Full Text
Introduction
There is a new public health crisis, coronavirus disease 2019 (COVID-19) pandemic threatening the world with the emergence and spread of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). This began as an outbreak in December 2019 in Wuhan city of Hubei Province, China as novel coronavirus (2019-nCoV). The World Health Organization (WHO) referred the disease condition as COVID- 19 [1] and the virus as “SARS related CoV-2” (SARS- CoV-2) [2]. The World Health Organization declared COVID-19 disease as a “public health emergency of international concern” (PHEIC) [3] on 30th January 2020 and as pandemic [4] on 11th March 2020 due to its rapid spreading ability. It covered more than 213 countries and territories around the world consisting of 24,854,140 confirmed cases and 8,38,924 deaths as of 30th August 2020 [5]. The number of COVID -19 patients has dramatically increased worldwide. The case fatality rate is estimated to range from 2 to 3% [6, 7]. The treatment in intensive care units has become a major challenge. Confirmed detection of the SARS-CoV-2 virus followed by isolation of the infected person at the earliest possible is the only measure to prevent the further transmission of this disease. Hence early diagnosis of COVID -19 is absolutely essential in present pandemic situation.
Real-time reverse transcription PCR (real-time RT-PCR) is the gold standard test for diagnosis of COVID-19. It requires specialized laboratory setup, equipment, skilled personnel and biosafety cabinet. Advantage of this test is its accuracy of detection. Average time taken is around 4-5 hours from receipt of sample. The time taken for sample transportation and increasing sample load also to be considered. In view of urgent need of quick and early detection of COVID-19 positive patients, rapid antigen detection test (point of care testing) has edge over it. This test also helps in proper implementation of the Govt. strategy to test, track and treat. The other advantage of performing this test, it helps in reducing in the anxiety and fear among healthcare workers involved in the patient care [8].
This study was conducted with the aim: (1) to evaluate the infection positivity rate of COVID-19 disease, (2) to assess the age and gender wise prevalence of the disease, (3) to compare the results of rapid antigen detection test and gold standard real time RT-PCR for early detection of SARS-CoV-2 virus, (4) to evaluate the sensitivity and specificity of rapid antigen detection test for COVID-19 disease, (5) to evaluate the infection positivity rate of COVID-19 disease among symptomatic and asymptomatic patients.
Materials and methods
An observational study was conducted in the department of Microbiology and Molecular biology of Krishna Institute of Medical Sciences, Secunderabad, Telangana, India. It is a multispecialty, tertiary care private hospital approved by National Accreditation Board for Hospitals and Healthcare (NABH) & National Accreditation Board for Testing & Calibration Laboratories (NABL). A total of 849 patients’ specimens (nasopharyngeal swab) were tested for COVID-19 by rapid antigen detection assay within a period of one and half month from middle of July 2020 to end of August 2020. Among them, 208 patients’ specimens were compared for both rapid antigen detection assay and real time RT-PCR test for COVID-19.
In the present study, only those of individuals were included who were tested by both rapid antigen detection test (Chromatographic immunoassay) and real time RT-PCR at the same point of time. The individuals who were tested by only one method were excluded from the study.
Suspected individuals, who tested negative for COVID-19 by rapid antigen test, were tested sequentially by RT-PCR to rule out infection as per Indian Council of Medical Research (ICMR) guidelines [8]. Few patients, who turned out positive for COVID-19 by rapid antigen detection test were also reconfirmed by RT-PCR test, although a positive test should be considered as a true positive and does not need retesting.
Specimens were collected from (i) all symptomatic patients of Influenza Like Illness (ILI) who were suspected of having COVID19 infection, (ii) all asymptomatic patients- who were undergoing chemotherapy; immunosuppressed patients including HIV+; patients diagnosed with malignant disease; transplant patients; elderly patients (>65 years of age) with co-morbidities such as lung disease, heart disease, liver disease, kidney disease, diabetes, neurological disorders, blood disorders and (iii) all asymptomatic patients - undergoing elective/emergency surgical procedures or non-surgical interventions such as bronchoscopy, upper GI endoscopy and dialysis [8]. The category of the patients and the number of patients in each category is depicted in table no 2.
Rapid antigen detection test
Rapid antigen detection test were performed by standard Q COVID -19 Ag kit of SD BIOSENSOR which is a chromatographic immunoassay. It is a qualitative, point of care (POC) test.
Procedure: Nasopharyngeal swabs were collected from patients by trained healthcare worker following full infection control practices including use of proper PPE. Swabs were immediately inserted into the extraction buffer tube provided in the kit. Buffer tubes were squeezed while stirring the swabs for 5 times [9]. The swabs were removed & discarded in Yellow bag following BMW Management rules, 2016 and latest guidelines provided by Central Pollution Control Board [10]. Nozzle caps (provided in the kit) were pressed onto the buffer tubes. Two drops of extracted specimens were poured into well of the cassette. Readings were recorded. Cassettes were observed for15-20 minutes before reporting as negative. Tests were conducted under strict medical supervision and within 10-15 minutes of specimen collection in extraction buffer. Temperature of the kit was always maintained between 2oC to 30oC.
All testing results using Ag detection assay were entered on the ICMR covid-19 portal and also communicated to the state authorities and officials of the Integrated Disease Surveillance Programme (IDSP) on a real-time basis.
All suspected individuals who test negative for COVID-19 by rapid antigen tests were tested sequentially by RT-PCR to rule out infection.
Real time RT PCR
Real time RT-PCR performed with the nasopharyngeal specimens were only included in the study.
Procedure: Nasopharyngeal swabs (dacron swabs) were collected from patients by trained healthcare workers and placed in a commercially available viral transport medium. Strict infection control practices were followed including use of proper PPE.
The sample material for the isolation of RNA was sent in appropriate sample collection systems. For correct sampling, instructions given by the WHO under the following link https://www.who.int/csr/sars/sampling/en/ [11] were followed.
The specimens were transported to the Molecular Biology Department by triple packaging following recommended guidelines [12]. Laboratory request forms and ICMR information forms [13] were filled appropriately.
For viral RNA extraction, QIAGEN QIAamp® Viral RNA Mini QIAcube Kit (validated RNA isolation kits) [14] was used. The process of extraction was carried out using QIAamp Mini spin columns in a standard microcentrifuge, on a fully automated extraction instrument QIAcube [15].
Quality control: Each lot of the QIAamp RNA Mini QIAcube Kit was tested against predetermined specifications to ensure consistent product quality.
The ViroQ SARS-CoV-2 Kit (BAG Diagnostics) was used for the in vitro, qualitative detection of SARS-CoV-2 RNA in respiratory specimens (nasopharyngeal swabs). The kit contains primers and fluorescent probes to amplify and detect gene fragments for SARS-CoV-2 and is based on a one step reaction with real-time PCR technology. Here an efficient cDNA synthesis is possible from RNA coupled with a real-time PCR system, Rotor-Gene Q in one tube.
Test principle: The test is performed with extracted RNA as starting material. The RNA is converted into cDNA with a reverse transcriptase enzyme and afterwards amplified in a PCR (Rotor-Gene Q). The primers are designed for the selective amplification of the trancripted cDNA of the viral genes - SARS-CoV-2/ RdRP Gene (RNA-dependent RNA-Polymerase) and Beta-CoV/ E Gene (Sarbeco, Envelope coding gene) for the detection of SARS-CoV-2 [16].
The amplicons are detected with likewise SARS-CoV-2 specific fluorescent dye-labelled hydrolysis probes-TaqMan® probes.
If amplicons are present, the probes are hydrolyzed by the Taq polymerase and a fluorescence signal is generated that increases proportionally with the amount of the PCR product. The fluorescence signals are measured by the optical detection unit of the real-time PCR cycler.
The test is performed in a single PCR reaction that detects the two viral genes RdRP & E gene and a universally expressed human housekeeping gene- Rnase P (Cell control) with different fluorescent colours. The detection of Rnase P indicates the correct sampling, RNA-Isolation and RT-PCR-amplification.
Assessment of clinical specimen test results were performed after the positive and negative controls have been examined and determined to be valid and acceptable. Internal quality control of new lots of the ViroQ SARS-CoV-2 kit were performed using a combination of RNA samples known to be positive or negative. To detect possible contaminations, negative controls are recommended. PCR reactions were performed with the RNAse free water as negative test control (NTC) for this purpose.
All reagents were stored in temperature recommended in the test kit.
All molecular techniques were performed in the BSL-2 [17] by well trained, qualified and experienced personnel in molecular techniques. Each step was performed following appropriate safety precautions, good laboratory practices and strict infection control practices. All biological materials and disposables were inactivated in autoclave (in an autoclavable bag) and discarded in yellow bag as per BMW management rules 2016 and latest guidelines provided by Central Pollution Control Board [10].
All testing results were entered on the ICMR COVID-19 portal, portal of Telangana state authorities and also communicated to District Medical and Health Officer and officials of Integrated Disease Surveillance Programme (IDSP) on a real-time basis.
Results
A total of 208 patients’ dual specimens (nasopharyngeal swab) were received for testing by rapid antigen detection assay and subsequently by real time RT PCR test. The results of both the tests were compared. Among them, 142 (68.26%) were males and 66 (31.73%) were females (Table 1). The patients belonged to symptomatic and asymptomatic categories as mentioned in Table 2.
Table 1: Age and gender wise distribution of patients included in the study (n=208).
|
Age groups
|
Males
|
Females
|
|
0-10 Years
|
0 (0%)
|
01 (1.51%)
|
|
11-20 Years
|
02 (1.40%)
|
0 (0%)
|
|
21-30 Years
|
11 (7.74%)
|
07 (10.6%)
|
|
31-40 Years
|
16 (11.26%)
|
05 (7.57%)
|
|
41-50 Years
|
30 (21.1%)
|
14 (21.21%)
|
|
51-60 Years
|
27 (19.01%)
|
16 (24.24%)
|
|
61-70 Years
|
33 (23.2%)
|
16 (24.24%)
|
|
71-80 Years
|
21 (14.7%)
|
06 (9.09%)
|
|
>80 Years
|
02 (1.40%)
|
01 (1.51%)
|
|
Total= 208
|
142 (68.26%)
|
66 (31.73%)
|
Table 2: Various categories of the patients included in the study (n=208).
|
Sr.No.
|
Category of the patients
|
No. of patients
|
|
1
|
Symptomatic patients: Patients of influenza like illness (ILI) who were suspected of having COVID19 infection,
|
123 (59.13%)
|
|
2
|
Asymptomatic patients:Patients who were undergoing chemotherapy; immunosuppressed patients including HIV+; patients diagnosed with malignant disease; transplant patients; elderly patients (>65 years of age) with co-morbidities such as lung disease, heart disease, liver disease, kidney disease, diabetes and blood disorders.
|
34 (16.34%)
|
|
3
|
Asymptomatic patients: Patients undergoing elective/emergency surgical procedures or non-surgical interventions such as bronchoscopy, upper GI endoscopy and dialysis.
|
51 (24.51%)
|
The number of patients tested as positive (either by any one method or by both the methods) were 56 (26.92%) and negative were 152 (73.07%) as shown in Figure 1. 56 positive patients include 34 (60.71%) males and 22 (39.28%) females. Among males the predominantly affected age group was 51-60 years (29.4%) followed by 61-70 years (26.4%), whereas among females the predominantly affected age groups were 21-30 years (27.2%) and 71-80 years (27.2%) as depicted in Figure 2.
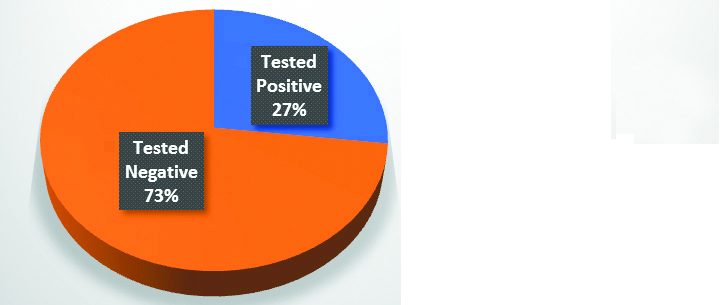
Figure 1: Percentage of positive and negative results (COVID-19 Infection positivity rate).
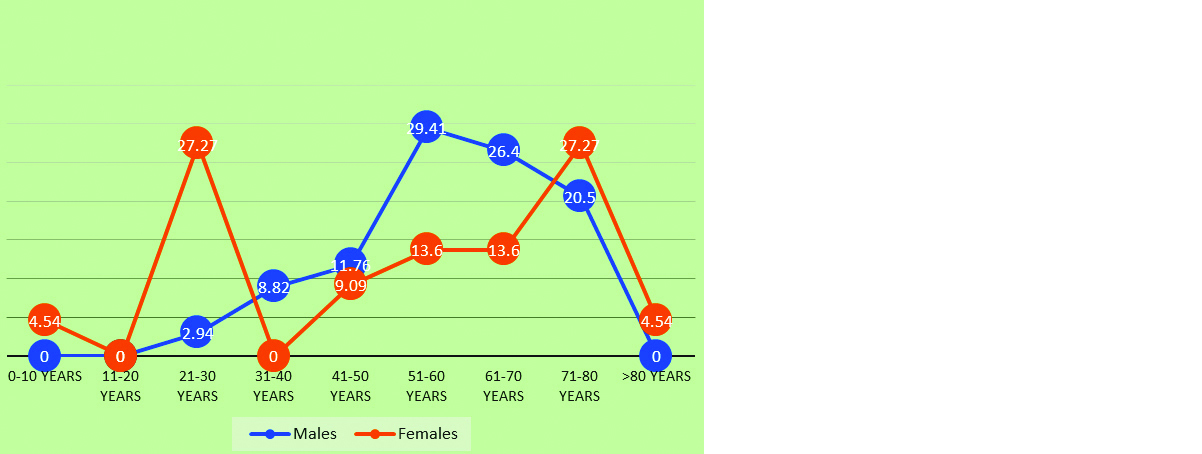
Figure 2: Age and gender wise distribution of positive patients in percentage (n=56).
Among 208 dual specimens, 29 (13.94%) specimens found to be positive by both rapid antigen detection assay and real time RT -PCR. 152 (73.07%) specimens showed negative by both the tests. 23 (11.05%) specimens showed negative by rapid antigen detection assay but positive by real time RT -PCR. 04 specimens (1.92%) showed positive by rapid antigen detection assay but negative by real time RT-PCR (Table 3).
Table 3: Comparative table of rapid antigen detection and real time RT PCR tests (n=208).
|
|
Rapid Ag Positive/
RT-PCR Positive
|
Rapid Ag Negative/
RT-PCR Negative
|
Rapid Ag Negative/
RT-PCR Positive
|
Rapid Ag Positive/
RT-PCR Negative
|
|
Results
|
29 (13.94%)
|
152 (73.07%)
|
23 (11.05%)
|
04 (1.92%)
|
These results generate a sensitivity of 55.76% and a specificity of 97.43% for rapid antigen detection assay (Table 4).
Table 4: Calculation of sensitivity and specificity rapid antigen detection assay (n=208).
|
Positive test results
|
Rapid Ag Pos / RT-PCR Pos
(True Positive)
n=29
|
Rapid Ag Pos / RT-PCR Neg
(False Positive)
n=04
|
|
Negative test results
|
Rapid Ag Neg / RT-PCR Pos
(False Negative)
n=23
|
Rapid Ag Neg / RT-PCR Neg
(True Negative)
n= 152
|
Sensitivity: 55.76% (TP/ TP+FN X100); Specificity: 97.43% (TN/ TN+ FP X100)
All positive and negative results were analysed among 123 symptomatic (59.13%) and 85 asymptomatic (40.86%) patients. It was observed that, among 29 true positive patients (positive results by both rapid antigen detection assay and real time RT- PCR), 19 (65.5%) were symptomatic whereas 10 (34.4%) were asymptomatic patients. Among 152 true negative patients (negative by both rapid antigen detection assay and real time RT-PCR), 95 (62.5%) were found to be symptomatic whereas, 56 (36.84%) were found to be asymptomatic.
Among 23 False Negative patients, who showed negative by rapid antigen detection test but positive by real time RT-PCR, 13 (56.52%) were symptomatic and 10 (43.47%) were asymptomatic, whereas among 04 False Positive patients (rapid antigen test positive but RT-PCR Negative), 02 (50%) were symptomatic and 02 (50%) were asymptomatic (Figure 3).

Figure 3: Comparative table of symptomatic and asymptomatic patients.
Among all 56 COVID-19 positive patients, 34 (60.71%) were symptomatic and 22 (39.2%) were asymptomatic (Figure 4).
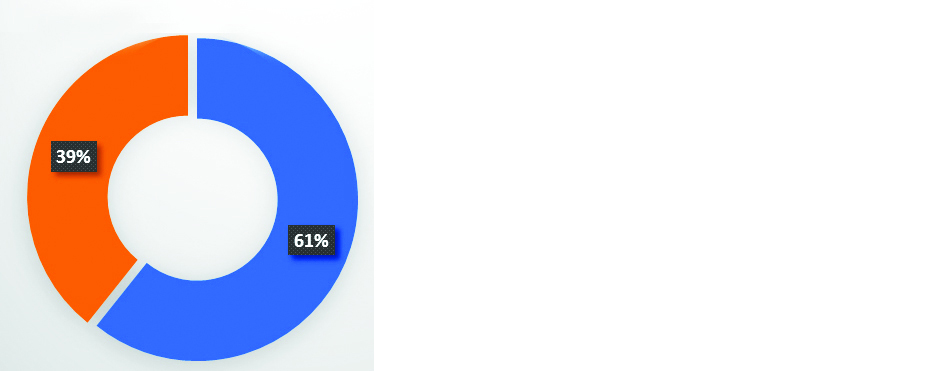
Figure 4: COVID-19 Infection positivity rate among symptomatic and asymptomatic patients (n=56).
Symptomatic positives were most commonly observed among the males within age group 61-80 years (53.8%) followed by the age group 41-60 years (34.61%). Among females, symptomatic positives were most commonly seen within the age group 41-60 years and 61-80 years (37.5%) (Figure 5).
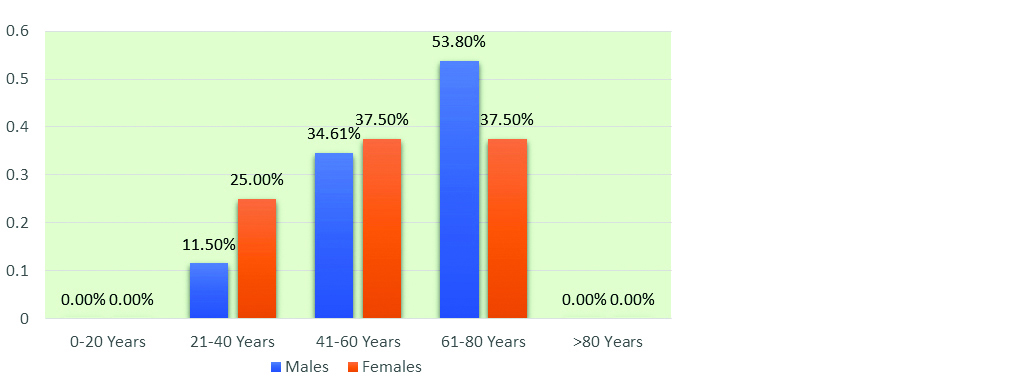
Figure 5: Age and gender wise distribution of symptomatic positive patients (n=34).
Asymptomatic positives were predominantly seen among males of 41-60 years of age group (63.63%) and among females of 61-80 years of age group (36.36%) (Figure 6).
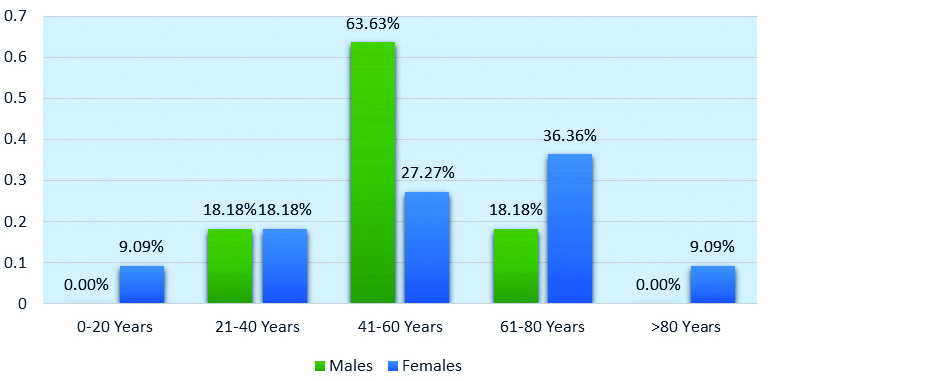
Figure 6: Age and gender wise distribution of asymptomatic positive patients (n=22).
Discussion
The results of the present study suggest that COVID-19 infection positivity rate is 26.92%. Males are predominantly affected (60.71%). Among males 51-60 years of age group is most vulnerable (29.4%) followed by 61-70 years of age group (26.4%) and 71-80 years of age group (20.5%). It has been observed that children and adolescent age group is not affected at all whereas the risk has become drastically increased after the age of 50. This can probably be explained as, the young people have good protective immune response which is required to eliminate the virus. Most of the individuals started developing co-morbidities after the age of 50 years resulting into impaired protective immune response [18]. Unlike males, females of age group 21-30 years also share the same percentage of COVID-19 infection as the females of 71-80 years of age group (27.27%). Females of young age group may be affected due to lack of endogenous protective immune response or having an inappropriate genetic background (eg: HLA), that elicits specific antiviral activity [18].
Therefore, good general health and strategies to boost immune responses is important to get protection against COVID-19 disease.
Our study indicates, 13.94% specimens are true positive and 73.03% are true negative. 11.05% indicates false negative (negative by rapid antigen but positive by RT-PCR) and 1.92% as false positive (positive by rapid antigen but negative by RT-PCR).
In our study, 11.05% indicates false negative result by rapid antigen detection assay. Rapid antigen detection assay has high specificity and low sensitivity, so the possibilities of false-negative results remain there. Presence of low concentration of antigen in specimen (below the detection limit of the test) and improper sample collection could be the other reasons for false-negative result by rapid antigen detection assay [9]. In the present study, 1.92% results were considered as false positive by rapid antigen detection test because they were positive by rapid antigen but negative by RT-PCR. A negative result by RT-PCR does not exclude a possible infection. Inappropriate specimen collection, presence of PCR inhibitors and mutations or polymorphisms in the primer and probe binding sites are some of the reasons which may cause false negative results. High susceptibility of the RT-PCR method for cross contaminations, may also cause false negative results [16].
Results of present study generates a sensitivity of 55.76% and a specificity of 97.43% for rapid antigen detection test which correlates with the findings as evaluated by ICMR [8, 19].
The present study reported the prevalence of symptomatic and asymptomatic covid-19 cases as 60.71% and 39.2% whereas in a similar study conducted by Mizumoto et al., in Yokohama, Japan, 2020 reported, 48.26% as symptomatic cases and 51.73% as asymptomatic cases [20]. In another study Shental et al., suggested that 10- 30% of SARS-CoV-2 infected patients are asymptomatic [21].
In the developing countries, diagnostic testing is limited to only on symptomatic patients due to limited number of laboratories, limited access to molecular tests, high cost and other resource constraints. On the other hand, a significant level of viral shedding may occur even during the incubation period or prior to symptom onset (pre-symptomatic period). Zou L et al in March 2020 also mentioned about the transmission potential of asymptomatic or minimally symptomatic patients [22]. Hence, rapid and accurate identification of pre-symptomatic and asymptomatic cases is very crucial in effective control of silent spread COVID-19 pandemic. Therefore, there is an urgent need to increase diagnostic testing capabilities in order to screen asymptomatic carriers.
There are number of methods available for detection of virus and to combat this disease in the present pandemic situation, but these available diagnostic methods have their own limitations.
The detection of viral nucleic acid by RT-PCR is the standard for non- invasive diagnosis of COVID-19. Advantage of this test is its accuracy. This method is most sensitive and specific [23, 24] but testing time could be relatively long [25] because of sample overload. Further, it requires specialized laboratory setup, expensive equipment, skilled personnel and biosafety cabinet (BSL-2).
Rapid antigen detection test can provide rapid results within 30 minutes, are relatively cheap, won’t require any extensive training and can be used in the laboratories as well as at patient bedside (point of care). Although antigen-based tests are not as sensitive as molecular tests especially during the early stages of infection, but more suitable for testing in the community for screening purpose. This test is also useful in the remote areas where the laboratory facilities for molecular tests are not available and the areas with limited access to polymerase chain reaction (PCR) reagents.
Conclusion
The speedy and exponential global spread of COVID-19 disease strongly urges the fast and economic diagnostic tools for mass screening to prevent the spread of virus. Such tests will be routinely required until a vaccine is developed. Rapid antigen detection testing has potential to become an important tool for mass screening for COVID-19 among asymptomatic individuals in particular situations. This will also provide rapid and moderately reliable results in the areas with limited access to molecular tests. As sample size of our study is not adequate, further research and more data availability is required.
Conflict of interest
Authors declare no conflicts of interest.
References
[1] World Health Organization. WHO Director-General’s remarks at the media briefing on 2019-nCoV on 11 February 2020. WHO; 2020. Accessed on 17 February 2020 from: https://www.who.int/dg/speeches/detail/who-director-general-s-remarks-at-themedia-briefing-on-2019-ncov-on-11-february-2020
[2] Gorbalenya AE, Baker SC, Baric RS, Groot RJ de, Drosten C, et al. Severe acute respiratory syndrome-related coronavirus: The species and its viruses - A statement of the Coronavirus Study Group. BioRxiv. 2020. Available from: https://doi.org/10.1101/2020.02.07.937862.
[3] World Health Organization. Statement on the Second Meeting of the International Health Regulations. Emergency Committee regarding the outbreak of novel coronavirus (2019-nCoV); 2005. Accessed on 17 February 2020 from: https://www.who.int/news/item/30-01-2020-statement-on-the-second-meeting-of-the-international-health-regulations-(2005)-emergency-committee-regarding-the-outbreak-of-novel-coronavirus-(2019-ncov)
[4] WHO Director-General's opening remarks at the media briefing on COVID-19 - 11 March 2020. Available from: https://www.who.int/director-general/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020
[5] Coronavirus disease (COVID-19) Weekly Epidemiological Update Data as received by WHO from national authorities, as of 10 am CEST 30 August 2020. Available from: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200831-weekly-epi-update-3.pdf?sfvrsn=d7032a2a_4
[6] Singhal T. A review of coronavirus disease-2019 (COVID-19). The Indian Journal of Pediatrics. 2020; 87(4):281–286.
[7] Chatterjee P, Agarwal A, Sarkar S, Gupta N, Gangakhedkar RR, et al. The 2019 novel coronavirus disease (COVID-19) pandemic: A review of the current evidence. Indian J Med Res. 2020; 151(2):147–159.
[8] Advisory on use of rapid antigen detection test for COVID-19. Indian Council of Medical Research, Department of Health Research. India: ICMR;14th June 2020. Available from: https://www.icmr.gov.in/pdf/covid/strategy/Advisory_for_rapid_antigen_test14062020.pdf
[9] STANDARD Q COVID-19 Ag (SD Biosensor)- literature.
[10] Guidelines for handling, treatment and disposal of waste generated during treatment/diagnosis/ quarantine of COVID-19 patients by Central Pollution Control Bord (Ministry of Environment, Forest & Climate change). Available from: http://www.uppcb.com/pdf/Guidelines_190320.pdf
[11] Sampling for severe acute respiratory syndrome (SARS) diagnostic tests by instructions given by the WHO. Available from: https://www.who.int/csr/sars/sampling/en/
[12] Specimen Collection, Packaging and Transport Guidelines for 2019 novel Coronavirus (2019-nCoV). ICMR-NIV/2019-nCoV/Specimens_01. 20/01/2020. Available from: https://www.mohfw.gov.in/pdf/5Sample%20collection_packaging%20%202019-nCoV.pdf
[13] ICMR Specimen Referral Form for COVID-19 (SARS-CoV2). tmc.cov.in> Sample requisition Form (SRF)- ICMR. Available form: https://tmc.gov.in/pdf/covid-19%20test/Sample%20Requisition%20Form%20(SRF)-%20ICMR.pdf
[14] QIAGEN QIAamp® Viral RNA Mini QIAcube Kit. Available form: www.qiagen.com.
[15] Fully automated extraction instrument QIAcube. Available form: www.qiagen.com/MyQIAcube.
[16] ViroQ SARS-CoV-2 Kit (BAG Diagnostics). Test kit for the qualitative detection of SARS-CoV-2 RNA. Available form: www.bag-diagnostics.com
[17] World Health Organization. Laboratory testing for 2019 novel coronavirus (2019-nCoV) in suspected human cases. Geneva: WHO; 2020.
[18] Shi Y, Wang Y, Shao C, Huang J, Gan J, et al. COVID-19 infection: the perspectives on immune responses. Cell Death & Differentiation. 2020; 27(5):1451–1454.
[19] Advisory on Newer Additional Strategies for COVID-19 testing. Indian Council of Medical Research Department of Health Research. 23rd June 2020. Available from: https://www.icmr.gov.in/pdf/covid/strategy/New_additional_Advisory_23062020_3.pdf
[20] Mizumoto K, Kagaya K, Zarebski A, Chowell G. Estimating the asymptomatic proportion of coronavirus disease 2019 (COVID-19) cases on board the diamond princess cruise ship, Yokohama, Japan, 2020. Eurosurveillance, 2020; 25:10:2000180.
[21] Shental N, Levy S, Skorniakov S, Wuvshet V, Shemer-Avni Y, et al. Efficient high throughput SARS-CoV-2 testing to detect asymptomatic carriers. medRxiv. Available from: https://www.medrxiv.org/content/10.1101/2020.04.14.20064618v1
[22] Zou L, Ruan F, Huang M, Liang L, Huang H, et al. SARS-CoV-2 viral load in upper respiratory specimens of infected patients. N Engl J Med. 2020; 382(12):1177–1179.
[23] Ramesh K, Suman N, Kaushik S, Sanjay M. COVID-19 diagnostic approaches: different roads to the same destination. Virus Dis. 2020; 31(2):97–105.
[24] Kasterena PBV, Veera BVD, Brinka SVD, Wijsmana L, Jongea JD, et al. Comparison of seven commercial RT-PCR diagnostic kits for COVID-19. J Clin Virol. 2020; 128:104412.
[25] Wang L, Wang Y, Ye D, Liu Q. Review of the 2019 novel coronavirus (SARS-CoV-2). based on current evidence. Int J Antimicrob Agents. 2020; 55(6):105948.