Full Text
Introduction
Acute megakaryoblastic leukemia (AMKL) is a rare form of leukemia, 3%– 5% of all de novo Acute myeloid leukemia (AML) cases [1]. In adults, AMKL is more uncommon, with only 0.6% of all AML cases [2, 3]. AMKL is more prevalent in children, constituting 4%–15% of all AML cases [2, 4]. It is a diagnostic challenge, due to myelofibrosis and very few circulating blasts [3].
WHO 5th edition hematolymphoid neoplasms proposed diagnostic essential criteria for AMKL as ≥20% blasts with megakaryocytic differentiation in the PS, and/or BMA, and any one positive megakaryocytic marker (CD41a, or CD61, or CD42b) on IPT. To diagnose AMKL, myeloproliferative neoplasm and other defined AML types must be excluded [1].
AMKL has 3 clinical groups based on molecular alterations. Children with DS AMKL show GATA1 mutations. Children with Non-DS AMKL are associated mostly with chromosomal translocations like RBM15::MRTFA, NUP98::KDM5A, CBFA2T3::GLIS2 and KMT2A rearrangements. Adults with AMKL show mutations in TP53, RB1, ASXL1 and DNMT3A [1]. Multivariate analysis observed AML with megakaryoblastic morphology as an independent adverse prognostic factor for OS [5, 6]. AMKL associated with DS has an excellent prognosis with high rates of remission. In contrast, Non-DS AMKL is more heterogeneous, and have a poor prognosis [1, 7]. AMKL in adults have an inferior prognosis than children [8, 9].
Materials and methods
A 6-year retrospective analytical study from January 2018 to May 2024 is conducted in the Department of Oncopathology in The Gujarat Cancer and Research Institute. Study is approved by the institutional research committee (IRC), as per the 1964 helsinki declaration and its subsequent amendments. Informed consent was obtained from all patients at the time of admission for the use of medical record data for scientific studies.
AMKL diagnosis was based on the bone marrow blast morphology and the megakaryoblastic lineage was confirmed by CD41a positivity on flow cytometry. Clinical information, reports of CBC parameters, wright-stained PS, BMA slides and reports, IPT reports, karyotyping reports, and survival data are collected from institutional LIS, MRD case files and slide archives at our institute.
Inclusion criteria: All de novo AMKL cases in children and adults that are IPT-proven diagnosed within the institute.
Exclusion criteria: All AML cases that are negative for megakaryoblastic IPT marker CD41.
Sample collection and preparation: The BMA and peripheral blood samples were collected in an EDTA vacutainer, and processed within 24 hours for morphology and IPT. Single-bevel pediatric BMB Needle (Ayka Medical Inc., Sonipat, Haryana), an adult BMB Jamshidi needle (BD India, Gurgaon, Haryana) were used for all experiments. Wright-stained PS, BMA smears, and hematoxylin and eosin (H&E) stained BMB sections are reviewed for all cases.
Immunophenotyping: For all 15 cases, PS or BMA samples were run in a multicolor flow cytometer (FACS Canto II, 6-color, 2-Laser, BD biosciences, New Jersey, USA), with primary monoclonal antibody panel: CD45 (v500), CD34 (Per CP cy5.5), CD19 (PE Cy 7), cCD79a (PE), sCD3(1), cCD3 (PE Cy7), CD7 (APC), c MPO (FITC), and secondary antibody panel: CD41a(PE), CD13 (PE), CD33 (PE Cy7), CD117 (APC), HLA-DR (APC-H7), CD14 (APC-H7), CD15 (FITC). Antibody, fluorochrome dye, clone, catalog number, and volume used for each sample are tabulated in table 1. Sample preparation flow chart is depicted in figure 1.
Table 1: Details of the primary and secondary antibody panel used in the study for diagnosing AMKL. PBS=Phosphate-Buffered Solution.
|
Primary antibody panel (Lineage-specific markers)
|
|
CD Marker
|
CD 45
|
CD19
|
cCD79a
|
CD34
|
cCD3
|
sCD3
|
c MPO
|
CD7
|
|
Fluorescent dye used
|
V500
|
PE Cy 7
|
PE
|
Per cp cy5.5
|
V450
|
APCH7
|
FITC
|
APC
|
|
Volume used for each sample
|
5 µl
|
5 µl
|
10 µl
|
10 µl
|
2 µl
|
5 µl
|
10 µl
|
5 µl
|
|
Clone
|
HI30
|
SJ25C1
|
HM47
|
8G12
|
UCHL1
|
SK7
|
5B8
|
MT701
|
|
Catalog number
|
560779
|
341103
|
333152
|
347213
|
560365
|
641415
|
340580
|
653312
|
|
Secondary antibody panel (Myeloid and megakaryocytic specific markers)
|
|
CD marker
|
cCD41a
|
CD13
|
CD33
|
CD117
|
HLA DR
|
CD14
|
CD15
|
|
Fluorescent dye used
|
PE Cy 7
|
PE
|
APC
|
PE cy 7
|
V450
|
APC
|
FITC
|
|
Volume used for each sample
|
5 µl
|
10 µl
|
5 µl
|
5 µl
|
5 µl
|
5 µl
|
10 µl
|
|
Clone
|
HIP8
|
L138
|
P67.6
|
104D2
|
G46-6
|
MoP9
|
MMA
|
|
Catalog number
|
561424
|
347406
|
345800
|
339206
|
561359
|
345787
|
340703
|
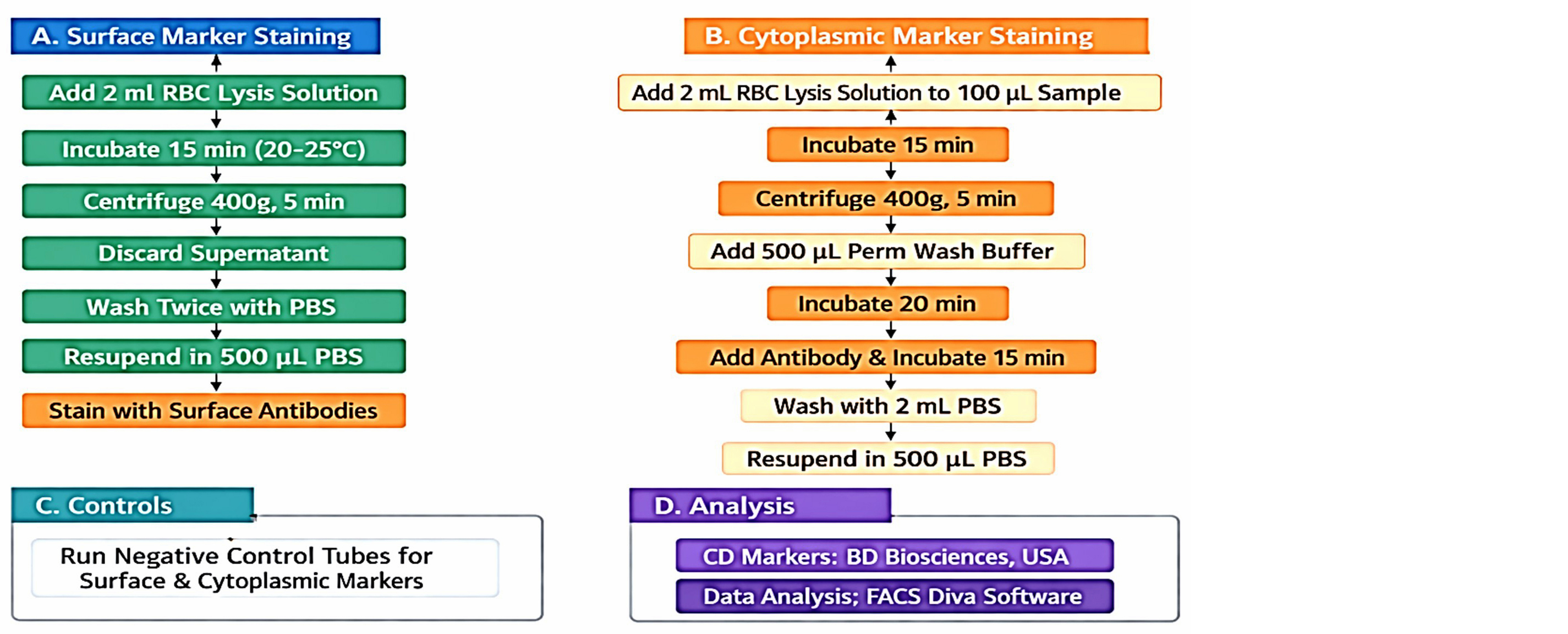
Figure 1: Flow cytometry sample preparation flow chart for Surface CD markers and cytoplasmic CD markers.
Flow cytometric data analysis: A minimum of 1,00,000 total cells were acquired. A side scatter (SSC) versus CD45 (v500) dot plot was used for gating blasts. According to flow cytometry principles, a cut-off value of 20% for surface and cytoplasmic markers was considered positive (BD Biosciences, New Jersey, USA).
Karyotyping was performed at our institute on bone marrow samples collected in heparin vacutainers, and chromosome metaphases were analyzed using IKAROS software to detect down syndrome.
Based on DS, AMKL cases are classified as DS-AMKL or Non-DS AMKL. All AMKL cases were analyzed for age, gender, complete blood count (CBC) parameters, PS and BMA blast percentage, blast count in PS follow-up (PS-FP), BMA follow-up (BMA-FP), down vs. non-down karyotype, CD marker expression with varied intensity in DS-AMKL vs. non-DS-AMKL, remission status, and RFS. Median values are taken as cut-off values to evaluate the differences in Hb, WBC, and Platelet levels between DS-AMKL and non-DS AMKL.
Chemotherapy details
Total 9 patients with AMKL received chemotherapy. 6 patients received chemotherapy with cytarabine and 2 patients with DS AMKL received cytarabine and daunorubicin therapy. One child with non-DS AMKL received high dose cytarabine. No one received bone marrow transplant (BMT), as most of the children succumbed to early death due to non-availability of compatible donor and adults did not receive BMT due to comorbid conditions.
Statistical analysis
The statistical package for the social sciences (SPSS) by IBM, united states (SAGE IBM SPSS), version 24.0 is used for statistical analyses. The kaplan–meier survival analysis was used to obtain the survival curves which were compared by the log-rank test and cox regression model to calculate the hazard ratios. P value <0.05 is statistically significant. Survival follow up data for 6 patients is obtained from hospital records and 7 patients from telephonic conversation with patient’s family members. The median period of follow-up is 28 months, but only one year OS data is mentioned for the uniformity of analysis, as after one year 10 out of 12 children died.
Results
AMKL is seen mostly in children <18 years (12/15) with a median age of 3 years. Only 3 patients are adults (>18 years). In 6 years, a total of 2215 AML cases, with 1791 in adults and 424 in children were diagnosed. Out of which 12 cases in children were AMKL (i.e., 2.8%) and 3 cases in adults (i.e., 0.67%). DS AMKL is observed in a total of 7/12 children, with two of them being 7 years of age, and the remaining infants. Non-DS AMKL is seen in 3 adults >50 years and 5 children, ranging in age from 6 months to 2 years. The incidence of AMKL is higher in children than in adults, with a ratio of 4:1, and M: F ratio of 1.5:1.
Complete blood count parameters: All AMKL cases showed anemia, with hemoglobin (Hb) levels ranging from 3.7 to 10.9 g/dL and a median Hb level of 7.7 g/dL; leucopenia to leukocytosis with WBC counts ranging from 870 to 1 lakh cells/cu mm and median WBC counts of 13,300 cells/cu mm.
AMKL showed either thrombocytopenia or normal platelet counts, with a median platelet count of 66,000 cells/μL. Median PS blasts were 35%, and median BMA blasts were 60%. DS AMKL had lower platelet counts, with higher Hb, WBC counts, and blast percentages in PS and BMA than Non-DS-AMKL, shown in figure 2.
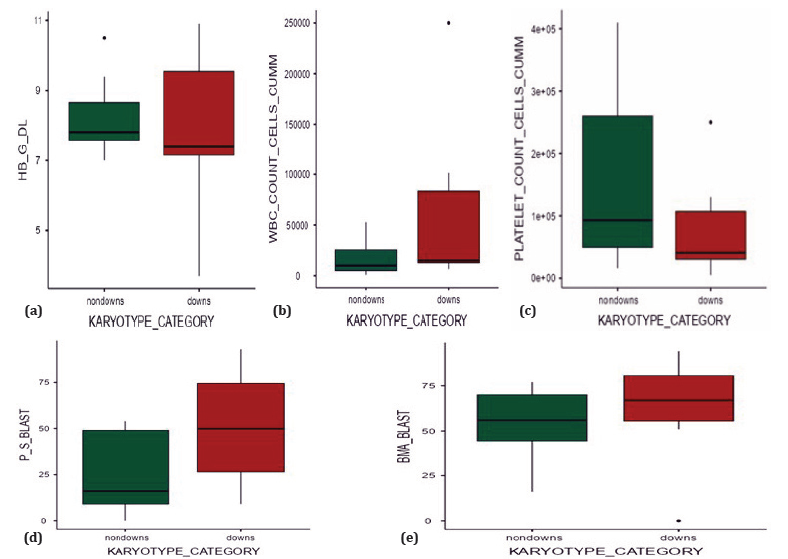
Figure 2: Graphs with differences in CBC parameters and blast percentage in DS AMKL (Brick red) and Non-DS AMKL (Dark green). Non-DS AMKL had lower Hb levels (a), lower WBC counts (b), and higher platelet counts (c) compared to DS AMKL. The percentage of PS blasts (d) and BMA blasts (e) is higher in DS-AMKL than in non-DS-AMKL.
Morphology: Wright-stained PS and BMA smears showed medium to large blasts with round, slightly irregular nuclei, fine chromatin, and one to three nucleoli, basophilic agranular cytoplasm with distinct cytoplasmic blebs or pseudopods (Figure 3a). On morphology blasts with basophilic cytoplasm with vacuoles are also seen in AML with monocytic differentiation and acute erythroblastic leukemia. Cytoplasmic blebs are seen in myeloblasts in AML M0 or M1 as well as with hydroxymethyl prednisolone therapy. Clustering of blasts, binucleation, cytoplasmic blebs with granules and platelet budding on surface of the blasts are morphological clues that helped in exclusion of other AML blasts with similar features and arriving at AMKL diagnosis. For definite diagnosis of AMKL, blasts should express megakaryoblast markers either CD41 or CD61 and all 15 cases in our study were IPT proven expressing CD41. BMB was done in five cases, showing diffuse infiltration of marrow spaces by poorly differentiated blasts (Figure 3b). A reticulin stain on the BMB showed grade 1 myelofibrosis.
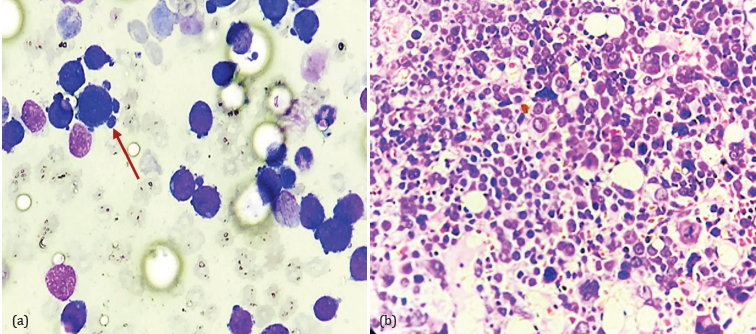
Figure 3: Morphology of AMKL blasts in BMA and BMB: (a) BMA (Wright stain, 400x) with large blasts showing cytoplasmic blebs and basophilic cytoplasm with fine cytoplasmic vacuoles (red arrow), (b) BMB (H&E stain 400X) showing diffuse infiltration of marrow spaces by poorly differentiated pleomorphic blasts with bi, tri, and multinucleation and atypical mitotic figures.
Karyotyping: Conventional karyotyping showed trisomy 21 (47 XX/XY) is seen exclusively in children (7/15) cases, while the remaining eight non-DS AMKL cases are seen in both children (5/8) and adults (3/8). Among 8 Non-DS AMKL cases, 4 showed normal karyotypes, 2 had complex karyotypes, and among other 2 cases one older adult had an isochromosome 17, and trisomy 8 in a 6-month-old infant.
Immunophenotypic analysis: In all cases, CD45-gated blasts showed positivity for the specific megakaryoblastic marker CD41a and expressed stem cell markers, including CD117, CD34, HLA-DR, and myeloid markers such as CD33 and CD13. Additionally, they showed aberrant T cell marker expression, specifically CD7. Myeloid lineage marker cMPO, B cell lineage markers (CD19, Ccd79a), and T cell lineage markers (cCD3, sCD3) were negative, as shown in figure 4.
Expression and intensity of CD markers in DS AMKL and Non-DS AMKL were varied. Differences in expression of CD markers in DS AMKL vs Non-DS AMKL is depicted in table 2.
Table 2: Differences in expression of CD markers in DS AMKL vs Non-DS AMKL.
|
Immunophenotypic CD Marker Intensity
|
Karyotype
Category
|
Counts
|
% of Total cells Positive
|
|
CD_41A_POSITIVE_BLASTS
|
DIM
|
Non-DS
|
0
|
0.00%
|
|
DS
|
2
|
13.30%
|
|
MODERATE
|
Non-DS
|
4
|
26.70%
|
|
DS
|
4
|
26.70%
|
|
MODERATE TO BRIGHT
|
Non-DS
|
3
|
20.00%
|
|
DS
|
1
|
6.70%
|
|
BRIGHT
|
Non-DS
|
1
|
6.70%
|
|
DS
|
0
|
0.00%
|
|
CD34_POSSIVE_BLASTS
|
DIM
|
Non-DS
|
3
|
20.00%
|
|
DS
|
1
|
6.70%
|
|
MODERATE
|
Non-DS
|
4
|
26.70%
|
|
DS
|
6
|
40.00%
|
|
BRIGHT
|
Non-DS
|
1
|
6.70%
|
|
DS
|
0
|
0.00%
|
|
CD117_POSITIVE_BLASTS
|
DIM
|
Non-DS
|
2
|
13.30%
|
|
DS
|
0
|
0.00%
|
|
MODERATE
|
Non-DS
|
6
|
40.00%
|
|
DS
|
7
|
46.70%
|
|
CD7_POSITIVE_BLASTS
|
DIM
|
Non-DS
|
4
|
26.70%
|
|
DS
|
1
|
6.70%
|
|
MODERATE
|
Non-DS
|
4
|
26.70%
|
|
DS
|
3
|
20.00%
|
|
MODERATE TO BRIGHT
|
Non-DS
|
0
|
0.00%
|
|
DS
|
3
|
20.00%
|
|
CD_33_POSITIVE_BLASTS
|
DIM
|
Non-DS
|
6
|
40.00%
|
|
DS
|
2
|
13.30%
|
|
MODERATE
|
Non-DS
|
2
|
13.30%
|
|
DS
|
5
|
33.30%
|
|
CD_13_POSITIVE_BLASTS
|
DIM
|
Non-DS
|
4
|
26.70%
|
|
DS
|
4
|
26.70%
|
|
MODERATE
|
Non-DS
|
4
|
26.70%
|
|
DS
|
3
|
20.00%
|
|
HLA_DR_POSITIVE_BLASTS
|
DIM
|
Non-DS
|
4
|
26.70%
|
|
DS
|
6
|
40.00%
|
|
MODERATE
|
Non-DS
|
3
|
20.00%
|
|
DS
|
1
|
6.70%
|
|
MODERATE TO BRIGHT
|
Non-DS
|
1
|
6.70%
|
|
DS
|
0
|
0.00%
|
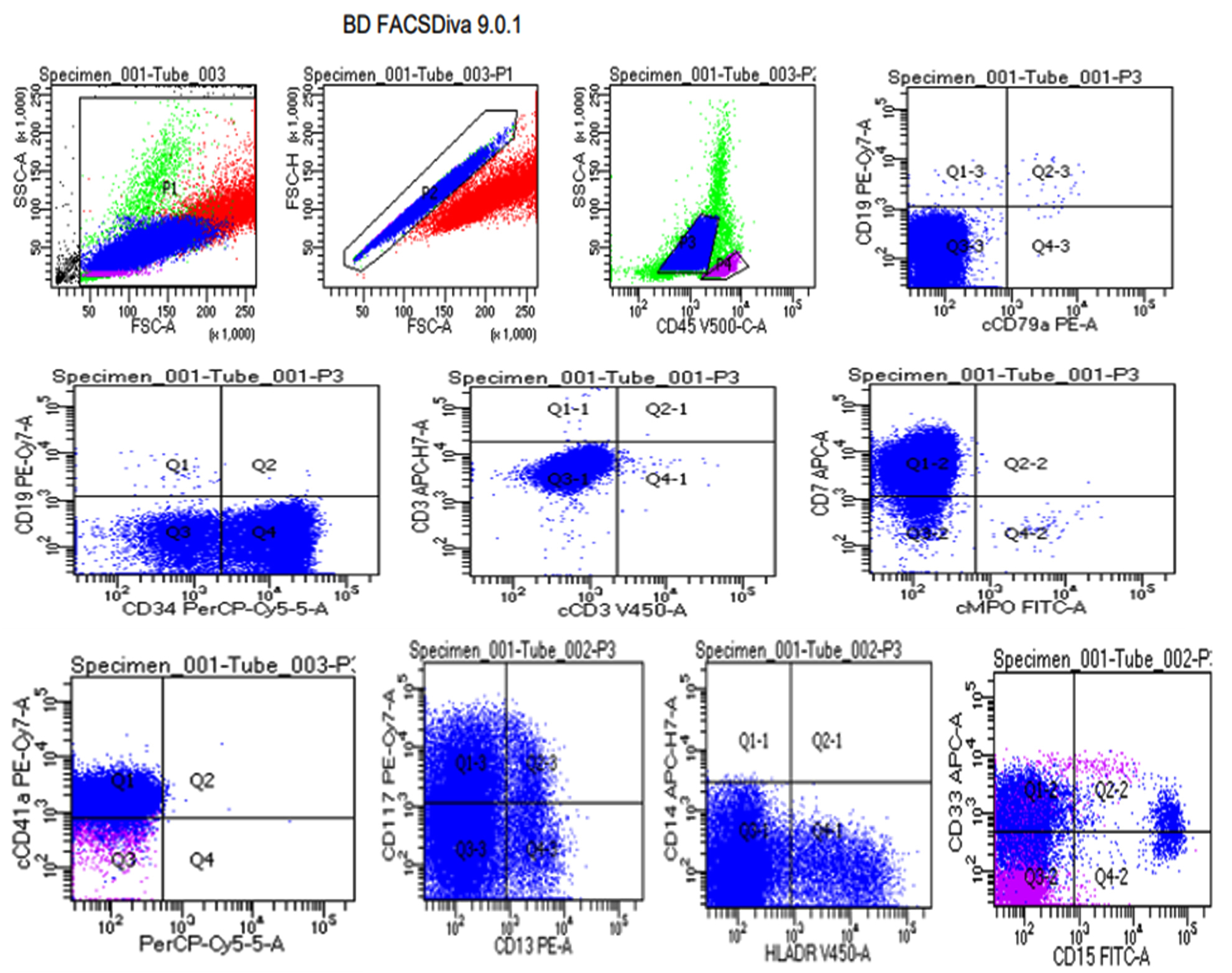
Figure 4: Flow cytometric analysis done by BD FACS Diva showing CD45-gated blasts expressing CD41a positive along with CD117, CD7, CD34, and CD33 co-expression diagnosed as AMKL. Blasts are negative for myeloid, B, and T lineage markers.
Overall survival
Kaplan–Meier survival analysis is presented in table 3, which compares demographics, CBC parameters and IPT markers, karyotyping, and remission status for OS.
Table 3: Kaplan-Meier analysis for Overall Survival.
|
Variable
|
Value
|
OS status
|
|
No. Died (%)
|
No. Alive (%)
|
p value
(Log rank)
|
|
Age
|
<18
|
10(66.66%)
|
2(13.33%)
|
0.448
|
|
>18
|
3(20%)
|
0(0%)
|
|
Sex
|
FEMALE
|
6(40%)
|
0(0%)
|
0.215
|
|
MALE
|
7(46.66%)
|
2(13.33%)
|
|
HB
|
<7.7
|
8(53.33%)
|
0(0%)
|
0.104
|
|
>7.7
|
5(33.33%)
|
2(13.33%)
|
|
WBC
|
<13300
|
6(40%)
|
1(6.66%)
|
0.919
|
|
>13300
|
7(46.66%)
|
1(6.66%)
|
|
Platelet
|
<66000
|
7(46.66%)
|
1(6.66%)
|
0.919
|
|
>66000
|
6(40%)
|
1(6.66%)
|
|
P S
|
<35
|
7(46.66%)
|
0(0%)
|
0.155
|
|
>35
|
6(40%)
|
2(13.33%)
|
|
BMA
|
<60
|
5(33.33%)
|
2(13.33%)
|
0.104
|
|
>60
|
8(53.33%)
|
0(0%)
|
|
Karyotype category
|
DOWNS
|
6(40%)
|
1(6.66%)
|
0.919
|
|
NON- DOWNS
|
7(46.66%)
|
1(6.66%)
|
|
PS FP
|
YES
|
3(20%)
|
1(6.66%)
|
0.423
|
|
NO
|
10(66.66%)
|
1(6.66%)
|
|
BM FP
|
YES
|
2(13.33%)
|
1(6.66%)
|
0.255
|
|
NO
|
11(73.33%)
|
1(6.66%)
|
|
Remission
|
YES
|
3(20%)
|
2(13.33%)
|
0.032
|
|
NO
|
10(66.66%)
|
0(0%)
|
|
Relapse status
|
YES
|
1(6.66%)
|
1(6.66%)
|
0.101
|
|
NO
|
12(80%)
|
1(6.66%)
|
|
Survival status
|
DIED
|
13(86.66%)
|
0(0%)
|
|
|
ALIVE
|
0(0%)
|
2(13.33%)
|
|
Chemo
|
Yes
|
7(46.66%)
|
2(13.33%)
|
0.215
|
|
No
|
6(40%)
|
0(0%)
|
Anemia with Hb levels ≤ 7.7 g/dL is seen in 46% of AMKL cases and is associated with a decreased median OS compared to higher Hb levels. Despite having low platelet counts, higher WBC counts, and higher blast percentages in PS and BMA, DS AMKL children had better OS than Non-DS AMKL. OS in children is 13%, and in adults it is 0%.
The median period of follow-up is 28 months. In one year, survival analysis, DS AMKL children at the end of 2 months had 42.90% survival but from fourth month onwards they had 28.6% survival rate, while Non-DS AMKL children showed uniform inferior survival rate of 20% in the entire year.
The median OS in children is 32 days. Most of the children in our study had an earlier death, even before the start of chemotherapy (7/12) or a few days to months of chemotherapy (3/12). The cause of death is acute cardiorespiratory arrest or AMKL itself.
9 AMKL cases received chemotherapy. Post-chemotherapy follow-up showed remission with complete hematological recovery in 5 cases, with the absence of blasts in PS and BMA, among which 2 cases showed relapse. Cause of death in relapse cases is AMKL itself. Cause of death in 2 non-relapse cases in DS AMKL children was high AMKL blasts causing Cardiac arrest. One child with Non-DS AMKL had a relapse but survived due to low BMA blast percentage and due to intensified chemotherapy. Adults had a median OS of 3 months, as one patient survived for a year with chemotherapy went into remission without any relapse, however he died due to cytarabine-induced cardiotoxicity.
AMKL cases that achieved remission (5/15) in their disease course had better OS compared to those without any remission, which shows a statistically significant P-value=0.032.
AMKL cases that received chemotherapy in their disease course had better OS (13.33%) compared to those without any chemotherapy (0%).
OS probabilities in children for time in months in DS AMKL and Non-DS AMKL are depicted in figure 5.
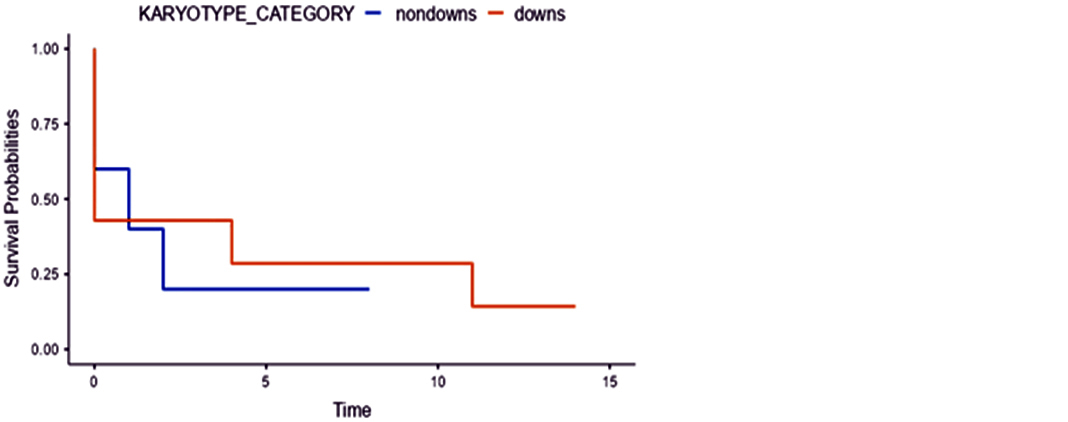
Figure 5: Overall survival probabilities in DS AMKL and Non-DS AMKL children with 2-month time interval. The graph shows a better OS in DS AMKL than Non-DS AMKL. Note: Survival Probability of 1.00 on the Y-axis = 100%.
Table 4 depicts details of all 15 patients and includes age at diagnosis, sex, karyotype, date of diagnosis, last known status, details of therapy received, remission status, relapse status, date and cause of death.
Table 4: Demographic, karyotypic, treatment, response and cause of death details of patients with AMKL.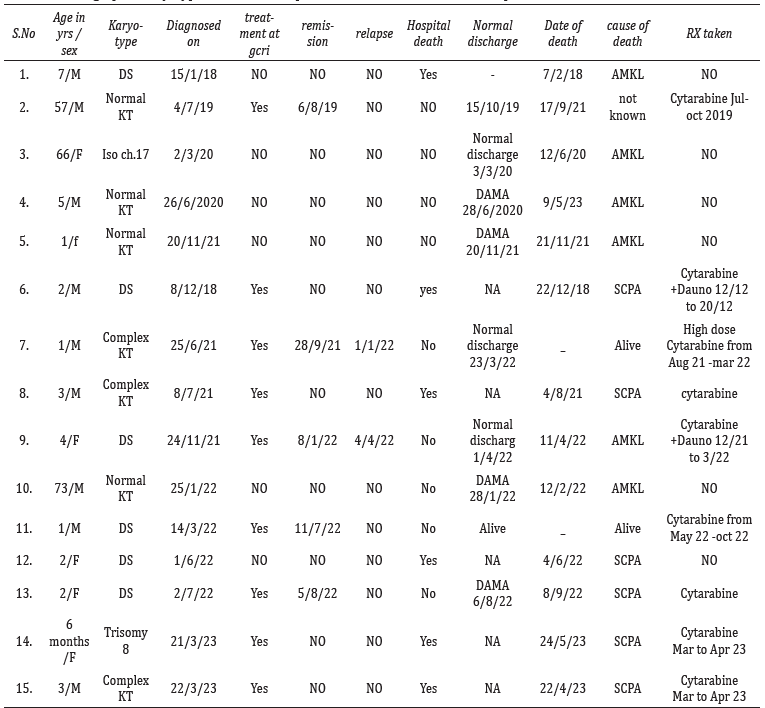
Abbreviations: SCPA= Sudden cardio pulmonary arrest, KT=Karyotype, DAMA=Discharge against medical advice.
A Cox proportional Hazard Ratio of 0.91 depicted in table 5.
Table 5: KARYOTYPE_CATEGORY with Cox Proportional Hazard Ratio (HR) in children.
|
Explanatory
|
Levels
|
all
|
HR (Univariable)
|
|
KARYOTYPE
|
Non-DS AMKL
|
5 (41.7)
|
-
|
|
|
DS AMKL
|
7 (58.3)
|
0.91 (0.24-3.44, p=0.893)
|
Discussion
AMKL is more common in children with median age of 3 years than in adults, with a 4:1 ratio, as observed by Brouwer et al. in the euro flow study. 10 AMKL is seen more in males.
CBC parameters differed between DS-AMKL and Non-DS-AMKL. Leukocytosis and thrombocytopenia are more common in DS-AMKL than in Non-DS-AMKL, but this difference did not significantly impact the survival. In contrast, anemia, with Hb <7.7 g/dL, more commonly seen in Non-DS AMKL. AMKL, with a higher BMA blast initially and on follow-up, had decreased OS. Similarly, AMKL with anemia, and AML M7 morphology are observed as independent negative factors for OS by Oki Y et al. at MD anderson cancer center [6].
In our study, AMKL patients with down and non-down karyotypes showed differences in the positivity and intensity of IPT markers, though they are not statistically significant. DS AMKL patients showed dim to moderate CD41a expression compared to moderate to bright CD41a expression in Non-DS AMKL. Dim to moderate expression of CD7 is seen in Non-DS AMKL, while the intensity is brighter in DS AMKL, and aberrant expression of CD7 is seen in all DS AMKL, similar to the findings of Javed et al [11]. The intensity of CD117 expression is dim to moderate in Non-DS AMKL, while DS AMKL shows moderate expression, which is in concordance with the findings of Brouwer et al. in the euro flow study [10]. Wang et al [12] and Koulmane laxminarayana et al [13] have also observed significant variation in CD marker expression between DS AMKL and non-DS AMKL. Observing these differences in IPT marker positivity and their intensity among AMKL patients serves as diagnostic clue for further cytogenetic workup, which impacts the survival outcomes.
The median follow-up period of all patients in our study is 28 months. OS in children is 13%, and in adults it is 0%, indicating poorer outcomes in adult AMKL. One child with DS AMKL is alive and one child with Non-DS AMKL who received high dose chemotherapy are alive. DS AMKL had better 1 year survival rates (28.6-42.9%) compared to Non-DS AMKL (20%). One Non-DS AMKL adult who survived for a year with chemotherapy later died due to chemotherapy-induced toxicity and age-related morbidity, while a child with Non-DS AMKL is alive after receiving intensified chemotherapy, indicating better OS in children than adults. None of the adults survived the 28-month follow-up period, indicating poor prognosis in adults.
In our study, the median OS in AMKL patients is 1 month, which correlates well with the median OS of 6 months by Zhao et al [3], the median OS of 13 months in Phillip A. Haadad et al [14], and a similar median OS in months seen by Pagano et al [4], Oki Y et al [6], Tallman et al [9].
Hazard ratio of 0.91, indicates 9% less risk of death in DS AMKL compared to Non-DS AMKL. Even though the difference in OS is minimal, DS AMKL has a better OS than Non-DS AMKL, as also observed by Hama et al [15].
Coronary and pulmonary vessels thrombosis, with emboli in atria and ventricles, leading to acute cardiorespiratory failure and sudden death is due to excessive production of platelet adhesion molecules by megakaryoblasts. This cause of death is also observed by Odani et al [16].
In our study AMKL cases with complete remission had better OS compared to those without any remission, with a statistically significant P-value=0.032. Similar findings were observed by Phillip A. Haadad et al [14], and Schweitzer J et al [17]. Children with Non-DS AMKL, who received intensified chemotherapy with high dose cytarabine, had better OS, which was also highlighted by Schweitzer J et al [17].
Study strengths: This article is one of the very few articles that focus on the integrated diagnosis of AMKL with CBC parameters, IPT markers, karyotyping, and morphology, along with assessing the overall survival in AMKL patients, comparing survival outcome with and without DS, which also compares the survival benefits in patients with remission and those who received chemotherapy.
Limitations: In our study, we could not check for the mutations and chromosomal translocations in AMKL. NGS and cytogenetics are not available at our institute during the study period and as it is a retrospective study samples are not available for molecular analysis in the present study.
Conclusion
Integrated diagnosis, incorporating morphological clues and CD41a expression, is essential in diagnosing AMKL. Age at diagnosis, differences in CBC parameters, percentage of blasts in BMA, down syndrome versus non-DS Karyotype, and remission status and chemotherapy status play a significant role in assessing overall survival in patients with AMKL. Differential expression of IPT markers in AMKL should prompt appropriate karyotyping and cytogenetic evaluation, as overall survival differs in DS AMKL and Non-DS AMKL.
Acknowledgements
The senior statisticians Asish batham, Prachi Amitkumar Kaloliya and Dr. Anand Shah, Associate professor, community oncology, for the statistical analysis in this study.
Conflicts of interest
Authors declare no conflicts of interest.
References
[1] Loeb K, Marcogliese A, Tembhare P. Acute megakaryocytic leukaemia. In: WHO Classification of Tumours Editorial Board. Haematolymphoid tumours. Lyon (France): International Agency for Research on Cancer; 2024. (WHO classification of tumours series, 5th ed.; vol. 11). Available from https://publications.iarc.who.int/Book-And-Report-Series/Who-Classification-Of-Tumours/Haematolymphoid-Tumours.
[2] Qi H, Mao Y, Cao Q, Sun X, Kuai W, et al. Clinical characteristics and prognosis of 27 patients with childhood acute megakaryoblastic leukemia. Med Sci Monit. 2020; 26:922662.
[3] Zhao G, Wu W, Wang X, Gu J. Clinical diagnosis of adult patients with acute megakaryocytic leukemia. Oncol Lett. 2018; 16:6988–6997.
[4] Pagano L, Pulsoni A, Vignetti M, Mele L, Fianchi L, et al. Acute megakaryoblastic leukemia: experience of GIMEMA trials. Leukemia. 2002; 16:1622–1626.
[5] Masoumi–Dehshiri R, Hashemi A, Neamatzadeh H, Zare–Zardeini H. A case report: acute myeloid leukemia (FAB M7). Iran J Pediatr Hematol Oncol. 2014; 4:188–190.
[6] Oki Y, Kantarjian HM, Zhou X, Cortes J, Faderl S, et al. Adult acute megakaryocytic leukemia: an analysis of 37 patients treated at M.D. Anderson Cancer Center. Blood. 2006; 107:880–884.
[7] Masetti R, Guidi V, Ronchini L, Bertuccio NS, Locatelli F, et al. The changing scenario of non–Down syndrome acute megakaryoblastic leukemia in children. Crit Rev Oncol Hematol. 2019; 138:132–138.
[8] McNulty M, Crispino JD. Acute megakaryocytic leukemia. Cold Spring Harb Perspect Med. 2020; 10:034884.
[9] Tallman MS, Neuberg D, Bennett JM, Francois CJ, Paietta E, et al. Acute megakaryocytic leukemia: the Eastern Cooperative Oncology Group experience. Blood. 2000; 96:2405–2411.
[10] Brouwer N, Matarraz S, Nierkens S, Hofmans M, Novakova M, et al. Immunophenotypic analysis of acute megakaryoblastic leukemia: a EuroFlow study. Cancers (Basel). 2022; 14:1583.
[11] Javed O, Aali A, Shaikh T, Khan H, Mansoor N. Clinical, morphological and immunophenotypic insights into pediatric acute megakaryoblastic leukemia: a multifaceted approach. Asian Pac J Cancer Prev. 2025; 41–47.
[12] Wang L, Peters JM, Fuda F, Li L, Karandikar NJ, et al. Acute megakaryoblastic leukemia associated with trisomy 21 demonstrates a distinct immunophenotype. Cytometry B Clin Cytom. 2015; 88:244–252.
[13] Koulmane Laxminarayana SL, Kohli S, Agrohi J, Belurkar S. Pediatric non–Down syndrome acute megakaryoblastic leukemia with unusual immunophenotype. Cureus. 2023; 15:35965.
[14] Haddad PA, Vutukuri S, Gupta A. Acute megakaryocytic leukemia: clinicopathologic determinants of survival—analysis of a pooled database. Blood. 2024; 144:5938.
[15] Hama A, Yagasaki H, Takahashi Y, Nishio N, Muramatsu H, et al. Acute megakaryoblastic leukaemia (AMKL) in children: a comparison of AMKL with and without Down syndrome. Br J Haematol. 2008; 140:552–561.
[16] Odani K, Abe J, Tsuyuki Y, Yanagita S, Shiogama K, et al. Acute coronary syndrome in acute myeloid leukemia with maturation accompanying megakaryocytic differentiation. Case Rep Pathol. 2020; 2020:8886298.
[17] Schweitzer J, Zimmermann M, Rasche M, et al. Improved outcome of pediatric patients with acute megakaryoblastic leukemia in the AML–BFM 04 trial. Ann Hematol. 2015; 94:1327–1336.