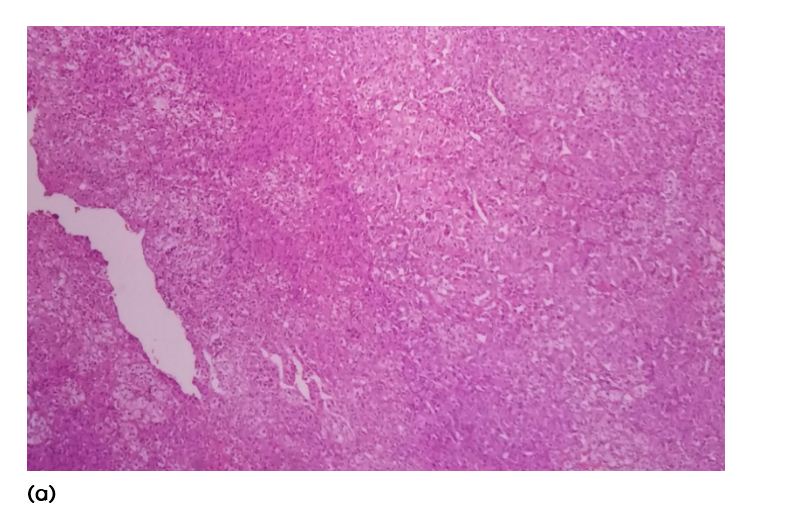
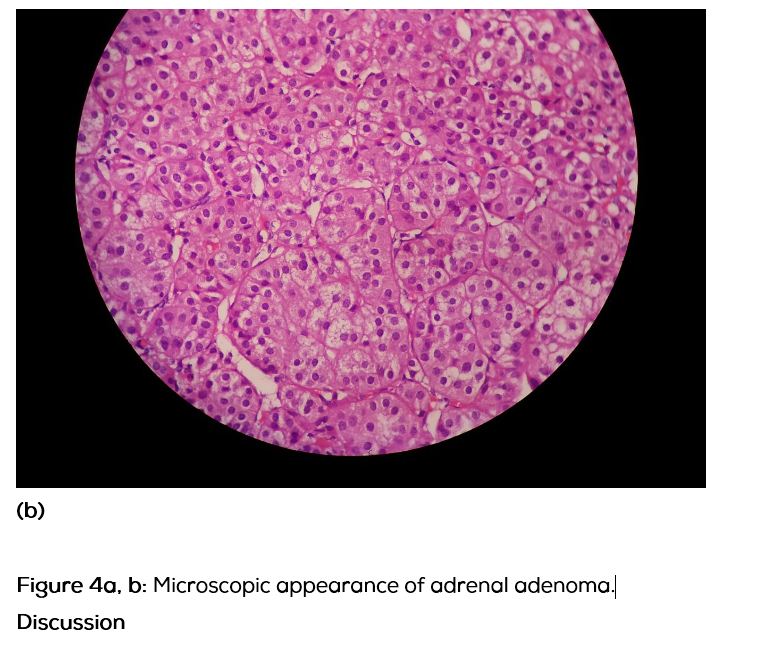
She was discharged on spironolactone and subsequently underwent laparoscopic left adrenalectomy. Her BP and electrolytes were within normal limits post-operatively. Histopathological examination of the specimen showed a 1 × 1 × 0.7cm well circumscribed yellow nodule (Figure 3) and microscopic appearance was of a nodule composed of cells with foamy cytoplasm and round nuclei (Figure 4a, b). No nuclear atypia seen. Features were compatible with adrenal adenoma with intact capsule. On post-operative follow-up, her BP and electrolytes were normal and she was not on any anti-hypertensive medication or aldosterone antagonist.
Many subtypes of primary aldosteronism have been described [2]. The most frequent causes of primary aldosteronism include: Bilateral idiopathic hyperaldosteronism (or idiopathic hyperplasia [IHA], 60 to 70 percent); unilateral aldosterone producing adenomas (APA) (30 to 40 percent).
Less common forms include unilateral hyperplasia or primary adrenal hyperplasia; familial hyperaldosteronism; and pure aldosterone-producing adrenocortical carcinomas and ectopic aldosterone-secreting tumors.
The clinical features of primary aldosteronism are in part determined by the renal actions of aldosterone. Aldosterone causes increased sodium reabsorption and increased secretion of cellular potassium into the lumen. Although hypokalemia is considered to be a "classic" sign of primary aldosteronism, some patients with primary aldosteronism due to an adrenal adenoma and more commonly those with adrenal hyperplasia are not hypokalemic. Hypokalemia is therefore an inconsistent finding [1, 3].
Other clinical features include mild hypernatremia, metabolic alkalosis, hypomagnesemia and muscle weakness, but there is no edema. Investigations for primary hyperaldosteronism were carried out [2, 5] for patients with Hypertension and hypokalemia; Severe or resistant hypertension; hypertension and an adrenal incidentaloma; hypertension and a family history of early-onset hypertension or stroke at a young age (<40 years); hypertension and first-degree relatives with documented primary aldosteronism.
The initial evaluation should consist of documenting that the PRA is reduced (typically undetectable), and the plasma aldosterone concentration (PAC) is inappropriately high for the PRA; the net effect is an elevated PAC/PRA [2].
An elevated PAC/PRA ratio alone is not always diagnostic of primary aldosteronism. Diagnosis is confirmed by demonstrating inappropriate aldosterone secretion. Aldosterone suppression testing can be performed with orally administered sodium chloride and measurement of urinary aldosterone excretion or with intravenous sodium chloride loading and measurement of the plasma aldosterone concentration [2].
The patient, in the case described above had spontaneous and refractory hypokalemia, metabolic alkalosis, almost undetectable PRA, and an elevated PAC/PRA ratio, suggestive of primary aldosteronism. Aldosterone suppression testing could not be done in our patient due to logistic reasons. However the suspicion for primary aldosteronism was very high because the PAC/PRA ratio was high in spite of being on angiotensin receptor blocker (which would normal be expected to give a false negative result).
Guidelines recommend that all patients with primary aldosteronism undergo adrenal CT as the initial study in subtype testing and to exclude adrenocortical carcinoma [2, 5]. Adrenal venous sampling is recommended if CT scan is normal or shows bilateral abnormalities [6]. It has been recommended to establish/exclude unilateral form of primary aldosteronism. This could not be done for our patient due to her reluctance to get an invasive test done. The fact that the surgery (unilateral adrenalectomy) was successful and she did not need any anti-hypertensive medication or aldosterone antagonist post-operatively, confirms the fact that her unilateral adrenal adenoma (confirmed on biopsy) was indeed a ’functional’ aldosterone producing adenoma.
For most patients with confirmed unilateral aldosterone hypersecretion (eg., adrenal adenoma or unilateral adrenal hyperplasia), unilateral adrenalectomy (preferably, laparoscopic adrenalectomy) is preferred over medical therapy [2, 5]. Hypokalemia should be corrected with spironolactone preoperatively. Patients with bilateral adrenal hyperplasia are treated with medical therapy, not adrenalectomy [2].
For patients with either bilateral adrenal hyperplasia or confirmed unilateral adrenal aldosterone hypersecretion (who refuse or are not for surgery), an aldosterone antagonist (eg., spironolactone or eplerenone) is the first choice for pharmacologic therapy [7]. Serum potassium, creatinine, and blood pressure should be monitored frequently in the first 4-6 weeks of postoperative period.
Conclusions
Diagnosis of PA should be suspected in hypertensive patients with hypokalemia in the absence of Cushing’s syndrome. PA was once regarded as rare disorder with a very low prevalence among unselected hypertensive patients, but it is now realized that significant number of cases with hypertension may have PA. It is important for physicians to have high degree of suspicion to recognize this disorder early and to carryout proper diagnostic tests and treatment, to avoid missing curable hypertension.
Acknowledgements
Acknowledgements are due to the Departments of Radiology and Imageology, Krishna Institute of Medical Sciences (KIMS), Secunderabad.
Conflicts of interest
The authors declare no conflict of interest.
References
1. Mulatero P, Stowasser M, Loh KC, Fardella CE, Gordon RD, et al. Increased diagnosis of primary aldosteronism, including surgically correctable forms, in centers from five continents. J Clin Endocrinol Metab. 2004; 89(3):1045–1050.
2. Young WF. Primary aldosteronism: renaissance of a syndrome. Clin Endocrinol (Oxf). 2007; 66(5):607–618.
3. Loh KC, Koay ES, Khaw MC, Emmanuel SC, Young WF Jr. Prevalence of primary aldosteronism among Asian hypertensive patients in Singapore. J Clin Endocrinol Metab. 2000; 85(8):2854–2859.
4. Catena C, Colussi G, Nadalini E, Chiuch A, Baroselli S, et al. Cardiovascular outcomes in patients with primary aldosteronism after treatment. Arch Intern Med. 2008; 168(1):80–85.
5. Funder JW, Carey RM, Fardella C, Gomez-Sanchez CE, Mantero F, et al. Case detection, diagnosis, and treatment of patients with primary aldosteronism: an endocrine society clinical practice guideline. J Clin Endocrinol Metab. 2008; 93(9):3266–3281.
6. Rossi GP, Auchus RJ, Brown M, Lenders JW, Naruse M, et al. An expert consensus statement on use of adrenal vein sampling for the subtyping of primary aldosteronism. Hypertension. 2014; 63(1):151–160.
7. Parthasarathy HK, Ménard J, White WB, Young WF Jr, Williams GH, et al. A double-blind, randomized study comparing the antihypertensive effect of eplerenone and spironolactone in patients with hypertension and evidence of primary aldosteronism. J Hypertens. 2011; 29(5):980–990.