Full Text
Introduction
Chronic lymphoproliferative disorders (CLPD) comprise approximately 90% of all lymphoid malignancies. The annual incidence was about 10 per 100,000 persons worldwide and showed a rising trend in developed countries [1]. However, their occurrence in Asian populations remained relatively infrequent, accounting for approximately 2% of all leukemia cases. Flow cytometry serve as a valuable diagnostic tool, providing a convenient and effective method for analyzing and categorizing CLPD into distinct entities. It also plays a crucial role in establishing the primary diagnosis, staging, assessing treatment response, and monitoring patients during follow-up [2].
CLPD represent a heterogeneous group of neoplasms characterized by the clonal proliferation of mature lymphocytes involving peripheral blood, bone marrow, and lymphoid tissues. Accurate subclassification is essential, as different subtypes exhibit variable clinical behaviour, prognostic implications, and therapeutic responses. Immunophenotyping by flow cytometry, in combination with clinical and morphological assessment, greatly improved diagnostic precision and helped distinguish between morphologically overlapping entities.
The present study analyzed the morphological, clinical, and immunophenotypic findings of all CLPD cases diagnosed over a one-year period at our institution, with the aim of determining the role of flow cytometry in the categorization of CLPD.
Materials and methods
This retrospective study was conducted over a period of one year, from January 1, 2024, to December 31, 2024. A total of 111 cases of chronic lymphoproliferative disorders (CLPD) were included. All cases were diagnosed using immunophenotypic analysis in the Oncopathology Department of the State Cancer Center. Cases with inconclusive flowcytometry analysis and without immunohistochemistry follow up, cases without morphological correlation and cases with reactive lymphocytosis were excluded form study. The study was approved by the Institutional Ethical Review Board.
Relevant clinico-radiological, hematological, and biochemical findings, along with immunophenotyping (IPT) data and cytogenetic reports, were retrieved from the hospital database. Peripheral smear, bone marrow aspiration, and trephine biopsy slides for each case were thoroughly examined.
Immunophenotyping
Immunophenotyping was performed using a FACS Canto eight-color flow cytometer (BD Biosciences) equipped with BD DIVA software, following the lyse–wash–stain–wash method. For each sample, 50,000 events were acquired and subsequently subjected to gating procedures to exclude debris on the SSC-A versus FSC-A plot. Singlets were then gated on the FSC-A versus FSC-H plot. The target lymphocyte population, characterized by low side scatter (SSC) and bright expression of CD45, was identified and selected using a scatter plot of SSC-A versus CD45 expression.
B-CLPD panel
The B-CLPD panel employed CD19 gating to analyze B cells characterized by low side scatter (SSC) and high CD19 expression. The markers included in the B-CLPD panel were CD45, CD19, CD20, CD79b, CD5, CD10, CD23, CD200, FMC7, CD43, and kappa and lambda light chains. Sequential addition of diagnostic markers was performed whenever considered necessary. For cases suspected of Hairy cell leukemia, additional markers such as CD11c, CD103, CD25, and CD123 were included. In cases with features suggestive of Plasma cell neoplasms, markers such as CD56, CD38, and CD138 were employed.
T-CLPD panel
The T-cell panel consisted of both surface and cytoplasmic markers, including cCD3, sCD3, CD5, CD7, CD2, CD4, CD8, and CD1a. In addition, NK-cell–associated markers CD16 and CD56 were analyzed to assist in identifying cases of NK-cell proliferative disorders.
Immunohistochemistry
In cases with inconclusive flow cytometric analysis, the bone marrow trephine biopsy/lymph node biopsy of same patient were subjected to immunohistochemistry analysis for completion of diagnosis. The applied panel are as follows:- B cell lymphomas panel: CD20, CD10, CD5,CD23, CD11b, cyclin D1,SOX11, BCL6, BCL2, Ki67, kappa, lambda, CD30,cMYC, MUM1; T/NKcell lymphoma panel: CD2, CD3, CD4,CD5,CD7, CD8, CD30,ALK, CD10, CD21,BCL6, CD56, CD16, Ki67.
Cytogenetic study
Heparinized samples of peripheral blood and bone marrow cells were cultured in RPMI-1460 medium. Cell proliferation was arrested at metaphase using colcemid. The samples were then processed on trypsin-treated slides, air-dried, and stained with Giemsa stain for G-banding of chromosomes.
Statistical analysis
Statistical analysis was performed using IBM SPSS software version 21. The Chi-square test was applied to compare the significance of parameters distinguishing chronic lymphocytic leukemia (CLL) from other CLPD subtypes. A p-value of <0.05 was considered statistically significant.
Results
The mean age of the patients was 58.4 years, with an age range of 21 to 92 years. Among the total 111 cases, males constituted 65.76% (73 cases), while females accounted for 34.24% (38 cases), giving a male-to-female ratio of 1.9:1. The occurrence of CLPD was predominantly observed in elderly males.
Clinicohematological profile
The most common clinical manifestation observed in the study cohort was lymphadenopathy (67%), with generalized lymphadenopathy present in 66.6% of these cases. Splenomegaly was noted in 59% of patients, while hepatomegaly was observed in 41.44%. Lymphocytosis was present in the majority of patients (81%), with a median absolute lymphocyte count (ALC) of 59,000 cells/mm³.
A smaller subset of patients exhibited cytopenias, including pancytopenia in 6.3% and bicytopenia in 2.7% of cases, while 9% of cases showed normal blood counts. The eight cases of pancytopenia included four cases of hairy cell leukemia, one case each of lymphoplasmacytic lymphoma (LPL), T-cell CLPD, follicular lymphoma (FL), and chronic lymphocytic leukemia (CLL).
The cut-off value for abnormal serum LDH was taken as 300 U/L. The majority of CLPD cases demonstrated LDH levels below 300 U/L. On comparison of hematological parameters, most cases in the CLL cohort had an ALC >59,000 cells/mm³ (p < 0.000) and LDH <300 U/L (p = 0.013), whereas the non-CLL cohort predominantly showed ALC <59,000 cells/mm³ (p < 0.000) and LDH >300 U/L (p = 0.013) (Table 1).
Table 1: Comparison of LDH and absolute lymphocyte Count (ALC) values in CLL and other CLPD cases.
|
Parameter
|
Value
|
CLL (%)
|
Other CLPD (%)
|
p value
|
|
LDH
|
<300
|
60 (82.19)
|
23 (60.52)
|
0.013
|
|
>300
|
13 (17.80)
|
15 (39.47)
|
|
ALC
|
<59,000
|
27 (36.98)
|
29 (76.31)
|
<0.000
|
|
>59,000
|
46 (63.01)
|
9 (23.68)
|
Involvement of body fluids was observed in six cases of CLPD, which included cerebrospinal fluid (CSF) in three cases, pleural fluid in two cases, and ascitic fluid in one case. Among these, five patients were previously diagnosed with lymphoproliferative disorders, and flow cytometry proved useful in confirming body fluid involvement in these cases. In one case, B-CLPD was primarily diagnosed on a CSF sample, and subsequent lymph node immunohistochemistry established the diagnosis of chronic lymphocytic leukemia (CLL).
Peripheral smear and bone marrow examination
Peripheral smear examination in cases presenting with lymphocytosis provided important diagnostic clues and guided the need for further confirmatory testing. The detailed findings from peripheral smear and bone marrow evaluation are summarized in Table 2 and illustrated in Figure 1.
Table 2: Peripheral smear findings in CLPD cases.
|
CLPD
|
Smear findings
|
|
CLL
|
peripheral lymphocytosis with smudge cells, where the atypical lymphocytes showed clumped soccer balls like chromatin
|
|
HCL
|
lymphoid cells with moderate amount of cytoplasm and spongy nuclear chromatin with circumferential cytoplasmic hairy projections (hairy cells).
|
|
FL
|
small lymphoid cells with cleaved nuclei
|
|
MZL
|
plasmacytoid lymphocytes
|
|
MCL
|
lymphocyte showing fish mouth appearance
|
|
LGL
|
Large lymphocytes, with moderate cytoplasm and cytoplasmic granules
|
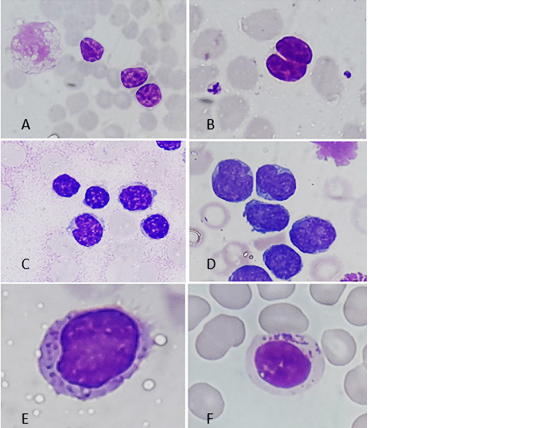
Figure 1: Morphology of lymphocytes in CLPDs (a) Chronic lymphocytic leukemia with smudge cells and lymphocytes with soccer ball type condensed chromatin. (b) Follicular lymphoma with lymphocytes showing cleaved nucleus. (c) Mantle cell lymphoma with lymphocyte showing fish mouth appearance, (d) Blastoid variant of MCL, (e) Hairy cell leukemia showing lymphocyte with spongy chromatin, moderate amount of cytoplasm and circumferential villous projections from cytoplasmic membrane, and (f) Large granular cell leukemia with lymphocyte showing coarse granules in cytoplasm.
The presence of smudge cells was not specific to CLL in this study and was also observed in other CLPD cases, mostly resulting from smearing artifacts and the fragile nature of leukemic cells. Bone marrow involvement was observed in 106 out of 111 cases (95.5%).
Cytogenetic study
Karyotyping in CLPD cases was often challenging because the cells frequently failed to grow in culture medium. Cytogenetic analysis was successfully performed in 39 cases. Among B-CLPD cases, hypodiploidy was observed in one case and trisomy 2 in another. In CLL cases, deletion of 13q was detected in one case, and trisomy 21 was observed in another. Fourteen cases exhibited a normal karyotype, while eleven cases did not yield analysable metaphases. Cytogenetic analysis in CLPD cases are difficult as these cases have low proliferative index which leads to less yield of analysable metaphase.
Immunophenotyping
Immunophenotypic analysis revealed that the majority of CLPD cases were B-CLPD (96.6%), with only a few cases classified as T-CLPD (3.6%). Among B-CLPD cases, chronic lymphocytic leukemia accounted for 65.76%, followed by mantle cell lymphoma (7%), hairy cell leukemia (5%), follicular lymphoma (4%), and splenic marginal zone lymphoma (2%). In 11% of cases, definitive subtyping of B-CLPD was not possible due to the absence of specific diagnostic markers; these cases were diagnosed as B-CLPD, and immunohistochemistry was recommended for further subtyping. The flow cytometry marker expression in CLL cases is summarized in Table 3.
Table 3: Flow cytometry marker expression in chronic lymphocytic leukemia (CLL) cases.
|
Markers
|
Intensity
|
CLL (%)
|
|
CD20
|
Bright
|
4 (5.479)
|
|
Moderate
|
61 (83.56)
|
|
Dim
|
7 (9.589)
|
|
None
|
1 (1.369)
|
|
CD5
|
Bright
|
1 (1.369)
|
|
Moderate
|
61 (83.56)
|
|
Dim
|
7 (9.589)
|
|
None
|
4 (5.479)
|
|
CD79b
|
Bright
|
0 (0)
|
|
Moderate
|
11 (15.06)
|
|
Dim
|
7 (9.589)
|
|
None
|
55 (75.34)
|
|
CD23
|
Bright
|
0 (0)
|
|
Moderate
|
51 (69.86)
|
|
Dim
|
15 (20.54)
|
|
None
|
7 (9.589)
|
|
CD200
|
Bright
|
1 (1.369)
|
|
Moderate
|
67 (91.78)
|
|
Dim
|
3 (4.109)
|
|
None
|
2 (2.739)
|
|
FMC7
|
Bright
|
0 (0)
|
|
Moderate
|
0 (0)
|
|
Dim
|
0 (0)
|
|
None
|
73 (100)
|
|
CD10
|
Bright
|
0 (0)
|
|
Moderate
|
0 (0)
|
|
Dim
|
0 (0)
|
|
None
|
73 (100)
|
|
CD43
|
Bright
|
0 (0)
|
|
Moderate
|
70 (95.89)
|
|
Dim
|
1 (1.369)
|
|
None
|
2 (2.739)
|
Eighty-seven percent of B-CLPD cases expressed only a single light chain in the gated population, with the majority being kappa light chain, thereby establishing the monoclonal nature of the abnormal lymphocytes. However, in 12% of cases, either complete absence of light chain expression (11 cases) or dual expression of light chains (2 cases) was observed.
Among 12 cases (11% of the total 111 CLPD cases) in which complete subtyping was not possible by flow cytometry, immunohistochemistry provided a definitive diagnosis in eleven cases, including marginal zone lymphoma (2 cases), splenic marginal zone lymphoma (1 case), small cell lymphoma (1 case), lymphoplasmacytic lymphoma (1 case), diffuse large B-cell lymphoma (1 case), and atypical CLL (1 case).
Four cases were diagnosed as T-CLPD by flow cytometry, comprising hepatosplenic T-cell lymphoma, large granular lymphocytic lymphoma, and T-cell prolymphocytic leukemia. One case of T-CLPD remained untyped even after immunohistochemistry correlation.
Discussion
Lymphoid malignancies may develop at any stage of B- or T-cell development and commonly present with lymphocytosis, generalized lymphadenopathy, marrow infiltration, hepatomegaly, or splenomegaly. Cases with persistent lymphocytosis over prolonged periods are generally classified as Chronic lymphoproliferative disorders (CLPD) [3]. Clear subtyping of CLPD is essential for definitive treatment, and diagnosis is established by integrating clinical, pathological, and flow cytometry findings. Immunohistochemistry (IHC) is recommended when flow cytometry is not feasible due to low cell counts, diluted bone marrow aspirate, or dry tap.
Consistent with other Indian studies by Dwivedi et al. and Gogia et al., CLPD in our study predominantly affected the elderly population, with peak incidence in the fifth to sixth decades, a mean age of 63 years, and a median age of 58 years [4, 5]. The youngest patient in our cohort was 20 years old, diagnosed with Hepatosplenic T-cell lymphoma (HSTCL), while the youngest CLL case was 33 years old. In a study by Montserrat et al. on CLL, the youngest cohort was 20 years old, with predominance of younger CLL cases (<50 years) in the 30–40-year age group [6].
Lymphocytosis was the most common presentation of CLPD; however, some entities, such as Hairy cell leukemia, are known to be associated with pancytopenia. Interestingly, in our study, one case each of CLL, Follicular lymphoma, Lymphoplasmacytic lymphoma (LPL), and T-CLPD presented with pancytopenia initially. In a study by Yan Li et al., pancytopenia was more commonly observed in Waldenström macroglobulinemia, Hairy cell leukemia, and Marginal zone lymphoma [7]. CLL may present with pancytopenia secondary to autoimmune destruction of blood cells. In our cohort, 4% (3 cases) of CLL patients developed autoimmune antibodies (direct Coombs test positive), with one of these cases presenting with pancytopenia, consistent with findings by Vitale et al., who reported 4–7% of CLL cases presenting with autoimmune cytopenias [8].
In our study, CLL was the most common B-CLPD, accounting for 67% of cases, followed by Mantle cell lymphoma (7%), Hairy cell leukemia (5%), Follicular lymphoma (4%), and Splenic marginal zone lymphoma (2%). These findings were in accordance with other studies, including Jemal et al., Oberley et al., Gujral et al., Simi et al., and Rai et al. [9–13]. The incidence of T-CLPD was only 3.6% in our study, similar to 4.2% reported in other Indian literature [4]. The different distribution of CLPD cases are as follows in Table 4.
Table 4: Distribution of CLPD cases in comparison to different studies.
|
CLPD
|
Our study
|
Oberley et al [10]
|
Simi et al [12]
|
Gujral et al [11]
|
Rai et al [13]
|
Dwivedi et al [4]
|
|
CLL
|
67%
|
66%
|
66.03%
|
68.5%
|
-
|
54.2%
|
|
MCL
|
7%
|
4%
|
4.31%
|
5.5%
|
-
|
-
|
|
FCL
|
4%
|
4%
|
6.7%
|
8.5%
|
-
|
-
|
|
MZL
|
2%
|
-
|
1.91%
|
5%
|
-
|
-
|
|
HCL
|
5%
|
2%
|
5.26%
|
5%
|
5.9%
|
2%
|
|
BCLPD unclassified
|
11.4%
|
18%
|
6.19%
|
7.5%
|
-
|
29.2%
|
|
BCLPD
|
96.4%
|
95%
|
90.4%
|
-
|
-
|
95.8%
|
|
TCLPD
|
3.6%
|
5%
|
9.4%
|
-
|
-
|
4.2%
|
Body fluid involvement by CLPD is uncommon. Pleural effusions complicate 6–20% of NHL cases [14]. A study from a tertiary medical center in Taiwan reported that lymphomatous effusions were more commonly caused by B-cell NHL (75.0%) than T-cell NHL (22.2%) or Classic Hodgkin lymphoma (2.8%) [15]. In our study, six cases (5.4%) presented with involvement of body fluids (3 CSF, 2 pleural fluid, 1 ascitic fluid), comprising five CLL and one mantle cell lymphoma case.
Flow cytometric analysis identified CLL cases by classical expression of CD5, CD23, CD200, monoclonal light chain restriction, and reduced expression of CD20 and CD79b, with absent FMC7. In our study, the sensitivity of CD200 for detecting CLL was 94%, whereas CD23 positivity had a sensitivity of 84%. This is consistent with Miao et al., who reported that CD200 had higher sensitivity than CD23 in distinguishing CLL from mantle cell lymphoma, likely due to increased CD23 expression in MCL [16]. CD5 negativity can occur in 7–20% of CLL cases [17]; in our cohort, CD5 negativity was observed in 5.4% of cases. CD200 expression helps identify CLL in atypical CD5/CD23-negative immunophenotypes [18]. Although CD200 expression is also observed in hairy cell leukemia (HCL), HCL can be distinguished from CLL by expression of CD25, CD11c, and CD103, along with absent CD5 and CD23.
In our study, one case of CLL exhibited an atypical immunophenotype with both CD5 and CD23 negative; CD200 positivity and FMC7 negativity aided in confirming the diagnosis. According to Rawstron et al., the minimal diagnostic panel for CLL includes CD19, CD5, CD20, CD23, kappa, and lambda [19]. However, this panel may be insufficient for identifying atypical cases, in which CD200 and FMC7 provide additional diagnostic confirmation. The combination of CD5 and CD23 positivity, CD200 positivity, and FMC7 negativity is diagnostic in most CLL cases, allowing accurate differentiation of CD5-positive, CD23-negative CLPD as either CLL or MCL [16, 20, 21].
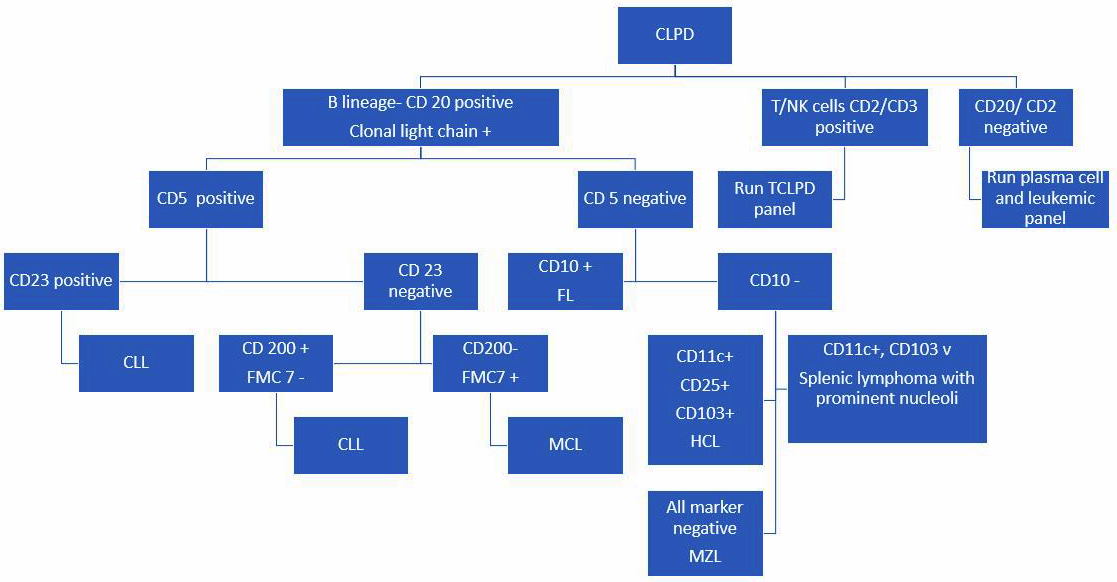
Figure 2: The concise representation of CLPD diagnostic algorithm.
In our study, 11% of B-CLPD cases remained unclassified by flow cytometry and required IHC for definitive diagnosis. Similarly, Miao et al. reported that 22.5% of cases remained unclassified by flow cytometry and required further investigation [16].
Flow cytometric analysis plays a crucial role in diagnosing and subtyping CLPD. It is a rapid and feasible method that provides definitive diagnosis in most cases, accurately identifying CLL and differentiating it from other morphological mimickers. It also quantifies abnormal lymphocytes in peripheral blood and provides reliable results even in cases with low diagnostic cell yield, such as hairy cell leukemia, where leukopenia and marrow fibrosis limit the detection of diagnostic cells. Only a minority of cases required IHC to confirm subtype and clonality. Novel IHC markers, such as LEF1 for CLL and MNDA and IRTA1 for marginal zone lymphoma, further aid diagnosis in challenging cases.
Cytogenetic studies are valuable for prognostic risk stratification in CLPD. In CLL, mutations in TP53 or deletions of 17p13, 11q, and 13q are associated with poor prognosis, whereas splenic marginal zone lymphoma has the most favorable prognosis, with 10-year survival rates up to 60–70%. CLPD can also transform into high-grade lymphomas, such as diffuse large B-cell lymphoma (Richter transformation), highlighting the importance of early diagnosis for optimal patient management.
Conclusion
Flow cytometry plays a central role in the evaluation and definitive diagnosis of chronic lymphoproliferative disorders (CLPD), enabling timely and appropriate patient management. It is also a valuable tool for confirming body fluid involvement in CLPD cases. The judicious application of diagnostic flow cytometry panels, integrated with morphological and clinical findings, optimizes resource utilization in a tertiary care setting and facilitates early and accurate patient management.
Conflict of interest
Authors declare no conflict of interest.
References
[1] UK Government. Chronic lymphoproliferative disorders. 2015. Available from: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/384520/chronic_lymphoproliferative_disorders.pdf
[2] Sah SP, Matutes E, Wotherspoon AC, Morilla R, Catovsky D. A comparison of flow cytometry, bone marrow biopsy, and bone marrow aspirates in the detection of lymphoid infiltration in B cell disorders. J Clin Pathol. 2003; 56:129–132.
[3] Braylan RC. Impact of flow cytometry on the diagnosis and characterization of lymphomas, chronic lymphoproliferative disorders and plasma cell neoplasias. Cytometry A. 2004; 58:57–61.
[4] Dwivedi E, Kushwaha R, Jain M, Yadav G, Verma SP, et al. To assess the correlation between bone marrow morphology and immunophenotypic findings in patients of chronic lymphoproliferative disorders (CLPD’s) and to assess the role of flowcytometric immunophenotyping in diagnosis and subclassification of CLPD’s. Int J Contemp Med Res [IJCMR]. 2019; 6(7).
[5] Gogia A, Sharma A, Raina V, Kumar L, Vishnubhatla S, et al. Assessment of 285 cases of chronic lymphocytic leukemia seen at single large tertiary center in Northern India. Leuk Lymphoma. 2012; 53:1961–1965.
[6] Montserrat E, Gomis F, Vallespi T, Rios A, Romero A, et al. Presenting features and prognosis of chronic lymphocytic leukemia in younger adults. Blood. 1991; 78:1545–1551.
[7] Wang Y, Huang Q, Wang Z, Lang T, Nie Y. Clinical and laboratory characteristics of 11 cases of chronic B lymphocyte proliferation disease without lymphoid proliferation. Cancer Res Clin. 2018; 6:51–56.
[8] Vitale M, Montalbano A, Salvetti C. Autoimmune complications in chronic lymphocytic leukemia in the era of targeted drugs. Cancers. 2020; 12:282.
[9] Jemal A, Siegel R, Ward E, Murray T, Xu J, et al. Cancer statistics, 2006. CA Cancer J Clin. 2006; 56:106–130.
[10] Oberley MJ, Fitzgerald S, Yang DT, Morgan A, Johnson J, et al. Value-based flow testing of chronic lymphoproliferative disorders: a quality improvement project to develop an algorithm to streamline testing and reduce costs. Am J Clin Pathol. 2014; 142:411–418.
[11] Gujral S, Polampalli SN, Badrinath Y, Kumar A, Subramanian PG, et al. Immunophenotyping of mature B-cell non Hodgkin lymphoma involving bone marrow and peripheral blood: critical analysis and insights gained at a tertiary care cancer hospital. Leuk Lymphoma. 2009; 50:1290–1300.
[12] Simi CM, Nair RA, Jacob PM, Jayasudha AV. Flow cytometry in chronic B-cell lymphoproliferative disorders: a retrospective study from a tertiary oncology centre, Trivandrum, India. J Clin Diagn Res. 2021.
[13] Rai V, Manimaran P, Saha A, Kailashiya V, Sawhney J, et al. Untangling the strands of hairy cell leukemia: the clinicopathological spectrum over eleven years at a tertiary care center. Discoveries (Craiova). 2023; 11:166.
[14] Weick JK, Kiely JM, Harrison EG, Carr DT, Scanlon PW. Pleural effusion in lymphoma. Cancer. 1973; 31:848–853.
[15] Wang RC, Chen YH, Chen BJ, Chuang SS. The cytopathological spectrum of lymphomas in effusions in a tertiary center in Taiwan. Diagn Cytopathol. 2021; 49:232–240.
[16] Miao Y, Cao L, Sun Q, Li XT, Wang Y, et al. Spectrum and immunophenotyping of 653 patients with B cell chronic lymphoproliferative disorders in China: a single centre analysis. Hematol Oncol. 2018; 36:121–127.
[17] Rossi D, Mauro G, Coco L. CD5 negative lymphocytosis mimicking typical B-chronic lymphocytic leukaemia. Description of 26 cases. Nouv Rev Fr Hematol. 1993; 35:451–455.
[18] El Desoukey NA, Afify RAA, Amin DG, Mohammed RF. CD200 expression in B-cell chronic lymphoproliferative disorders. J Investig Med. 2012; 60:56–61.
[19] Rawstron AC, Kreuzer KA, Soosapilla A, Spacek M, Stehlikova O, et al. Reproducible diagnosis of chronic lymphocytic leukemia by flow cytometry: an European Research Initiative on CLL (ERIC) & European Society for Clinical Cell Analysis (ESCCA) Harmonisation project. Cytometry B Clin Cytom. 2018; 94:121–128.
[20] Ahmad E, Garcia D, Davis BH. Clinical utility of CD23 and FMC7 antigen coexistent expression in B cell lymphoproliferative disorder subclassification. Cytometry. 2002; 50:1–7.
[21] Garcia DP, Rooney MT, Ahmad E, Davis BH. Diagnostic usefulness of CD23 and FMC-7 antigen expression patterns in B-cell lymphoma classification. Am J Clin Pathol. 2001; 115:258–265.