Original Research
2020
December
Volume : 8
Issue : S1
Tocilizumab treatment and outcomes in severe COVID-19 patients: Retrospective study from a tertiary care institute in western India
Peta RK, Panchal HP, Patel A, Parikh S, Khanikar D, Himthani N
Pdf Page Numbers :- 6-10
Ravindra Kumar Peta1,*, Harsha P Panchal1,*, Apurva Patel1, Sonia Parikh1, Duncan Khanikar1, and Nikhil Himthani1
1Department of Medical Oncology, The Gujarat Cancer Research Institute, Ahmedabad, Gujarat, India
*Corresponding authors: Dr. Harsha P Panchal, Prof and HOD, Department of medical oncology, The Gujarat Cancer REsearch Institute, Ahmedabad, Gujarat, India. Mobile: +91 9825940769; Email: drharshapanchal@gmail.com. Dr. Ravindra Kumar Peta (DM Medical oncology), Resident, Department of Medical Oncology, The Gujarat Cancer Research Institute, Ahmedabad, Gujarat, India. Mobile: +91 9023739722; Email: Ravindra0504090@gmail.com
Received 24 September 2020; Revised 9 November 2020; Accepted 17 November 2020; Published 25 November 2020
Citation: Peta RK, Panchal HP, Patel A, Parikh S, Khanikar D, Himthani N. Tocilizumab treatment and outcomes in severe COVID-19 patients: Retrospective study from a tertiary care institute in western India. J Med Sci Res. 2020; 8(S1):6-10. DOI: http://dx.doi.org/10.17727/JMSR.2020/8S1-1
Copyright: © 2020 Peta RK et al. Published by KIMS Foundation and Research Center. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Coronavirus disease 2019 (COVID-19) is a pandemic caused by a novel beta coronavirus severe acute respiratory syndrome coronavirus (SARS-CoV-2). The symptoms range from mild to severe in nature. The severity of respiratory symptoms is due to the cytokine storm. The tocilizumab, interleukin-6 inhibitor, can prevent the cytokine release and decrease the mortality.
Patients and methods: This is a retrospective observational study of 20 COVID-19 positive cases who received tocilizumab.
Results and conclusion: There were 75% males with a mean age of 47.20±9.68 years in our study. 50% had diabetes mellitus, 35% had hypertension, 5% had Chronic kidney disease, 5% had obesity and 5% had hypothyroidism. Mortality was reduced to 65% with tocilizumab administration. There was statistically significant reduction of C- reactive protein (p<0.00052) and IL-6 (interleukin) (p<0.023) after administration of tocilizumab.
Keywords: tocilizumab; interleukin-6; COVID-19; C-reactive protein
Full Text
Introduction In the past century, there were four major global pandemics [1] (1918 pandemic by H1N1 Virus, 1957-58 pandemic by H2N2 virus, 1968 pandemic by H3N2 virus and 2009 pandemic by H1N1pdm09 virus), before the present coronavirus disease 2019 (COVID-19) pandemic. Severe acute respiratory syndrome coronavirus (SARS-CoV-2), a novel beta coronavirus, which caused coronavirus disease 2019(COVID-19) was first identified in China in December 2019 [2-4]. This was followed by global spread causing SARS-CoV-2 pandemic, that was declared by World Health Organization (WHO) on 11, March 2020 [5].
The SARS-CoV-2 causes mild to moderate illness in majority of the patients. Presentation may vary from being asymptomatic to having clinical symptoms like fever and dyspnea, with or without pneumonia [6]. However, some of the patients may develop severe pneumonia with or without Acute respiratory distress syndrome (ARDS) and multiorgan failure requiring urgent admission to intensive care unit (ICU) for critical support and management [6, 7].
Currently, 215 nations are affected by COVID-19 pandemic [8]. In India, there are about 28 lakh cases with 54,000 deaths till 20th August 2020 [8]. There are no therapeutic options approved for COVID-19 and many antivirals, IL-6 inhibitors are being clinically evaluated for the therapeutic efficacy [9].
The pathogenesis of COVID-19 is unclear, but in severe cases there is laboratory evidence of systemic inflammation similar to cytokine release syndrome (CRS) with increase of proinflammatory cytokines like ferritin, IL-6, D dimer, etc. [10-12]. Hence, blocking of IL-6 can reduce the cytokine storm [10]. Tocilizumab (TCZ), a humanized monoclonal antibody is an IL-6 inhibitor is being used to treat rheumatoid arthritis, CRS induced by chimeric antigen receptor T-cell (CAR-T) therapy and other autoinflammatory conditions [13, 14]. In an initial single arm Chinese trial, administration of TCZ showed radiological improvement, reduction of body temperature and oxygen supplementation in 21 COVID-19 positive patients [15].
The aim of our study was to evaluate data and report the first experience with TCZ in 20 patients with severe SARS-CoV-2 infection in a Tertiary care Hospital in Western India.
Patients and methods
This was a retrospective observational study with inclusion of all SARS-CoV-2 RTPCR positive patients who received tocilizumab. Statistical analysis was done using Statistical Package for the Social Sciences (SPSS), version 25.0. Descriptive analysis was presented as Mean ± standard deviation for continuous variables and percentage for categorical variables. Statistical tests of significance (paired t- test) was performed for analysis of survived patients excluding the expired patients. P-value of <0.05 was considered statistically significant.
Results
The 20 patients who were in ICU at the time of receipt of TCZ were evaluated for baseline characteristics and outcomes (table 1). The majority were males (75%, 15) and the mean age was 47.20±9.68 years. Comorbid conditions included diabetes mellitus (50%,10), hypertension (35%, 7), chronic kidney disease (5%, 1), Obesity (5%, 1) and hypothyroidism (5%,1). Of total patients, 6 (30%) were having single co-morbid conditions and 7 (35%) were having two comorbid conditions. Respiratory symptoms (80%, 16), myalgia (45%, 9), fever (35%, 7), cough (20%, 4) and others like headache, diarrhea were the major complaints. All patients have bilateral pulmonary infiltrates on chest radiographic images pre TCZ and improvement was seen post TCZ (Figure 1).
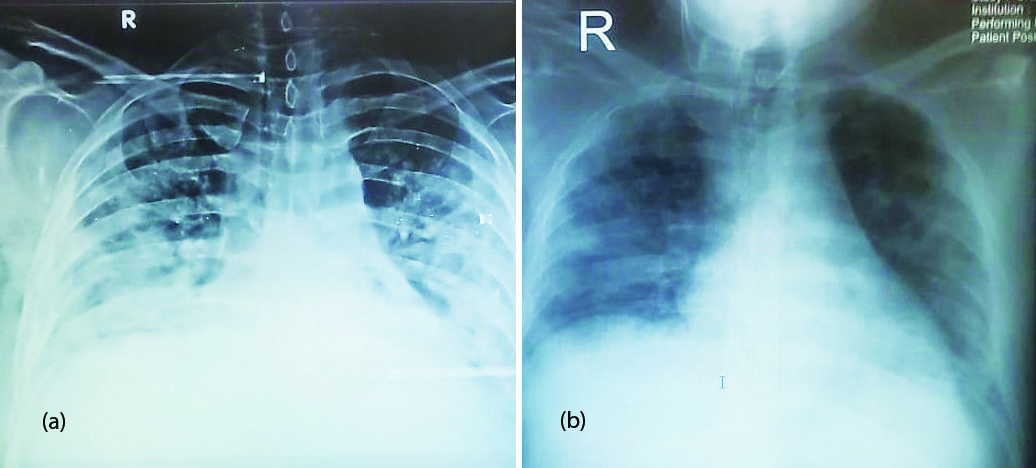
Figure 1: Chest x-ray PA view of a patient showing improvement after tocilizumab administration.
The procalcitonin was raised in one patient but the culture sensitivity was negative. There were 5 patients (25%) on NRBM (Non Re-Breather Mask), 12(60%) on BIPAP and 3(15%) on IPPV pre TCZ. Mortality reduced by 65%(13) after TCZ administration. The Mean duration of hospital stay for all patients was 13.90±8.44 days.
All patients were initially treated with azithromycin, hydroxychloroquine and methyl prednisolone. The patients who were progressing in symptoms and in radiographic images were then administered TCZ at a dose of 6-8mg/kg. The mean IL-6 pre TCZ was 981.70±650.98. All patients were initially treated with azithromycin, hydroxychloroquine and methyl prednisolone, and were progressing in symptoms and in radiographic images. Patients were then administered TCZ at a dose of 6-8mg/kg.
All patients who survived were evaluated for laboratory parameters pre and post TCZ (n=13). There was statistically significant decrease of C- reactive protein (p<0.00052) and ferritin (p<0.023) post TCZ administration (Table 2).
Table 1: Patient baseline characteristics (pre TCZ).
|
Patient characteristics (total number of patients N = 20)
|
|
Age (Mean±SD)
|
Mean 47.20±9.68 years
|
|
Gender (%, frequency)
|
Female
|
25% (5)
|
|
Male
|
75% (15)
|
|
Comorbid conditions (%, frequency)
|
Diabetes mellitus
|
50% (10)
|
|
Hypertension
|
35% (7)
|
|
Chronic kidney disease
|
5% (1)
|
|
Obese (BMI >30)
|
5% (1)
|
|
Hypothyroid
|
5% (1)
|
|
1 comorbid condition
|
30% (6)
|
|
2 comorbid conditions
|
35% (7)
|
|
No comorbid conditions
|
35% (7)
|
|
Clinical symptoms (%, frequency)
|
Fever
|
35% (7)
|
|
Myalgia
|
45% (9)
|
|
Respiratory symptoms
|
80% (16)
|
|
Cough
|
20% (4)
|
|
Others (headache, diarrhea)
|
15% (3)
|
|
Complete blood count
(Mean±SD)
|
Hemoglobin (gm/dl)
|
12.34±2.00
|
|
Total leukocyte count (x109 /L)
|
9.626±4.176
|
|
Platelet count (x109 /L)
|
256±107
|
|
Renal function test (Mean±SD)
|
Serum creatinine(mg/dl)
|
1.06±1.14
|
|
Liver function test
(%, frequency)
|
Elevated liver enzymes
|
50% (10)
|
|
IL-6 (pg/ml) (Mean±SD)
|
981.70±650.98
|
|
Procalcitonin
(%, frequency)
|
Negative
|
95% (19)
|
|
Positive
|
5% (1)
|
|
Oxygen therapy
(%, frequency)
|
Non Re -Breather Mask
|
25% (5)
|
|
BIPAP
|
60% (12)
|
|
IPPV
|
15% (3)
|
|
Treatment outcomes
(%, frequency)
|
Survived
|
65% (13)
|
|
Expired
|
35% (7)
|
|
Duration of hospital stay
(Mean±SD)
|
For all patients
|
13.90±8.44 days
|
|
For survived patients
|
17.69±7.55 days
|
|
For expired patients
|
6.86±4.74 days
|
Table 2: Various parameters before and after tocilizumab in survived patients (n = 13).
|
Laboratory parameters
|
Pre- TCZ
Mean±SD
|
Post-TCZ
Mean±SD
|
P-Value
|
|
Hemoglobin (gm/dl)
|
12.13±2.01
|
12.60±1.86
|
0.308
|
|
Total leukocyte count (x109 /L)
|
9.588±4.292
|
10.103±4.867
|
0.801
|
|
Absolute neutrophil count (x109 /L)
|
8.171±4.251
|
9.433±5.688
|
0.571
|
|
Absolute lymphocyte count (x109 /L)
|
1.008±0.625
|
0.981±0.695
|
0.893
|
|
Neutrophil lymphocyte ratio (NLR)
|
10.31±6.24
|
16.71±16.24
|
0.190
|
|
Platelet count (x109 /L)
|
269±107
|
370±113
|
0.016
|
|
C- Reactive protein (mg/L)
|
79.69±55.57
|
7.93±8.11
|
0.000502
|
|
LDH (U/L)
|
549.83±229.23
|
433.08±137.25
|
0.147
|
|
D-dimer (ng/ml)
|
1862±4242.65
|
1219.54±1734.34
|
0.633
|
|
Ferritin (ng/ml)
|
660±564.43
|
387.70±318.52
|
0.023
|
Discussion
In our study we analyzed the patient baseline characteristics and outcomes in 20 patients. We also analyzed the laboratory parameters pre and post TCZ in the survived patients (n=13). The mortality decreased to 65% (13) after TCZ in our study with a Mean duration of hospital stay 13.90±8.44 days.
Several studies have reported the benefit of tocilizumab in severe COVID-19 patients [15-17]. Xu et al., in their study described 21 patients with severe to critical COVID-19 disease. Their study included 2 patients on mechanical ventilation and seven on oxygen supplementation. All patients were discharged alive after tocilizumab administration with no significant adverse effects [15].
Klopfenstein et al., in their case control study of 20 patients showed lower composite endpoint of mortality and ICU admission in the tocilizumab group. The difference in mortality in the standard therapy and tocilizumab group was not statistically significant [18].
In another study, Toniati et al., described 100 consecutive patients treated with multiple doses of tocilizumab for severe to critical COVID-19 disease. There was 18% mortality in patients receiving non invasive ventilation and 24% mortality in intubated patients. Only 15% patients are discharged [19]. Most of the studies had some pitfalls with small study sample and lack of control group.
Few studies have reported that tocilizumab is ineffective in treatment of severe COVID-19 pneumonia. Interim analysis of phase 3 randomized controls COVACTA trial of tocilizumab vs placebo, did not show improvement in mortality but decreased hospital stay by a week [20]. A randomized clinical trial done by Rosas et al., on 452 severe COVID-19 pneumonia patients showed that tocilizumab did not improve the clinical status or mortality [21].
Conclusion
Our study showed that there was 65% (13) reduction in mortality in patients with severe COVID-19 disease treated with TCZ. The mean duration of hospital stay in our study for all patients was 13.90±8.44 days. Many randomized trials are required for confirming the efficacy, effect on various laboratory parameters and also to know the adverse effects.
Limitations
The limitations of our study are that it is a retrospective observational single arm study. The size of the sample in the present study was small which is a limitation and the results cannot be generalized. The study excluded expired patients to compare the various parameters and inflammatory cytokines pre and post TCZ. This could lead to selection bias.
Conflicts of interest
Authors declare no conflicts of interest.
References
[1] Centers for Disease Control and Prevention, August 10, 2018. Past pandemics. Available from: https://www.cdc.gov/flu/pandemic-resources/basics/past-pandemics.html
[2] Zhu N, Zhang D, Wang W, et al. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med. 2020; 382(8):727–733.
[3] Gorbalenya AE, Baker SC, Baric RS, Groot RJD, Drosten C, et al. The species severe acute respiratory syndrome‐related coronavirus: classifying 2019‐nCoV and naming it SARS‐CoV‐2. Nat Microbiol. 2020; 5(4):536–544.
[4] World Health Organization. Report of the WHO‐China Joint Mission on Coronavirus Disease 2019 (COVID‐19). Geneva, Switzerland: World Health Organization; 2020. Available from: https://www.who.int/docs/default-source/coronaviruse/who-china-joint-mission-on-covid-19-final-report.pdf
[5] World Health Organization. Coronavirus disease 2019 (COVID‐19) Situation Report 51 (11 March 2020). Geneva, Switzerland: World Health Organization; 2020. Available from: https://www.who.int/docs/defaultcoronavirusdisease2019.source/coronaviruse/situation-reports/20200311-sitrep-51-covid-19.pdf?sfvrsn=1ba62e57_10
[6] Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID‐19) outbreak in China: summary of a report of 72 314 cases from the Chinese center for disease control and prevention. JAMA. 2020; 323(13):1239–1242.
[7] Huang C, Wang Y, Li X, Ren L, Zhao J, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020; 395(10223):497‐506.
[8] Worldometer, Coronavirus- WORLD/COUNTRIES/INDIA, August 20, 2020. Available from: https://www.worldometers.info/coronavirus/country/india/
[9] Centers for Communicable Disease Control and Prevention. Information for clinicians on therapeutic options for COVID‐19 patients: Centers for Communicable Disease Control and Prevention, 2020. Available from: https://www.cdc.gov/coronavirus/2019-ncov/hcp/therapeutic-options.html
[10] Zhang CM, Wu Z, Li JW, Zhao H, Wang GQ. The cytokine release syndrome (CRS) of severe COVID-19 and Interleukin-6 receptor (IL-6R) antagonist tocilizumab may be the key to reduce the mortality. Int J Antimicrob Agents. 2020; 55(5):105954.
[11] Park WY, Goodman RB, Steinberg KP, Ruzinski JT, Radella F, et al. Cytokine balance in the lungs of patients with acute respiratory distress syndrome. Am J Respir Crit Care Med. 2001, 164(10 Pt 1):1896–1903.
[12] Wang Z, Han W. Biomarkers of cytokine release syndrome and neurotoxicity related to CAR-T cell therapy. Biomarker Res. 2018; 6:29387417.
[13] Scott LJ. Tocilizumab: A review in rheumatoid arthritis. Drugs. 2017; 77:1865–1879.
[14] Kotch C, Barrett D, Teachey DT. Tocilizumab for the treatment of chimeric antigen receptor T cell-induced cytokine release syndrome. Expert Rev Clin Immunol. 2019; 15(8):813–822.
[15] Xu X, Han M, Li T, Sun W, Wang D, et al. Elective treatment of severe COVID-19 patients with tocilizumab. Proc Natl Acad Sci USA. 2020; 45:32350134.
[16] Luo P, Liu Y, Qiu L, Liu X, Liu D, et al. Tocilizumab treatment in COVID-19: A single center experience. J Med Virol. 2020; 92(7):814–818.
[17] Fu B, Xu X, Wei H. Why tocilizumab could be an effective treatment for severe COVID-19? J Transl Med. 2020; 18(1):164.
[18] Klopfenstein T, Zayet S, Lohse A, Balblanc JC, Badie J, et al. Tocilizumab therapy reduced intensive care unit admissions and/or mortality in COVID-19 patients. Med Mal Infect. 2020; 50(5):397–400.
[19] Toniati P, Piva S, Cattalini M, Garrafa E, Regola F, et al. Tocilizumab for the treatment of severe COVID-19 pneumonia with hyperinflammatory syndrome and acute respiratory failure: A single center study of 100 patients in Brescia, Italy. Autoimmunity reviews. 2020; 19(7):102568.
[20] Furlow B. COVACTA trial raises questions about tocilizumab's benefit in COVID-19. Lancet Rheumat. 2020; 2(10):e592.
[21] Rosas I, Bräu N, Waters M, Go RC, Hunter BD, et al. Tocilizumab in hospitalized patients with COVID-19 pneumonia. medRxiv. 2020 Jan 1. Available from: https://www.medrxiv.org/content/10.1101/2020.08.27.20183442v2.full.pdf