Full Text
Introduction
The burden of colorectal cancer (CRC) has risen rapidly in the economically developed Asian countries and India is no exception. Unlike a few other cancers, CRC seems to be on the rise in India. Although accurate figures on the estimated annual increase of the disease are not available in India, it is established that CRC is the third most common cancer globally [1-6]. Reliable time trends for India are available only from the Bombay registry which shows a significant increase in the incidence of colon cancer for both men and women over two decades. There is a lack of national and professional guidelines on colorectal cancer screening in our country, and resources are scarce with variable governmental support. Mortality continues to rise from colorectal cancers that could well be prevented. There are a number of factors that may influence origin of this tumor types including environmental and genetic factors linked to enhanced surveillance. Incidence of colorectal cancer is highest in developed countries such as the United States and Japan, and lowest in developing countries like Africa and Asia.
Incidence is slightly higher in men than women, and is highest in African American men. The burden of CRC has risen rapidly in some economically developed Asian countries like Japan, South Korea and Singapore [7, 8]. CRC is the third most common malignant neoplasm worldwide and the second leading cause of cancer deaths in the United States. It is estimated that there will be 142,570 new cases diagnosed in the United States in 2010 and 51,370 deaths due to this disease [9, 10]. The incidence is higher in men than in women. It ranges from 50.0 per 100,000 per year in Hispanic men to 68.4 per 100,000 per year in African American men. In women it ranges from 35.1 per 100,000 per year in Hispanics to 51.7 per 100,000 per year in African Americans. The age-adjusted mortality rates for men and women are 24.8 per 100,000 per year in men and 17.4 per 100,000 per year in women [11, 12]. About 6% of Americans are expected to develop the disease within their lifetime and about half of those will die from it. Age-specific incidence and mortality rates show that the vast majority of cases are diagnosed after age 50 years; about 7% of CRCs occur younger than age 50 years [13].
A more recent study of British vegetarians found a 12% non-significant increase in CRC among the vegetarians in comparison to meat eaters. The multi European EPIC study which followed 142,605 men and 335,873 women for a median of 8.7 years showed a small inverse association between intake of total fruits and vegetables and CRC risk in non-tobacco users. This study also reported a 20% increase risk (non-significant) for CRC in current smokers with high fruit and vegetable consumption [14, 15].
Colon cancer in India
National Cancer Control Program helps in identifying the burden of cancer cases in India. However, there is no policy on screening for colon cancer separately. The three year report of Population Based Cancer Registries (PBCR 2006-2008) under National Cancer Control Program reported age adjusted incidence rates for colon cancer in the Imphal West district which was highest followed by Bangalore and Mumbai [1, 2, 4]. In the PBCR, Bangalore, the number of incident colon cancer cases among males is 3.88% and the number of incident colon cancer cases among females is 2.72%. However, the Hospital Based Cancer Registry (HBCR)-2004-2006 reported the number of incident colon cancer cases among males was 1.52% and among females the colon cancer incidence was 0.87% (ncrpindia.org/Annual _Report.aspx). Most of the time the colon cancers are detected when patient has symptoms and reports to the hospital. Data available from Tata Memorial Hospital, Mumbai demonstrates a total of 555 cases of colorectal cancer (CRC) diagnosed over a 32 year period (1941-1972). However in the year 2006 alone 560 cases of CRC were treated in the same institution indicating a significant increase in number of patients. This rise in patient load was explained by awareness among the subjects, enhanced surveillance for early detection, improved technologies for early detection and increase in population in general with increase in number of elderly patients in particular as it is common in this age group. It was also expected that with improvement in economic status there will be a further increase in number of cases [2]. Previous data from Tata Memorial hospital showed that even 30 years ago, the incidence of CRC was higher in Zoroastrians (Parsis) which is a socio-economically well off community.
Unpublished data from Kasturba Hospital, Manipal, Karnataka showed a total number of 108 patients diagnosed / treated for colon cancer during the period from Jan 1st to 31st Dec 2008. The average age at diagnosis was 57.5 years with a slightly male preponderance. At the time of diagnosis, 60% of the malignancies were in Duke‘s stage B and 30% were in Duke‘s stage C with an expected 5 year survival rates of 64% and 27% respectively. An alarming number of 796 patients during the following consecutive years, 2009, 2010 and 2011 were diagnosed and verified. There appeared to be a steady but small increase in the incidence during these years with a male preponderance. Thirty per cent of the cancer was confined to rectum. Rectosigmoid and ascending colon were affected in 8% each of these patients.
A study conducted in Kashmir Valley registered 212 cases out of which 53.3% were Colon cancers with a male to female ratio of 1.2:1. The article by Javid et al. reports that the incidence rates for CRC in Kashmir (an area with distinct cultural and food habits) are enigmatically as low as in other parts of India. This study also found that the CRC in Kashmir is more common in men, in older individuals and in the urban regions. Like the rest of India, rectal cancer was more common than colon cancer [3]. The crude incidence rate of colorectal cancer was 3.65/ 100,000; it was 3.78 in males and 3.50/ 100,000 in females. The incidence rates for colorectal cancer in Muslims and Hindus were different. The crude incidence for colorectal carcinoma was highest for district Srinagar 6.19/ 100,000 (urban area) and lowest for district Kupwara (rural area) 1.59/ 100,000. The highest numbers of cases were detected in the age group 55–59 years (n = 34). The age specific rate for colorectal carcinoma was highest in the age group 55–59 years (17.21/ 100,000) followed by 65–69 years (14.86/ 100,000).
The intra country variation of the incidence rates of CRC across India is limited, unlike the striking north south differences in the incidence rates of stomach cancer and gall bladder cancer. Population based time trend studies show a rising trend in the incidence of CRC in India [4-6]. Among the groups that have a high incidence of CRC are those with hereditary conditions, such as familial adenomatous polyposis and hereditary nonpolyposis CRC (inherited in an autosomal dominant manner). Combined, the two groups account for no more than 6% of CRCs. More common conditions associated with an increased risk include a personal history of CRC or adenomas; first-degree relative with CRC; a personal history of ovarian, endometrial, or breast cancer; and a personal history of long-standing chronic ulcerative colitis or Crohn colitis [16, 17]. These high-risk groups account for about a quarter of all CRCs. Limiting screening or early cancer detection to only these high-risk groups is expected to miss the majority of CRCs in the western population [18]. Although there is as yet no scientific evidence for starting a CRC screening programme in India, as the incidence of CRC is still several folds lower than in the developing and developed countries, the rising trend may necessitate initiation of screening programmes in selected population as above [19, 20].
Five-year survival is 90% if the disease is diagnosed while still localized (i.e., confined to the wall of the bowel), but only 68% for regional disease (i.e., disease with lymph node involvement) and only 10% if distant metastases are present [21]. Recently, due to reduced exposure to risk factors, early detection efforts, polypectomy and improved treat has been attributed to reduced trends in CRC incidence and mortality rates [22]. However, in the near term, even greater incidence and mortality reductions could be achieved if a greater proportion of adults received regular screening. Although prospective randomized trials and observational studies have demonstrated mortality reductions associated with early detection of invasive disease, as well as removal of adenomatous polyps a majority of adults are not receiving regular age- and risk-appropriate screening or have never been screened at all [23, 24-26].
Polyps (adenomatous and non-adenomatous)
Worldwide cancer screening programs have reduced mortality through a reduction in incidence of advanced disease. Modern CRC screening can achieve this goal through the detection of early-stage adenocarcinomas, detection and removal of adenocarcinomas and adenomatous polyps respectively, the latter generally accepted as non-obligate precursor lesions. Although, adenomatous polyps are common in adults overage 50 years, the majority of polyps will not develop into adenocarcinoma. However, histology and size of the polyp will determine their clinical outcome as these may represent approximately one-half to two-thirds of all colorectal polyps and are associated with a higher risk of CRC [27]. Therefore, most CRC screening studies evaluate polyps greater than or equal to 10 mm and define as invasive CRC, as well as advanced adenomas along with histologically having high-grade dysplasia or significant villous components. These have been described in detail and suggest importance of colorectal polyps in the development of CRC is largely indirect; however, evidences are convincing [28–30].
Screening for colon cancer
Today there is a range of options for CRC screening in the average-risk population, with current technology falling into 2 general categories: stool tests, which include tests for occult blood or exfoliated DNA; and structural exams, which include flexible sigmoidoscopy (FSIG), colonoscopy, double-contrast barium enema (DCBE), and computed tomographic colonography (CTC). Stool tests are best suited for the detection of cancer, although they also will deliver positive findings for some advanced adenomas, while the structural exams can achieve the dual goals of detecting adenocarcinoma as well as identifying adenomatous polyps [31]. These tests may be used alone or in combination to improve sensitivity or, in some instances, to ensure a complete examination of the colon if the initial test cannot be completed. Although screening tests for CRC vary in terms of the degree of supporting evidence, potential efficacy for incidence and mortality reduction, cost-effectiveness and acceptability, any one of these options applied in a systematic program of regular screening has the potential to significantly reduce deaths from CRC. Table 1 shows the American Cancer Society recommendations for the early detection of cancer in average-risk asymptomatic [32].
Table 1: American Cancer Society recommendations for the early detection of cancer in average-risk asymptomatic.
|
Population
|
Test or procedure
|
Frequency
|
|
Men and women 50 aged ≥ 50 years
|
FOBT (fecal occult blood test) with at least 50% test sensitivity for cancer,, or FIT (fecal immunochemical test) with at least 50% test sensitivity for cancer, or
|
Annual, starting at age age 50 years
|
|
Stool DNA test, or
|
Interval uncertain, starting at Age 50 years
|
|
Flexible sigmoidoscopy, or
|
Every 5 years, starting at age 50 years
|
|
FOBT and flexible sigmoidoscopy, or
|
Annual FOBT (or FIT) and flexible sigmoidoscopy every 5 years, starting at age 50 years with at least 50% test sensitivity
|
|
DCBE (Double Contrast Barium Enema), or
|
Every 5 years, starting at age 50 years
|
|
Colonoscopy
|
Every 10 years, starting at age 50 years.
|
|
CTC (Computed Tomography Colonography)
|
Every 5 years, starting at age 50 years
|
However, certain limitations preclude the use of CTC as a screening modality in the Indian context. The incidence of CRC in India is still several folds lower than in most developing and developed countries. A large percentage of individuals needing colorectal cancer screening in India cannot afford CTC. Widespread use of CTC for colorectal cancer screening is limited due to the need for reader training and the limited opportunities currently available. It may become an acceptable option if made available to the population in select patients. Therefore, the screening for CRC in Indian context may be limited to a) Individuals diagnosed earlier with adenomatous polyps, b) Individuals who have undergone surgery to remove prior colorectal cancer, c) Individuals with immediate relatives diagnosed with precancerous colorectal polyps or colorectal cancer, d) Individuals with inflammatory bowel disease like Crohn’s disease and e) Individuals suffering from one out of two specific syndromes i.e. hereditary nonpolyposis colon cancer (HNPCC) or familial adenomatous polyposis
Computed tomography colonography/ virtual colonoscopy (CTC)
Computed tomography colonography/ virtual colonoscopy (CTC), also referred to as virtual colonoscopy, is a minimally invasive imaging examination of the entire colon and rectum. CTC uses CT to acquire images and advanced 2D and 3D-image display techniques for interpretation. Since its introduction in the mid-1990s, there have been rapid advancements in CTC technology. Multidetector CT permits image acquisition of thin 1 to 2 mm slices of the entire large intestine well within breath-hold imaging times. Computer imaging graphics allow for visualization of 3D endoscopic flight paths through the inside of the colon, which are simultaneously viewed with interactive 2D images. The integrated use of the 3D and2D techniques allows for ease of polyp detection, as well as characterization of lesion density and location. The 2D images also allow for evaluation of the extra colonic structures. Adequate bowel preparation and gaseous distension of the colon are essential to ensure a successful examination. Patients typically undergo full cathartic preparation along with a clear liquid diet the day before the study, similar to the requirements for colonoscopy. Tagging of residual solid stool and fluid with barium and / or iodine oral contrast agents is being increasingly used and validated in large trials. At CT, a small-caliber rectal catheter is inserted into the rectum, followed by automated or manual insufflations of room air or carbon dioxide. Intravenous contrast generally is not given to patients undergoing screening but can be helpful in some patients with more advanced symptoms. Typically, the entire procedure on the CT table takes approximately10 minutes, with no sedation or recovery time needed. Research into non cathartic approaches to minimize the bowel preparation is underway, but this technique has not yet been validated in a multicenter screening trial [33-35]. However, under conditions where same-day or next-day referral for colonoscopy would be possible, one drawback of non-cathartic CTC is that a cathartic bowel preparation would still be required prior to removal of polyps.
Efficacy of CTC
The test performance characteristics of CTC for polyp detection are derived by using optical colonoscopy (OC) as the reference standard. Early single-center CTC clinical trials involving small, polyp-rich cohorts provided encouraging initial results and served as proof of concept that paved the way for larger multicenter screening trials [36-38]. Two early trials by Cotton et al. and Rocky et al. included approximately 600 subjects each and observed per-patient sensitivity for large polyps of 55% and 59%, respectively [39, 40]. However, these 2 studies did not evaluate screening in an asymptomatic population, nor did they apply the latest CTC techniques. A recently initiated multi-institutional screening trial using more advanced CTC techniques demonstrated more favorable performance. Pickhardtet et al. studied 1,233 asymptomatic adults and introduced the techniques of stool tagging and primary 3D polyp detection, neither of which were used in the 2 earlier multi-institutional trials [41]. This trial reported 94% sensitivity for large adenomas, with a per-patient sensitivity for adenomas 6 mm of 89%. It was demonstrated that specificity (when polyps were matched for size) was 97.4% for lesions 1 cm but declines to 84.5% for all lesions to all lesions 6 mm.
In 2005, two meta-analyses reviewed the cumulative published CTC performance data, including both high-risk and screening cohorts with one analysis representing 33 studies on 6,393 patients [42, 43]. On a per-patient basis, pooled CTC sensitivity and specificity for large (10 mm) polyps was found to be 85% to 93% and 97%, respectively. Pooled sensitivity and specificity for detection of small polyps (6 to 9 mm) was 70% to 86% and 86% to 93%, respectively. Of note, the pooled CTC sensitivity for invasive CRC was 96%, comparable with the reported sensitivity for optical colonoscopy [35, 42, 44].
There also are a number of CTC trials currently in progress within the United States and Europe. Initial results from smaller screening trials utilizing 3D polyp detection by Cash et al. and Graser et al. have shown CTC performance characteristics similar to that of Pickhardt et al., providing at least a measure of independent validation for this screening technique [45, 46]. Also of particular interest is the recently completed ACRIN Study 6664, a National CT Colonography Trial, which is sponsored and funded by the National Cancer Institute. The primary aim of this trial was to assess CTC performance for large adenomas and advanced neoplasia in a large screening cohort of 2,500 patients across 15 institutions. State-of-the-art techniques included oral contrast tagging, colonic dissention with automated carbon dioxide delivery, multi detector row CT (16 slice) with thin collimation, and both 2D and 3D polyp detection on dedicated CTC software systems. Specialized training and achievement of a high level of expertise were required of the radiologists prior to participation in the study. Preliminary findings announced at the 2007 annual meeting of ACRIN on September 28, were consistent with other recent studies using state of the art techniques.
Beyond validation, a recent study demonstrated the efficacy of CTC to select patients who would benefit from therapeutic polypectomy. Kim et al. recently reported comparative results from primary CTC (with selective recommendation for therapeutic colonoscopy) and primary OC screening arms among 120 and 163 mostly asymptomatic adults, respectively [14, 47]. Although this study did not randomize participants to CTC versus OC, apart from a slightly higher proportion of individuals with a family history in the OC group, the 2 groups were similar. Similar rates of advanced neoplasia were found in each group with 3.2% in the CTC group and 3.4% in the OC group [47].
CTC has a number of potential advantages compared with conventional fiber optic colonoscopy. It is a noninvasive technique, requires no sedation, and can be completed in a much shorter time. The majority of studies assessing the relative acceptability of CTC and conventional colonoscopy in patients who have undergone both tests on the same day have demonstrated a preference for CTC. CTC is also safer than colonoscopy. Colon perforation occurs in 1:1000 patients who undergo conventional colonoscopy, and the mortality rate is 1: 5000. Although experience with CTC is much more limited, no CTC related colon perforations have been reported, and it is likely that the morbidity and mortality associated with CTC will be similar to those for air contrast barium enema (perforation rate of 1:10000 and mortality rate of 1:50000). CTC has the potential to examine the colon completely in nearly all patients, whereas conventional colonoscopy is incomplete in 5–10% of average patients. In addition, CTC eliminates blind spots that can be problematic with conventional colonoscopy. For example, CT colonography is able to demonstrate lesions behind haustral folds and beyond bends in the colon because of its ability to provide an endo luminal view of the colon in both forward and reverse directions and its ability to demonstrate the colon in both 2D and 3D perspectives. For the same reasons, localization of colonic lesions is more accurate with CTC than with fiber optic colonoscopy.
The National CT Colonography Trial of the American College of Radiology Imaging Network was designed to assess the accuracy of CT colonography in detecting histologically confirmed, large colorectal adenomas and cancers (≥ 10 mm in diameter), with optical colonoscopy (the current clinical standard for colorectal cancer screening) and histologic review used as the reference standard" In this study of asymptomatic adults, CT colonographic screening identified 90% of subjects with adenomas or cancers measuring 10 mm or more in diameter [48].
Benefits of CTC
CTC provides a time-efficient procedure with good accuracy and minimal invasiveness. No sedation or recovery time is required, nor is a chaperone needed to provide transportation after the procedure. Time permitting, patients can return to work on the same day.
CTC is a minimally invasive test; the risk for colonic perforation during screening is extremely low. In the collective experience of the International Working Group on Virtual Colonoscopy, there were no cases of perforation in over 11,000 screening CTC examinations, and out of nearly 22,000 total CTC examinations (screening and diagnostic), there was only one symptomatic perforation, corresponding to a symptomatic perforation rate of 0.005% [49]. Colonic distension with low-pressure carbon dioxide delivery may be safer than insufflations of room air. Rates of perforation are part of the quality metrics being collected by the ACR. CTC produces an image not only of the colon but also the upper and lower abdomen, there is a chance that incidental extra colonic findings will be observed.
Although the overall rates of extra colonic findings have been reported to range from 15% to 69%, the incidence of clinically significant extra colonic findings at CTC has ranged from 4.5% to 11% in various patient cohorts [50-53]. In an asymptomatic screening population, the incidence of unsuspected but potentially important extra colonic findings is approximately 4.5%, but findings of minimal or moderate potential clinical significance, such as cholelithiasis (6%) and nephrolithiasis (8%), are more common [53].
Standardization of the evolving technology and consensus related to the reporting of findings will be essential for effective implementation of CTC screening. A consensus statement of a standardized reporting structure for CTC findings was recently published, modeled after the Breast Imaging Reporting and Data System's (BI-RADS) reporting of mammography [54]. This reporting structure, termed the "CT Colonography Reporting and Data System (C-RADS)" describes how to report lesion size, morphology, and location, with a summary category score per patient.
Screening of average-risk adults with CTC should commence at age 50 years. The interval for repeat exams after a negative CTC has not been studied, and is uncertain. However, if current studies confirm the previously reported high sensitivity for detection of cancer and of polyps 6 mm, it would be reasonable to repeat exams every 5 years if the initial CTC is negative for significant polyps until further studies are completed and are able to provide additional guidance. CTC surveillance could be offered to those patients who would benefit from screening but either decline colonoscopy or who are not good candidates for colonoscopy for one or more reasons.
Ultimately, the important issue is not whether CTC is better or worse than colonoscopy or other screening tests for colorectal polyp detection. The objective of colorectal cancer screening is to prevent cancers and save lives. The unfortunate fact is that current compliance for colorectal cancer screening programs is very low, and patients continue to die from colorectal cancers that could have been prevented. If implementation of a technique like CTC can improve colorectal cancer screening compliance, then many lives can be saved.
Limitations of CTC
CTC requires the same full cathartic bowel preparation and restricted diet as colonoscopy, which may decrease patient adherence. As an "imaging-only" non-therapeutic evaluation of the colon, patients with polyps of significant size will require therapeutic colonoscopy for subsequent polypectomy. Thus, it is possible to offer same-day polypectomy to patients for whom colonoscopy is recommended without the need for additional bowel preparation, although this convenience for patients requires coordination between radiology and gastroenterology departments [55]. CTC is similar to endoscopy and DCBE with respect to the quality of interpretation being highly operator dependent, and thus initiatives towards training and certification are important. Detection of flat lesions has been variable, ranging from sensitivities of 13% to 65% in early CTC studies to 80% when using multi detector CT and combined 3D-2D polyp detection [56, 57]. However, debate continues over the prevalence and significance of flat colorectal lesions [58-60].
There is controversy over the long-term potential harms associated with radiation dose effects from CT examinations. One aspect of this controversy relates to risk-estimation models, and the other pertains to the long-term risk of cancer from single and repeated medical imaging exposures [61, 62]. In a recent position statement issued by the Health Physics Society, the health effects of low-dose radiation exposure (defined as below 50 to 100 mSv—a threshold many times higher than typical CTC levels) were considered to be "either too small to be observed or are non-existent [63]. Nevertheless, although this risk may be theoretical, there is a growing concern that more individuals are receiving multiple diagnostic evaluations with ionizing radiation over a lifetime and that for some individuals the doses over a lifetime can reach levels that are sufficiently high to be of concern. It is important to put these issues into context with respect to screening with CTC. Using the linear, no-threshold radiation-risk estimate, a CTC examination in a 50-year-old individual with an estimated organ dose to the colon of 7 to 13 mSv (65 mAs) is estimated to add an additional 0.044% to the lifetime risk of colon cancer [64]. Because organ radio sensitivity declines with increasing age, this organ dose is halved for the same examination taking place at age 70 years. In this same evaluation, the additional lifetime risk of cancer in any site associated with a single CTC examination at age 50 years was 0.14%, although the authors stated with optimized techniques this risk could be reduced by a factor of 5- to10-fold. More efficient dose protocols using 50 mAs on 4D CT, similar to the ACR- defined protocols, have demonstrated decreased estimated organ dose ranges of 5 to 8 mSv [65].
While acknowledging there is uncertainty about potential harms from single or multiple CTC screening examinations, current ACR quality metrics for CTC define low-dose parameters as a best practice for minimizing risk to patients [66]. The management of CTC findings is an important part of a CTC screening program. At this time, there is consensus that all patients with one or more polyps 10 mm or 3 or more polyps 6 mm should be referred for colonoscopy [67]. The management of patients with fewer polyps (< 3) in which the largest polyps 6 to 9 mm remains controversial. Such polyps are routinely removed if found at OC because of the opportunity and the risk, albeit low, of advanced neoplasia. However, in studies that have been limited to screening cohorts, among individuals whose largest polyp is 6 to 9 mm in size, the prevalence of advanced features tends to be low (3.4% to 6.6%) [68, 69]. At this time, there is ongoing research using CTC surveillance to evaluate the natural history of polyps in this size range. Based on expert consensus and until further evidence is available to provide additional guidance, a reasonable approach at this time for patients with 6 to 9 mm polyps identified on CTC is to recommend therapeutic colonoscopy. Patients who decline referral to colonoscopy or who are not good candidates for colonoscopy should be offered surveillance with CTC.
Optimal management of patients whose largest polyp is <6 mm detected on CTC is uncertain. Experts from the American Gastroenterological Association, the American College of Gastroenterology and the ACR have reported a range of policies on how to handle these lesions [70, 71]. There is general agreement that the risk of advanced features in patients whose largest polyp is 5 mm is very low. In a recent study that is able to provide this estimate in a screening cohort, the prevalence of advanced neoplasia in patients whose largest polyp was 5 mm was 1.7% [69]. At this time, there is a pressing need for multidisciplinary consensus on the reporting and clinical management of patients whose largest polyp is < 6 mm.
Obstacles to widespread use of CTC for colorectal cancer screening
Several obstacles to the widespread use of CTC for colorectal cancer screening are evident. The most important obstacle is that the cost of CTC as a screening procedure is not covered by the vast majority of third party payers. Currently in the United States, individuals who undergo CTC for screening purposes pay for the study themselves. Thus, a large percentage of individuals needing colorectal cancer screening cannot afford CTC. Other important issues related to the widespread use of CTC for colorectal cancer screening are the need for reader training and the limited opportunities currently available to acquire it. Experience with CTC trials has shows that interpretation of these examinations is associated with a learning curve. In terms of detection of colon cancer and advanced neoplasia, which is the primary goal of screening for CRC and adenomatous polyps, recent data suggest CTC is comparable to OC for the detection of cancer and polyps of significant size when state-of-the-art techniques are applied. Based on the accumulation of evidence since the time CTC was introduced, the expert panel concludes that there are sufficient data to include CTC as an acceptable option for CRC screening.
Technique of virtual colonoscopy
The integrated use of both 3D and 2D techniques allows polyp detection as well as characterization. However excellent bowel preparation is the prerequisite for imaging the colon. Thin section helical CT of the abdomen and pelvis in the prone and supine position are essential (Figures 1 and 2).
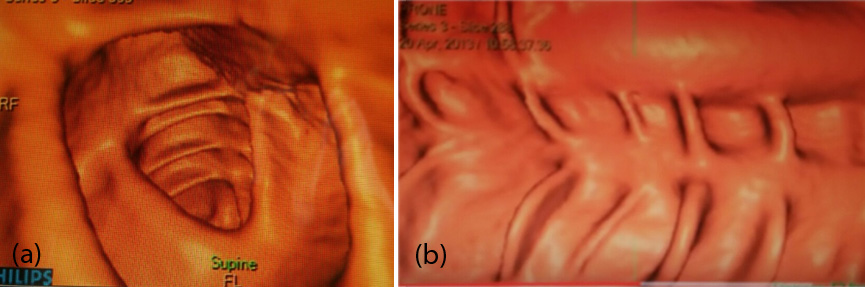
Figure 1: (a) Axial view of CT colonoscopy shows normal haustral folds and mucosal surface; (b) Longitudinal palette view of CT colonoscopy demonstrates spread out lumen.
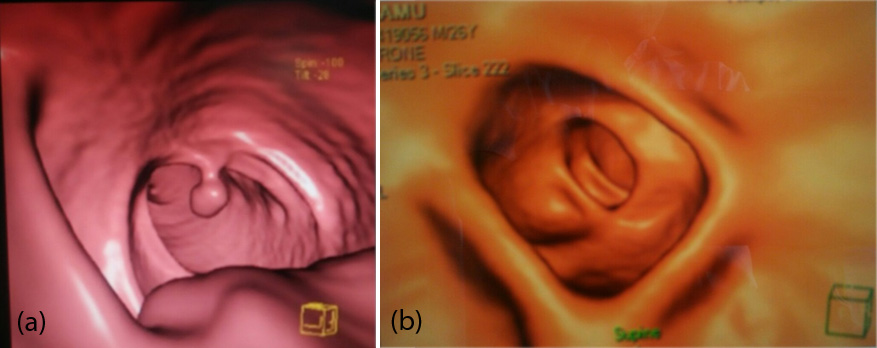
Figure 2: (a) Axial view of virtual colonoscopy reveals a pedunculated polyp; (b) CT colonoscopy in axial view shows a sessile polyp at 7o position.
Only liquid diet is given on the day before the procedure. Bowel preparation (1 sachet of Peglec dissolved in 1.5 L of water and given at the rate of 100 ml every 10 min) is started at least 6 h before patient is taken for CT scan. Oral Domperidone 1 tablet is administered half an hour prior to starting bowel preparation. If patient shows any signs or symptoms of intestinal colic, the preparation will be discontinued. Approximately 2 liters of room air is used for colon insufflations. About forty puffs are required for adequate distension as the patient starts to feel uncomfortable. Acquisition of the abdomen and pelvic scans is performed within a single breath hold in the supine position. The primary search for colorectal polyps and cancers employs 3D endo luminal search technique and correlation with 2D sections to identify the extramural component. Mobility of the lesion in supine and prone images indicates residual fecal material, pedunculated polyp or a rotating colon segment.
Conclusion
The objective of colorectal cancer screening is to detect cancer in its earliest stage to arrest the progress and offer long time survival and even cure. Community based outreach screening program would improve patient education and help overcome the hurdles in collection of stool samples and embarrassment of colonoscopy. Many lives can be saved if implementation of a technique like CTC can improve colorectal cancer screening program. However, limitations preclude the use of CTC as a screening modality in the Indian context. A large percentage of individuals needing colorectal cancer screening in India cannot afford CTC. Widespread use of CTC for colorectal cancer screening is limited due to the need for reader training and the limited opportunities currently available. It is an acceptable option if made available to the population in select patients.
Acknowledgements
Krishna Institute of Medical Sciences (KIMS), Secunderabad.
Conflicts of interest
Author declares no conflicts of interest.
References
[1] Mohandas KM. Colorectal cancer in India: Controversies, enigmas and primary prevention. Indian J Gastroenterol. 2011; 30(1):3-6.
[2] Jussawalla DJ, Gangadharan P. Cancer of the colon: 32 years of experience in Bombay, India. J Surg Oncol.1977; 9: 607–622.
[3] Javid G, Zargar SA, Rather S, Khan AR, Khan BA, et al. Incidence of colorectal cancer in Kashmir Valley, India. Indian J Gastroenterol. 2011; 30(1):7–11.
[4] Swaminathan R, Selvakumaran R, Esmy PO, Sampath P, Ferlay J, et al. Cancer pattern and survival in a rural district in South India. Cancer Epidemiol. 2009; 33(5):325–331.
[5] Yeole BB. Trends in cancer incidence in esophagus, stomach, colon, rectum and liver in males in India. Asian Pac J Cancer Prev. 2008; 9(1):97–100.
[6] Rastogi T, Devesa S, Mangtani P, Mathew A, Cooper N, et al. Cancer incidence rates among South Asians in four geographic regions: India, Singapore, UK and US. Int J Epidemiol. 2008; 37(1):147–160.
[7] Bray F, Ren JS, Masuyer E, Ferlay J. Global estimates of cancer prevalence for 27 sites in the adult population in 2008. Int J Cancer. 2013; 132(5):1133–1145.
[8] Curado MP. Cancer incidence in five continents, Volume IX. IARC Sci Publ. 2008; 160:1–837.
[9] Shike M, Winawer SJ, Greenwald PH, Bloch A, Hill MJ, et al. Primary prevention of colorectal cancer. The WHO Collaborating Centre for the Prevention of Colorectal Cancer. Bull World Health Organ. 1990; 68(3):377–385.
[10] American Cancer Society. Cancer facts and figures 2010. Atlanta, Ga: American Cancer Society, 2010.
[11] Ries LAG, Eisner MP, Kosary CL, Hankey BF, Miller BA, et al. SEER cancer statistics review, 1975–2002. Bethesda, MD. National Cancer Institute. 2005.
[12] Edwards BK, Howe HL, Ries LA, Thun MJ, Rosenberg HM, et al. Annual report to the nation on the status of cancer, 1973–1999, featuring implications of age and aging on US cancer burden. Cancer. 2002; 94(10):2766–2792.
[13] Imperiale TF, Wagner DR, Lin CY, Larkin GN, Rogge JD, et al. Results of screening colonoscopy among persons 40 to 49 years of age. N Engl J Med. 2002; 346(23):1781–1785.
[14] Key TJ, Appleby PN, Spencer EA, Travis RC, Allen NE, et al. Cancer incidence in British vegetarians. Br J Cancer. 2009; 101(1):192–197.
[15] van Duijnhoven FJ, Bueno-De-Mesquita HB, Ferrari P, Jenab M, Boshuizen HC, et al. Fruit, vegetables, and colorectal cancer risk: The European prospective. Investigation into Cancer and Nutrition. Am J Clin Nutr. 2009; 89(5):1441–1452.
[16] Fuchs CS1, Giovannucci EL, Colditz GA, Hunter DJ, Speizer FE, et al. A prospective study of family history and the risk of colorectal cancer. N Engl J Med. 1994; 331(25):1669–1674.
[17] Levin B, Rozen P, Young GP. How should we follow up colorectal premalignant conditions? In: Rozen P, Young G, Levin B, et al. Colorectal Cancer in Clinical Practice: Prevention, Early Detection, and Management. London, UK: Martin Dunitz. 2002; 67–76.
[18] Winawer SJ, Fletcher RH, Miller L, Godlee F, Stolar MH, et al. Colorectal cancer screening: Clinical guidelines and rationale. Gastroenterology. 1997, 112(2):594–642.
[19] Mellath MK. Colorectal cancer screening:current stateus. Trop Gastroenterol. 2014; 35(1):1–4.
[20] ICMR consensus document for management of colorectal cancer. 2014.
[21] Ries LAG, Melbert D, Krapcho M, Mariotto A, Miller BA, et al. SEER Cancer Statistics Review. Bethesda MD: National Cancer Institute. 2007; 1975–2004.
[22] Espey DK, Wu XC, Swan J, Wiggins C, Jim MA, et al. Annual report to the nation on the status of cancer, 1975–2004, Featuring cancer in American Indians and Alaska Natives. Cancer. 2007; 110(10):2119–2152.
[23] Selby JV, Friedman GD, Quesenberry CP Jr, Weiss NS. A case-control study of screening sigmoidoscopy and mortality from colorectal cancer. N Engl J Med. 1992; 326(10):653–657.
[24] Mandel JS, Church TR, Bond JH, Ederer F, Geisser MS, et al. The effect of fecal occult-blood screening on the incidence of colorectal cancer. N Engl J Med. 2000; 343(22):1603–1607.
[25] Meissner HI, Breen N, Klabunde CN, Vernon SW. Patterns of colorectal cancer screening uptake among men and women in the United States. Cancer Epidemiol Biomarkers Prev. 2006; 15(2):389–394.
[26] Smith RA, Cokkinides V, Eyre HJ. Cancer screening in the United States, 2007: A review of current guidelines, practices, and prospects. CA Cancer J Clin. 2007; 57(2):90–104.
[27] O'Brien MJ, Winawer SJ, Zauber AG, Gottlieb LS, Sternberg SS, et al. The National Polyp Study. Patient and polyp characteristics associated with high-grade dysplasia in colorectal adenomas. Gastroenterology. 1990; 98(2):371–379.
[28] Bond JH. Colon polyps and cancer. Endoscopy. 2003; 35(1):27–35. [29] Winawer SJ, Fletcher RH, Miller L, Godlee F, Stolar MH, et al. Colorectal cancer screening: Clinical guidelines and rationale. Gastroenterology. 1997; 112(2):594–642.
[30] Winawer SJ. Natural history of colorectal cancer. Am J Med. 1999; 106(1A):3S–6S.
[31] Lieberman DA, Weiss DG. One-time screening for colorectal cancer with combined fecal occult-blood testing and examination of the distal colon. N Engl J Med. 2001; 345(8):555–560.
[32] Smith RA, Cokkinides V, Brooks D, Saslow D, Brawley OW. Cancer Screening in the United States, 2010: A review of current American Cancer Society guidelines and issues in cancer screening. Ca Cancer J Clin. 2010; 60(2):99–119.
[33] Zalis ME, Perumpillichira J, Del Frate C, Hahn PF. CT colonography: digital subtraction bowel cleansing with mucosal reconstruction initial observations. Radiology. 2003; 226(3):911–917.
[34] Iannaccone R, Laghi A, Catalano C, Mangiapane F, Lamazza A, et al. Computed tomographic colonography without cathartic preparation for the detection of colorectal polyps. Gastroenterology. 2004; 127(5):1300–1311.
[35] Lefere P, Gryspeerdt S, Marrannes J, Baekelandt M, Van Holsbeeck B. CT colonography after fecal tagging with a reduced cathartic cleansing and a reduced volume of barium. AJR Am J Roentgenol. 2005; 184(6):1836–1842.
[36] Hara AK, Johnson CD, Reed JE, Ahlquist DA, Nelson H, et al. Detection of colorectal polyps with CT colography: Initial assessment of sensitivity and specificity. Radiology. 1997; 205(1):59–65.
[37] Fenlon HM, Nunes DP, Schroy PC, Barish MA, Clarke PD, et al. A comparison of virtual and conventional colonoscopy for the detection of colorectal polyps. N Engl J Med. 1999; 341(20):1496–1503.
[38] Yee J, Akerkar GA, Hung RK, Steinauer-Gebauer AM, Wall SD, et al. Colorectal neoplasia: Performance characteristics of CT colonography for detection in 300 patients. Radiology. 2001; 219(3):685–692.
[39] Cotton PB, Durkalski VL, Pineau BC, Palesch YY, Mauldin PD, et al. Computed tomographic colonography (virtual colonoscopy): A multicenter comparison with standard colonoscopy for detection of colorectal neoplasia. JAMA. 2004; 291(14):1713–1719.
[40] Rockey DC, Paulson E, Niedzwiecki D, Davis W, Bosworth HB, et al. Analysis of air contrast barium enema, computed tomographic colonography, and colonoscopy: prospective comparison. Lancet. 2005; 365(9456):305–311.
[41] Pickhardt PJ, Choi JR, Hwang I, Butler JA, Puckett ML, et al. Computed tomographic virtual colonoscopy to screen for colorectal neoplasia in asymptomatic adults. N Engl J Med. 2003; 349(23):2191–2200.
[42] Halligan S, Altman DG, Taylor SA, Mallett S, Deeks JJ, et al. CT colonography in the detection of colorectal polyps and cancer: systematic review, meta-analysis, and proposed minimum data set for study level reporting. Radiology. 2005; 237(3):893–904.
[43] Mulhall BP, Veerappan GR, Jackson JL. Meta-analysis: computed tomographic colonography. Ann Intern Med. 2005; 142(8):635–650.
[44] Farrar WD, Sawhney MS, Nelson DB, Lederle FA, Bond JH. Colorectal cancers found after a complete colonoscopy. ClinGastroenterolHepatol. 2006; 4(10):1259–1264.
[45] Cash B, Kim C, Cullen P. Accuracy of computed tomographic colonography for colorectal cancer screening in asymptomatic individuals. Gastroenterology. 2006; 130:A46.
[46] Graser A, Kolligs F, Kramer H. Results from the "Munich Colorectal Cancer Prevention Trial": Comparison of low-dose 64-MDCT colonography and video colonoscopy in a screening population. European Society of Gastrointestinal and Abdominal Radiology/Society of Gastrointestinal Radiologists. 2006; (26)19–23.
[47] Kim DH, Pickhardt PJ, Taylor AJ, Leung WK, Winter TC, et al. CT colonography versus colonoscopy for the detection of advanced neoplasia. N Engl J Med. 2007; 357(14):1403–1412.
[48] Johnson CD, Chen MH, Toledano AY, Heiken JP, Dachman A, et al. Accuracy of CT Colonography for Detection of Large Adenomas and Cancers N Engl J Med. 2008; 359(12):208–1217.
[49] Pickhardt PJ. Incidence of colonic perforation at CT colonography: review of existing data and implications for screening of asymptomatic adults. Radiology. 2006; 239(2):313–316.
[50] Hara AK, Johnson CD, MacCarty RL, Welch TJ. Incidental extracolonic findings at CT colonography. Radiology. 2000; 215(2):353–357.
[51] Gluecker TM, Johnson CD, Wilson LA, Maccarty RL, Welch TJ, et al. Extracolonic findings at CT colonography: evaluation of prevalence and cost in a screening population. Gastroenterology. 2003; 124(4):911–916.
[52] Yee J, Kumar NN, Godara S, Casamina JA, Hom R, et al. Extracolonic abnormalities discovered incidentally at CT colonography in a male population. Radiology. 2005: 236(2):519–526.
[53] Pickhardt PJ, Taylor AJ. Extracolonic findings identified in asymptomatic adults at screening CT colonography. AJR Am J Roentgenol. 2006; 186(3):718–728.
[54] Zalis ME, Barish MA, Choi JR, Dachman AH, Fenlon HM, et al. CT colonography reporting and data system: a consensus proposal. Radiology. 2005; 236(1):3–9.
[55] Pickhardt PJ, Taylor AJ, Kim DH, Reichelderfer M, Gopal DV, et al. Screening for colorectal neoplasia with CT colonography: initial experience from the 1st year of coverage by third-party payers. Radiology. 2006; 241(2):417–425.
[56] Fidler JL, Johnson CD, MacCarty RL, Welch TJ, Hara AK, et al. Detection of flat lesions in the colon with CT colonography. Abdom Imaging. 2002; 27(3):292–300.
[57] Pickhardt PJ, Nugent PA, Choi JR, Schindler WR. Flat colorectal lesions in asymptomatic adults: implications for screening with CT virtual colonoscopy. AJR Am J Roentgenol. 2004; 183(5):1343–1347.
[58] Bond JH. Doubling time of flat and polypoid colorectal neoplasms: defining the adenoma-carcinoma sequence. Am J Gastroenterol. 2000; 95(7):1621–1623.
[59] Zauber AG, O‘Brien MJ, Winawer SJ. On finding flat adenomas: is the search worth the gain? Gastroenterology. 2002; 122(3):839–840.
[60] Park SH, Lee SS, Choi EK, Kim SY, Yang SK, et al. Flat colorectal neoplasms: definition, importance, and visualization on CT colonography. AJR Am J Roentgenol. 2007; 188(4):953–959.
[61] Amis ES Jr, Butler PF, Applegate KE, Birnbaum SB, Brateman LF, et al. American College of Radiology white paper on radiation dose in medicine. J Am Coll Radiol. 2007; 4(5):272–284.
[62] Brenner DJ, Hall EJ. Computed tomography—an increasing source of radiation exposure. N Engl J Med. 2007; 357(22):2277–2284.
[63] Health Physics Society, Radiation Risk in Perspective. Position statement of the Health Physics Society. 2004.
[64] Brenner DJ, Georgsson MA. Mass screening with CT colonography: Should the radiation exposure be of concern? Gastroenterology. 2005; 129(1):328–337.
[65] Macari M, Bini EJ, Xue X, Milano A, Katz SS, et al. Colorectal neoplasms: Prospective comparison of thin-section low-dose multi-detector row CT colonography and conventional colonoscopy for detection. Radiology. 2002; 224(2):383–392.
[66] American College of Radiology, ACR Practice Guideline for the Performance of Computed Tomography (CT) Colonography in Adults. Reston, VA: American College of Radiology. 2006.
[67] Kim DH, Pickhardt PJ, Hoff G, Kay CL. Computed tomographic colonography for colorectal screening. Endoscopy. 2007; 39(6):545–549.
[68] Kim DH, Pickhardt PJ, Taylor AJ. Characteristics of advanced adenomas detected at CT colonographic screening: implications for appropriate polyp size thresholds for polypectomy versus surveillance. AJR Am J Roentgenol. 2007; 188(4):940–944.
[69] Moravec M, Lieberman D, Holub J. Rate of advanced pathologic features in 6–9 mm polyps in patients referred for colonoscopy screening. Gastrointest Endosc. 2007; 65:822.
[70] Rex DK, Lieberman D. ACG colorectal cancer prevention action plan: update on CT-colonography. Am J Gastroenterol. 2006; 101(7):1410–1413.
[71] Rockey DC, Barish M, Brill JV, Cash BD, Fletcher JG, et al. Standards for gastroenterologists for performing and interpreting diagnostic computed tomographic colonography. Gastroenterology. 2007; 133(3):1005–1024.