Full Text
Laryngospasm is the one of the common causes of airway obstruction in children perioperatively. Laryngospasm is a protective reflex closure of the glottis as a result of abnormal stimulus. Most of the time, the resulting hypoxia abolishes the reflex and the spasm tends to be self-limited. Laryngospasm causes about 40% of post-extubation airway obstruction. As per various studies, the incidence [1] of laryngospasm in paediatric population ranges from 0.04 to 14%.
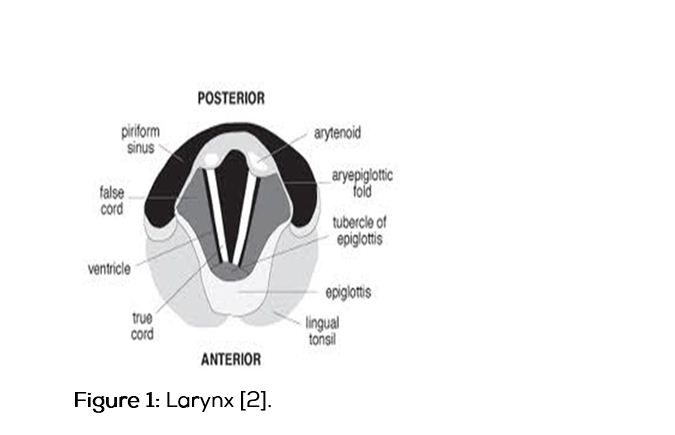
Physiology: Laryngeal reflex (glottic closure reflex; quick protective response), laryngeal closure (vocal cord adduction, short lived) protects lungs from aspiration of foreign material. laryngospasm: a prolonged form of vocal cord adduction (closure of the true vocal cords alone or the true and false vocal cords) [2].
Pathophysiology: Fink described two types of laryngospasm: i. Expiratory stridor, which is an active closure of the glottis secondary to adductor spasm. ii. Inspiratory stridor, which is a passive closure of the glottis secondary to a Ball-Valve mechanism. Adductor spasm can be treated by continuous positive airway pressure (CPAP). Ball-valve effect by forced inflation of the pharynx merely distends the pyriform fossa and presses the arytenoid against each other [3-5].
Clinical features: Stridor- A harsh high pitched noise. Use of accessory muscles causes tracheal tug, intercostal, subcostal and sternal recession. Rocking respiratory movements are paradoxical, hypoxia -61% (desaturation and cyanosis), bradycardia-6%, obstructive negative pressure pulmonary edema-4%, cardiac arrest-0.5% [4, 5].
Clinical diagnosis: Partial spasm (Incomplete airway obstruction)-An audible inspiratory or expiratory sound-characteristic stridulous noise (early diagnosis by precordial stethoscope). Mismatch between the patient’s respiratory effort and the disproportionally small movements of the reservoir bag when the patient is breathing through pediatric circuit (paradoxical respiratory movements of the thorax and abdomen, ineffective chest wall movement, exaggerated abdominal movement) and tracheal tug and reduced tidal volume.
Complete spasm (complete airway obstruction)-severe laryngospasm: Absence of breath sounds despite adequate chest wall movement (sine qua non), paradoxical respiratory movements and tracheal tug.
Risk factors: Anesthetic factors are inadequate depth of anesthesia, irritant volatile agents, inexperienced anesthesiologist. Local stimulation of the larynx by saliva, blood, vomitus, foreign bodies and instruments (laryngoscope, LMA’S and suction catheters) [4-6].
External factors are paediatric population, upper respiratory tract infection (URTI), hyper reactive airways, smokers, surgical stimulation, upper air way procedures, moving/ transferring patient, anal/ cervical stimulation (the Brewer-Luckhardt reflex).
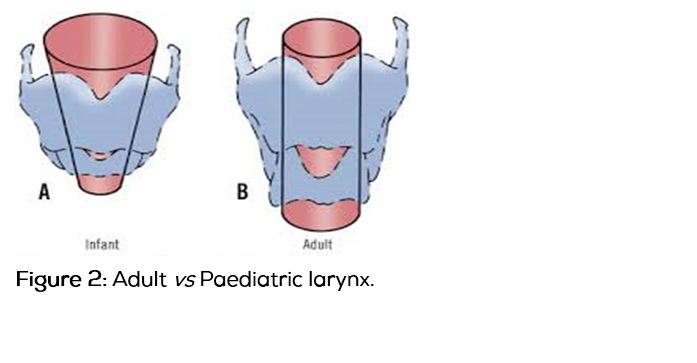
|
Infant
|
Adult
|
|
Large occiput
|
Flat occiput
|
|
Narrow finger like epiglottis Ω that is funnel shaped
|
· Normal tongue
· Flatter Epiglottis
· Narrowest at cords
|
|
Cricoid at C4 level
|
Cricoid at C7
|
|
· Vocal cords angled
· Obligate nasal breathers
|
Cords perpendicular to tracheal axis
|
Table 1: Pediatric vs Adult airway.
Prevention of laryngospasm: Laryngospasm can be prevented by identifying and treating the risk factors preoperatively. Induction may be done with newer inhalational agents like sevoflurane, IV induction may done with propofol which are less irritant to the airway when compared to the other agents available. IV line insertion should be done after deep anesthesia. Premedication with anticholinergics reduces the sectretions in the airway. Laryngospasm can be prevented by intubating after adequate depth and relaxation (using muscle relaxants) [7-9].
Emergence phase: Position the patient on his side with head down to prevent secretions and gastric contents irritating the airway. Suction of secretions from the oropharynx should be gentle and avoid further stimulation. Giving lignocaine 1mg/kg or propofol 0.5mg/kg IV at the time of extubation also protects from the laryngospasm [10]. Producing the artificial cough at the time of extubation will also help in reducing the incidence of laryngospasm (artificial cough tech.-positive pressure inflation of the lungs before extubation is followed by forced exhalation after extubation which expels any secretions, this in turn decreases laryngospasm). Extubation should be done when the patient is fully awake.
Management [6-10]: Management should be aimed at maintaining the oxygenation by early recognition and prompt treatment. Call for help as mild spasm can progress rapidly to complete air way obstruction and cyanosis. Identify and remove the offending stimulus. Switch to 100% O2 via circuit. Apply firm jaw thrust–pressing on the laryngospasm notch–this may help in relieving the laryngospasm due to a combination of airway opening and stimulation. If the spasm is not relieved apply CPAP (100% O2) attempt gentle bag mask ventilation (risk of stomach inflation). Then deepen anesthesia by propofol IV 0.5 mg/kg, then remove any blood/ secretions from airway. If the spasm is still not relieved we can give non-depolarising agent like suxamethonium IV 0.1 to 2 mg/kg to facilitate mask ventilation, if required patient may be intubated to maintain oxygenation. One should always be ready with injection atropine as hypoxic bradycardia is very common. Once laryngospasm has been relieved, inserting NG (Nasogastric) tube to decompress the stomach is to be considered. Patient can develop negative pressure pulmonary edema so they should be monitored closely in the post-operative period.
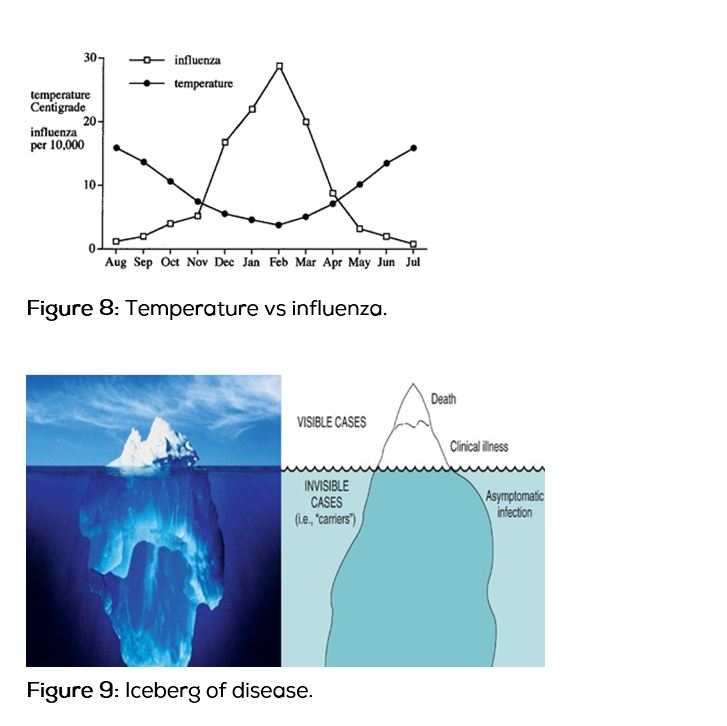
Iceberg phenomenon: What is “iceberg of disease”? It is a metaphor emphasising that for virtually every health problem the number of cases of disease ascertained (those visible) is outweighed by those not discovered (those invisible) (Figure 4). How the iceberg of disease phenomenon thwarts assessment of the true burden of disease, the need for services and the selection of representative cases for epidemiological study [12, 13].

Screening is the application of tests to diagnose disease (or its precursors) in an earlier phase of the natural history of disease (often in well people) than is achieved in routine medical practice. The key to successful screening is a simple test which can be applied to large populations with minimum harm and has a high degree of accuracy. The potential of screening is vast but there are important limitations.
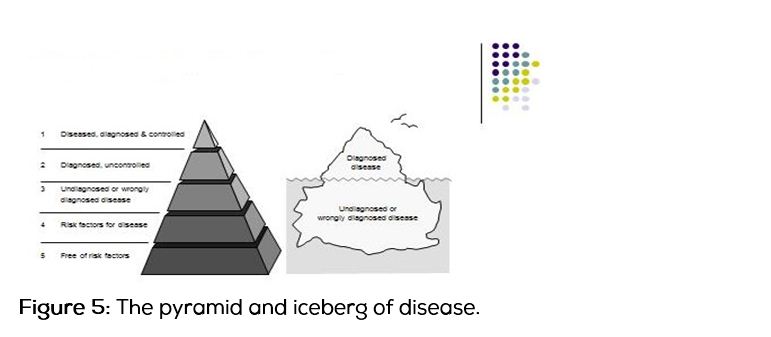
The unmeasured burden of disease (the metaphors of the iceberg and the pyramid): For most health problems there are large numbers of undiscovered or misdiagnosed cases of disease. Serious and killing disorders such as diabetes, atrial fibrillation and hypertension are other good examples of this iceberg phenomenon. Cases that have been correctly diagnosed can be likened to the tip of the iceberg, visible and easily measured. In most diseases, as with the iceberg, the larger presence lurks unseen, unmeasured and easily forgotten.
Figure 5 illustrates this idea and develops the iceberg concept in the form of a pyramid of disease by using its clear structure and shape. Blocks 1 and 2 correspond to the iceberg above the sea-level and 3 to 5 below sea level. Epidemiology that forgets the iceberg phenomenon of disease is weak and potentially misleading
Unidentified cases may be different to identified ones, both in terms of the natural history or spectrum of disease. Where symptoms and disease progression and outcome are related, the undiagnosed cases are likely to be less severe.
When symptoms and signs are not evident in the early stages of disease, as in high blood pressure or chronic glaucoma, undiagnosed cases may be just as severe as diagnosed ones. Epidemiological studies based on selected cases from the tip of the iceberg may give an erroneous view. Prostate cancer based on cases diagnosed in hospital would lead to the view that the disease is usually, if not always, progressive. Unselected cases show that prostatic cancer can in some cases be a static, or slowly progressive, phenomenon. Patients who are at the tip of the iceberg are more likely to have multiple health problems than others.
People with cardio-respiratory problems and diabetes are more likely to be admitted to hospital, than people with only one of these two problems. This is the basis of the bias known as Berkson's bias.
Clinical study and justification
Despite the advances in monitoring and the standard of care in the practice of anaesthesia, laryngospasm remains one of the main causes of respiratory complications seen, especially in children. Laryngospasm is usually easily detected and managed, but may present atypically and/or be precipitated by factors which are not immediately recognised. In this study we have evaluated the seasonal variations in the incidence of laryngospasm. If poorly managed, it has the potential to cause morbidity and mortality such as severe hypoxemia, pulmonary aspiration, and post-obstructive pulmonary oedema.
H(a)-alternate hypothesis: There is variation in incidence of laryngospasm with change in season.
H(o)-null hypothesis: There is no variation in incidence of laryngospasm with change in season.
Aims & objectives: To study the variation in incidence of laryngospasm with seasonal changes.
Materials & methods
Study design is prospective study. Study sample are 1000 patients in the age group from 1 to 5 years. Inclusion criteria are belonging to ASA grade 1 and posted for elective surgeries lasting 30 to 60 minutes. Exclusion criteria are pre-existing upper respiratory tract infection.History of laryngospasm during previous procedures.Airway abnormalities. Surgeries involve the airway, head and neck, congenital anomalies of the airway and more than 2 attempts at intubation. Anaesthesia for all cases was conducted under the supervision of paediatric anaesthesiologist.After obtaining informed consent, the patients were pre medicated with: midazolam, glycopyrollate, ondansetron and fentanyl. Induction and intubation with propofol and vecuronium was done. Maintenance was carried on with: N2O:O2 (3:2), sevoflurane 1%. After the completion of the surgical procedure, the neuromuscular blockade was adequately reversed with neostigmine. After gentle suctioning of the oropharynx and fulfilment of extubation criteria, the trachea was extubated. Laryngospasm if any was treated by: Air way manipulation, applying CPAP to the airway with 100% O2, CPAP + deepening anaesthesia (propofol), CPAP + deepening anaesthesia +Inj. suxamethonium. CPAP + deepening anaesthesia + inj suxamethonium + tracheal intubation.
After obtaining the approval of the institutional ethical committee, this study was undertaken from the period “2009 to 2013” and the study variables are Age, ASA grade, Month/ season of surgery,Surgical procedure, Duration of procedure. Data was analysed by using micro soft excel 2007, SPSS 16.0 and the study tools are pre-tested & pre-designed questionnaire.
Results and discussion
In the present study the incidence of laryngospasm was significantly varied with seasonal changes (P < 0.05) (Table 2 and Table 3). As P value is 0.0000, we reject null hypothesis and will say that there is variation in incidence of laryngospasm with change in season and it is statistically highly significant.
Table 2: Distribution of study population with incidence of laryngospasm.
|
Period of study
|
Total no. of children that underwent surgery
|
No. of children that had an incidence of laryngospasm
|
% of children that had incidence of laryngospasm
|
|
November to February
|
270
|
35
|
12.76%
|
|
March to January
|
350
|
5
|
1.43%
|
|
July to October
|
380
|
12
|
3.16%
|
Note: The Pearson chi-square, uncorrected for continuity is 40.09, P = < .0001
Table 3: Two-way contingency.
|
|
variation of incidence in laryngospasm with change in season
|
|
|
laryngospasm present
|
laryngospasm absent
|
|
March to June
|
5
18.20
( 9.57)
|
345
331.80
( 0.53)
|
350
|
|
July to October
|
12
19.76
( 3.05)
|
368
360.24
( 0.17)
|
380
|
|
November to February
|
35
14.04
( 31.29)
|
235
255.96
( 1.72)
|
270
|
|
|
52
|
948
|
1000
|
Note: χ2 = 46.320, df = 2, χ2/df = 23.16, P(χ2 > 46.320) = 0.0000; Expected values are displayed in italics; individual χ2values are displayed in (parentheses).
p value is highly significant i.e. < 0.05 (yes there is variation of incidence in laryngospasm with change in season),
Null hypothesis: There is no variation of incidence in laryngospasm with change in season as p value is 0.0000, we reject null hypothesis and will say that there is variation of incidence in laryngospasm with change in season statistically.
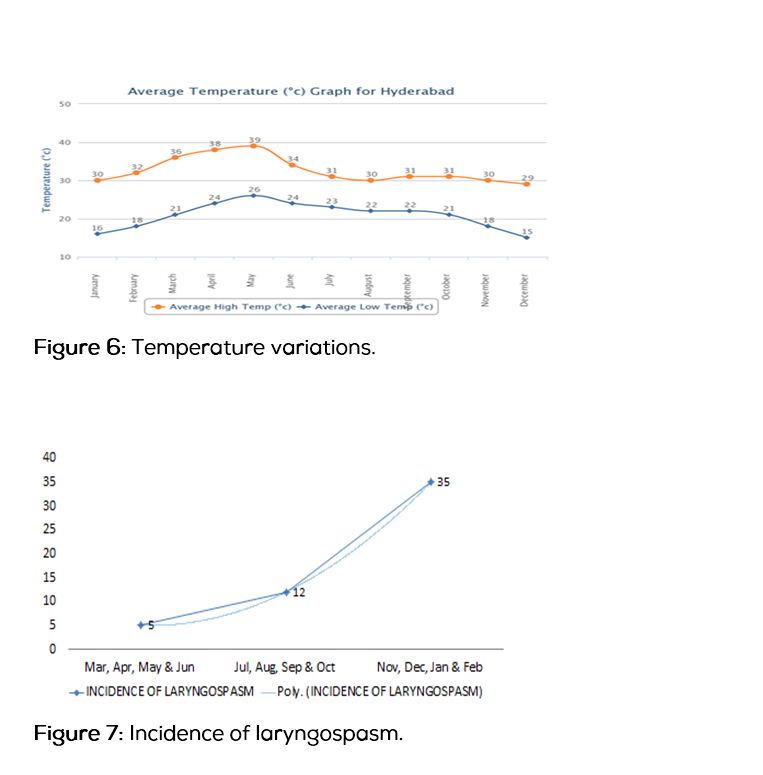
In Hyderabad city, low temperatures (Figure 6) are recorded during winter season, i.e. November, December, January and February. In this study incidence of laryngospasm (Figure 7) is more during this period. Rate of influenza infection (Figure 8) also increases during this period. As temperature falls infection rate increases. According to ice berg phenomenon (Figures 9 and 10) as rate of infections increases, subclinical infections also must be more during this period. It clearly shows there is seasonal variation in the incidence of laryngospasm, during winter season as temperature falls.
Iceberg of disease
The Iceberg of disease is a metaphor emphasising that for virtually every health problem the number of cases of disease ascertained (those visible) is outweighed by those not discovered (those not visible) [12]. Unidentified cases may be different to, identified ones both in terms of the natural history or spectrum of disease. Time between exposure to the agent and the development of disease is called the Incubation period. The Iceberg of disease is a metaphor emphasising that for virtually every health problem the number of cases of disease ascertained (those visible) is outweighed by those not discovered (those not visible). Unidentified cases may be different to identified ones, those both in terms of the natural history or spectrum of disease.
Time between exposure to the agent and the development of disease is called the Incubation period.Natural history of disease is the uninterrupted progression of disease from its initiation by exposure to the casual agents to either spontaneous resolution, containment by the body’s repair mechanisms, or to a clinically detectable problem.
In winter season incidence of influenza is in peak level, this incidence is tip of the iceberg, so subclinical infections [11, 14-18] are also more during this season. Undiagnosed cases are likely to be less severe, in subclinical URTI the airways are hyper reactive but less severe, is the cause for increased incidence of laryngospasm during winter season.
Conclusions
Increased incidence of laryngospasm in winter season is observed due to subclinical upper respiratory tract infections. Anaesthesiologists should be aware of this fact, well in advance, in particular during winter months, for prompt treatment of laryngospasm. Extubation in deeper planes and Injection propofol 0.5 mg/kg before extubation will help to get the response or laryngospasm.
Conflict of Interest
The authors declare no conflict of interest.
References
1. Olsson GL, Hallen B. Laryngospasm during anaesthesia. A computer-aided incidence study in 13,929 patients. Acta Anaesthesiol Scand. 1984; 28(5):567–575.
2. Rex MA. A review of the structural and functional basis of laryngospasm and a discussion of nerve pathways involved in reflexes and its clinical significance in man and animals. Br J Anaesth. 1970; 42(10):891–899.
3. Fink BR. The etiology and treatment of laryngospasm. Anesthesiology. 1956; 17:569–577.
4. Landsman IS. Mechanisms and treatment of laryngospasm. Int anesthesiol clin. 1997; 35(3):67–73.
5. Roy WL, Lerman J. Laryngospam in paediatric anesthesia. Can J Anaesth. 1988; 35(1):93–98.
6. Viswanathan T, KlugerMT, Webb RK, Westhorpe RN. Crisis management during anaesthesia: laryngispasm. Qual Saf Health Care. 2005; 14(3):e3.
7. Burgoyne LL, Anghelescu DL. Intervention steps for treating Laryngospasm in pediatric patients. Paediatr Anaesth. 2008; 18(4):297–302.
8. Chatopadhyay S, Rudra A, Sengupta S. Laryngospasm in pediatric anesthesia: a review. Indian Journal of Anesthesiology Research. 2013; 1:97–104.
9. Roy WL, Lerman J. Laryngospasm in paediatric anaesthesia. Can J Anaesth. 1988; 35(1):93–98.
10. Afshan G, Chohan U, Qamar-Ul-Hoda M, Kamal RS. Is there a role of a small dose of propofol in the treatment of laryngeal spasm? Paediatr Anaesth. 2002; 12(7):625–628.
11. Schreiner MS, O'Hara I, Markakis DA, Politis GD. Do children who.experience laryngospasm have an increased risk of upper.respiratory tract infection? Anesthesiology 1996; 85(3):475–480.
12. Bhopal RS. Concepts of Epidemiology. Oxford, Oxford University Press, 2002, pp317. Available on December 2014 from http://www.oup.co.uk/isbn/0-19-263155-1
13. Monto AS. Viral infections in the community: epidemiology, agents and interventions. Am J Med. 1995; 99(6B):24S–27S.
14. Reina J, Ferres F, Gutierrez O, Ruiz de Gopegui E, Gonalez-Cardenas M. Study of the clinical and epidemiological characteristcs of respiratory infections caused by adenovirus in a paediatric population (1997-2003). An Pediatr (Barc). 2004; 61(2):137–142.
15. Fleming DM, Zamber M, Bartelds AI, DeJong JC. The duration and magnitude of influenza epidemics: a study of surveillance data from sentinel general practices in England, Wales and the Netherlands. Eur J Epidemiol. 1999; 15(5):467–473.
16. van Gageldonk-Lafeber AB, Heijnen ML, Bartelds AI, Peters MF, van der Plas SM, et al. A case control study of Acute Respiratory Tract infection in general practice pts in Netherlands. Clin Infect Dis. 2005; 41(4):490–497.
17. Sarkar S, Biswas R, Gaur SD, Sen PC, Reddy DC. A study on sore throat and beta-haemolytic streptococcal pharyngitis among rural school children in Varanasi with reference to age and Season. Indian J Public Health. 1988; 32(4):190–198.
18. Chibber S, Sabharwal U, Chugh TD. Streptococcal throat carriage in the normal population. Indian J Med Micro. 1985; 3:283–285.