Full Text
Introduction
Micronucleus (MN) serves as valuable biomarkers in fine needle aspiration cytology (FNAC) smears of breast lesions. Micronuclei are small, extranuclear bodies that arise from chromosome fragments or whole chromosomes that fail to incorporate into the daughter nuclei during cell division. Micronuclei typically result due to lagging acentric fragments or as a result of failure to incorporate whole chromosomes into daughter nuclei during mitosis [1]. It is also a sensible indicator of the genetic damage and linked to various chromosome aberrations such as defective mitotic figures, chromosome fragmentation, mitotic cell death and catastrophe [2].
Micronuclei are morphologically similar to normal nuclei, except for the smaller size (ranging between 1/3rd and 1/16th of the nucleus), when stained by conventional smears. Micronucleus scoring (MN score) studies done in various preneoplastic and neoplastic lesions of oral, cervical, hepatic and urothelial regions have shown that there exists a clearcut correlation between degree of malignancy and MN score [3].
There are only a few literatures about occurrence of micronuclei in breast aspirates and its correlation with the type of lesion and grading of tumours. Borderline lesions like atypical hyperplasia and low-grade carcinoma in situ, or in papillary lesions etc. are difficult to ascertain on FNAC and in such situations, micronuclei may become important as they provide an objective and reproducible diagnosis.
The aim of this study was to investigate the potential relationship between micronucleus frequency and epithelial breast lesions, both benign and malignant, and to assess whether micronucleus scoring could serve as a supportive tool in categorizing diagnostically challenging borderline cases.
Materials and methods
This study is a prospective study conducted in the Department of Pathology at Mysore Medical College and Research Institute, Mysore over the period one year (January 2024 to November 2024). 208 breast cytology smears were examined. Ethics committee clearance was taken as per the Institutional protocol.
Fine needle aspiration cytology (FNAC) smears of breast lesions were collected. Informed consent was taken along with detailed clinical history and examination in case proforma.
Inclusion and exclusion criteria: This study included female patients presenting with breast lesions, with a preference for evaluating cells that were isolated or lying singly for accurate micronucleus counting. Male breast lesions were excluded, along with the cases of known breast carcinoma that had undergone neoadjuvant chemotherapy or radiotherapy. Additionally, cases involving fibrocystic disease, abscesses, and non-epithelial malignancies were not considered. Cellular clusters with obscured nuclear or cytoplasmic boundaries, as well as overlapping cells, degenerated cells, apoptotic cells, and cytoplasmic fragments, were also excluded from analysis.
Cytological assessment: Fine needle aspiration was conducted under aseptic conditions. The smears were stained with Haematoxylin and Eosin along with Grocott’s methenamine silver stain. All slides were reviewed, and the diagnoses were reported.
The Grocott’s Methenamine Silver (GMS) stain is a highly effective method for detecting micronuclei, offering several key advantages. It is highly sensitive, enabling the identification of even small micronuclei, and specific, minimising false positives and enhancing diagnostic accuracy. The distinct staining results make interpretation straightforward and rapid, facilitating quick analysis and decision-making. Additionally, it is a cost-effective technique. The GMS stain is also compatible with other stains like H&E, allowing for comprehensive cellular analysis, and supports quantitative assessment of micronuclei, providing valuable insights into their frequency and distribution The silver stain is beneficial in this context because it provides a high contrast between the micronuclei and the surrounding cellular material, making them easier to identify and count. These features make GMS stain a versatile and reliable tool [4].
Criteria for micronuclei: The HUman MicroNucleus (HUMN) project, an international collaborative effort, provided standardized guidelines for scoring micronuclei according to which micronucleus should have diameter ranging from 1/16 to 1/3 of the main nucleus. The staining intensity must be comparable to, or slightly weaker than that of the main nucleus. Additionally, micronuclei should be in the same plane of focus as the main nucleus and should be clearly separated from the main nucleus to ensure accurate identification [5].
Micronucleus scoring: Micronuclei will be counted in 1000 epithelial cells on breast cytology smears under oil immersion (100X magnification) by two independent observers. Structures resembling micronuclei—such as apoptotic bodies, staining artifacts, inflammatory cells, and fragmented nuclei which are excluded by counting the micronucleus in an intact cell without cell overlapping, clear intact cytoplasm, and having exact texture of chromatin as the main nucleus [6].
Pathologist review: Two pathologists independently observed the slides, and a mean value of their scores was taken to ensure reliability. Interobserver reliability was assessed using Cohen’s kappa value.
Categorization of cases: Cases were classified into five categories according to the International Academy of Cytology Yokohama System for Reporting Breast Fine-Needle Aspiration Biopsy Cytopathology [7] where the categories included were Insufficient (non-diagnostic), benign, atypical ductal hyperplasia, suspicious of malignancy, and carcinoma.
Robinson's criteria for grading malignant cases: This system assesses six cytological parameters, with each parameter scored between 1-3. The various parameters used are cell dissociation, cell size, cell uniformity, nucleolus, nuclear margin, nuclear chromatin [3].
Grading of tumours [3]: Grade I: Sum of scores between 6-11; Grade II: Sum of scores between 12-14; Grade III: Sum of scores between 15-18.
Statistical analysis
The data was collected, statistically evaluated and represented in terms of frequency distribution table. The categorical and counting variables were presented by frequencies and percentages in a tabular column. The date was analyzed using Statistical Package for Social Science (SPSS) for Windows, Version 20.0.
Results
In the present study, a total of 208 breast cytology cases meeting the defined inclusion and exclusion criteria were analyzed. These cases were diagnosed and categorised into ‘The Yokohama System for Reporting Breast Cytopathology’ [1]. Out of 208 cases, benign cases -Category 2 were 145 (69.7 %), which includes benign breast disease 16 cases (7.7%), fibroadenoma 88 cases (42.3 %), fibroadenosis 3 cases (1.4 %), usual ductal hyperplasia 34 cases (16.3%), phyllodes 4 (1.9%), Category 3 - atypical ductal hyperplasia, 18 cases (1.9%), Category 4 - suspicious of malignancy, 5 cases (2.4%) and Category 5 – malignancy 40 cases (19.2%) (Table 1).
Table 1: Yokohoma category distribution.
|
Benign breast disease
|
Percentage
|
|
|
Fibroadenoma
|
42.3%
|
|
|
Fibroadenosis
|
1.4%
|
|
|
Usual ductal hyperplasia
|
16.3%
|
|
|
Phyllodes
|
1.9%
|
|
Atypical ductal hyperplasia
|
8.7%
|
|
Suspicious of malignancy
|
2.4%
|
|
Carcinoma
|
19.2%
|
|
Total -208
|
100%
|
The Yokohama classifications demonstrate a distinct and gradual transition across different age groups. Yokohama Category 2 is most commonly observed in younger individuals (under 20 and 21–30 years) and shows a declining trend in successive age brackets. Category 3 emerges predominantly in the 21–30 age group, increases in frequency between 31–50 years, and then shows a mild decline with advancing age. Category 4 begins to appear more prominently from the 31–40 age group, peaks between 41–60 years, and remains present thereafter. Category 5 shows minimal representation in the younger population, gradually rises from 31–50 years, and becomes the most frequently encountered category in individuals aged 61–70 years. This age-related distribution reflects a continuous and age-correlated progression across the Yokohama categories, highlighting the evolving nature of breast lesions with increasing age (Figure 1).
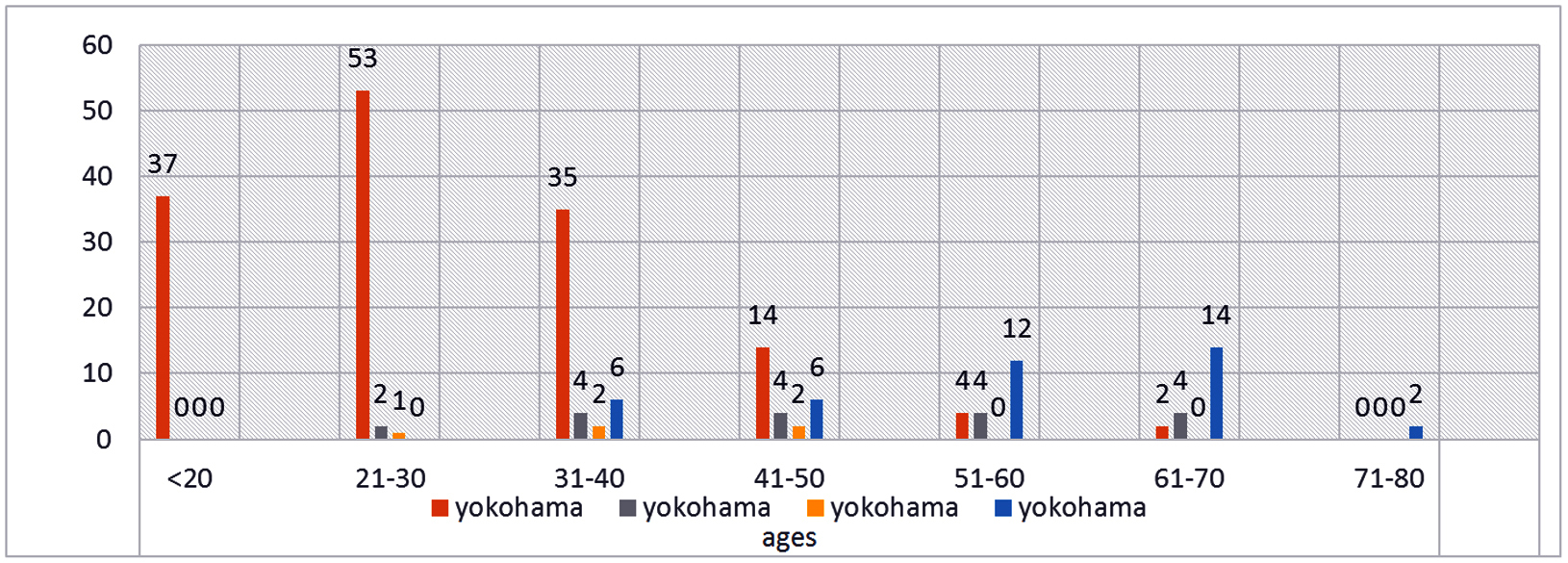
Figure 1: Age distribution according Yokohoma classification.
Category-2, benign, in which benign breast disease having the lowest MN scores were observed in this category, with an average score of 0.3 and a range of 0-1. This minimal MN count reflects the lack of significant genetic damage or chromosomal instability, consistent with the benign nature of these lesions. Fibroadenomas exhibited slightly higher MN scores, averaging 0.8 with a range of 0-2. While still indicative of benign conditions, the slightly elevated scores may reflect some degree of proliferative activity, albeit non-malignant. The average MN score for fibroadenosis was 1.3, with a range of 0-2. These slightly increased scores could be attributed to the mild cellular changes associated with this condition. However, the scores remain within the benign spectrum. Usual ductal hyperplasia demonstrated an average MN score of 1.2, with a range of 1-3. While categorized under benign conditions, the presence of higher MN counts compared to other benign lesions reflects the increased cellular activity associated with hyperplasia. Phyllodes tumours had an average MN score of 2.0, with a range of 2-3. This intermediate score may indicate the biphasic nature of phyllodes tumours, as they have both benign and malignant potential, necessitating careful evaluation (Figure 2 and Table 2).
Category-3, Atypical Ductal Hyperplasia (ADH) showed a significantly higher MN score, averaging 3.4 and ranging from 2-5. As a borderline lesion, the increased MN score reflects the genomic instability and potential for progression to malignancy, underscoring the diagnostic importance of MN scoring in such cases (Figure 3 and Table 2).
Category-4, Suspicious of Malignancy (SOM) Lesions exhibited an average MN score of 5.2, with a range of 5-6. This reflects a marked increase in chromosomal damage, aligning with the heightened risk of malignancy in such cases (Figure 4 and Table 2).
Table 2: Average micronucleus score in categories of Yokohoma classification.
|
Yokohoma category
|
Average micronucleus
|
Range of micronucleus
|
|
Benign breast disease
|
0.3
|
0-1
|
|
Fibroadenoma
|
0.8
|
0-2
|
|
Fibroadenosis
|
1.3
|
0-2
|
|
Usual ductal hyperplasia
|
1.2
|
1-3
|
|
Phyllodes
|
2.0
|
2-3
|
|
Atypical ductal hyperplasia
|
3.4
|
2-5
|
|
Suspicious of malignancy
|
5.2
|
5-6
|
|
Carcinoma
|
13.5
|
7-24
|
Category-5, Carcinoma, highest MN scores were observed in carcinoma cases, with an average score of 13.5 and a wide range of 7-24. Malignant cases were sub categorised using the Robinson's cytological scoring system. Grade 1 (Well-differentiated) the MN scores averaged 8.9, with a range of 7-11. These tumours are well-differentiated, with cellular structure resembling normal tissue. Grade 2 (Moderately differentiated) tumours exhibited an average MN score of 13.8, with a range of 12-15. Grade 3 (Poorly differentiated) had the highest MN scores, with an average of 17.4 and a range of 15-22. The progression in MN scores from Grade 1 to Grade 3 underscores the correlation between MN scoring and the degree of malignancy. These findings validate the utility of MN scoring in assessing tumour aggressiveness and grading malignant breast lesions effectively (Figures 5, 6, 7 & Table 3).
Table 3: Average micronucleus score in malignant cases.
|
Robinsons cytological scoring system for malignant cases
|
Average micronucleus
|
Range of micronucleus
|
|
Grade 1
|
8.9
|
7 -11
|
|
Grade 2
|
13.8
|
12-15
|
|
Grade 3
|
17.4
|
15-22
|
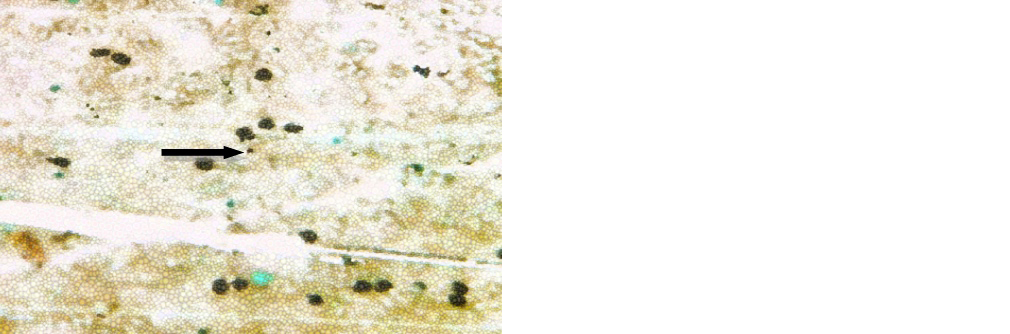
Figure 2: Benign100x (GMS).
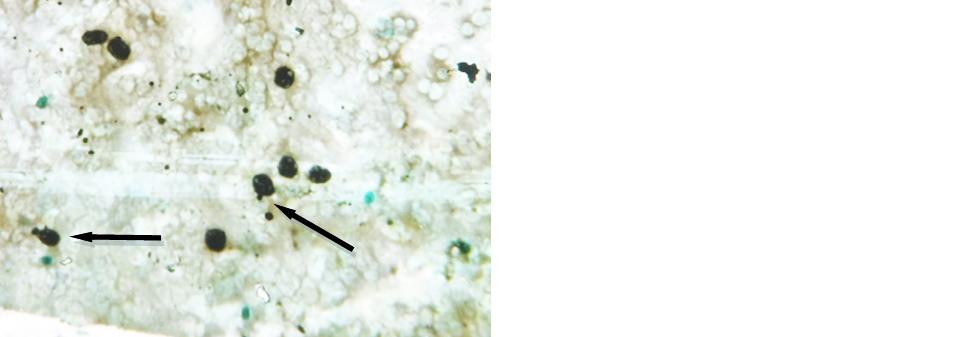
Figure 3: ADH, 100x (GMS).

Figure 4: SOM, 100x (GMS).

Figure 5: Carcinoma, 100x (GMS).
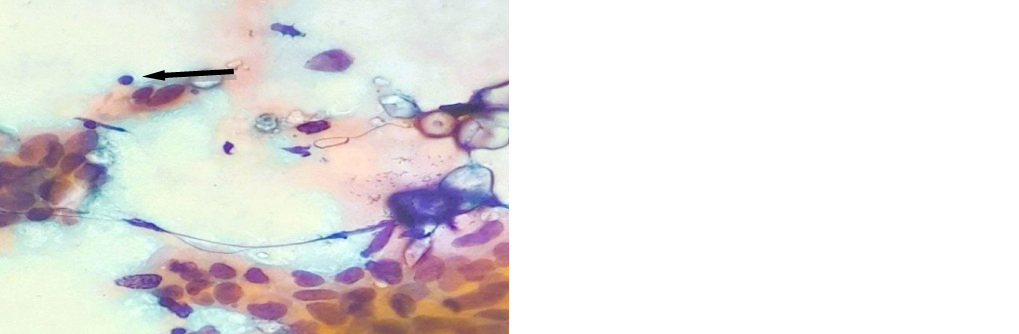
Figure 6: Carcinoma, 100x (Pap stain).
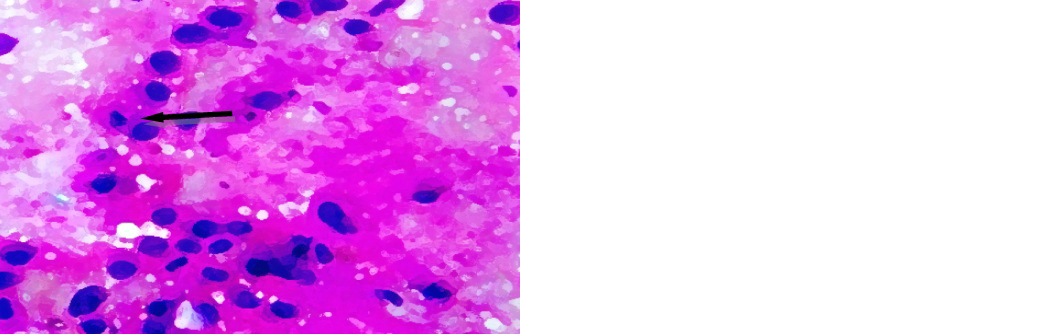
Figure 7: Carcinoma, 100x (H &E stain).
Discussion
The presence of micronuclei in cells is widely recognized as a reliable indicator of genomic instability, a hallmark of cancerous cells. In this study, the frequency of micronuclei in from breast cancer patients was evaluated using Gorocott’s methenamine silver staining. MN formation, caused by errors in cell division and chromosome disjunction, serves as an indicator of underlying genetic damage.
The present study utilized Grocott's Methenamine Silver (GMS) stain, known for its high sensitivity and contrast, to assess micronucleus (MN) scores across various breast cytological categories. Results indicated a progressive increase in MN scores with the severity of the condition, ranging from 0.3 in benign breast disease to 13.5 in carcinoma.
Saranyabai et al. [1] used Haematoxylin and Eosin (H&E) staining, which is effective for general cytological evaluations but less precise in MN detection compared to GMS. Carcinoma cases reported higher MN scores (20), reflecting some variability due to the staining method. Sylvia et al. [2] study used Leishman staining, primarily applied in haematological evaluations, with carcinoma cases recording slightly lower MN scores (19.2). While effective, this method struggled with detecting finer nuclear details in breast cytological samples, highlighting the advantages of more sensitive techniques such as GMS staining (Tables 4 & 5).
Till date, no previous studies are available which has evaluated micronuclei in breast cytology using Grocott’s Methenamine Silver (GMS) stain. This study was undertaken on an experimental basis to assess the feasibility and compatibility of GMS staining for micronucleus identification on cytological smears.
GMS staining, due to its high sensitivity and strong contrast, represents an effective non-invasive method for detecting micronuclei (MNs), which can aid in the early detection of breast cancer and risk assessment. A gradual increase in MN scores is observed from benign to malignant lesions, reflecting progressive genomic damage across this spectrum. Therefore, GMS staining may serve as a valuable adjunct tool for classifying breast lesions on cytology, particularly in borderline or grey zone cases such as ductal hyperplasia. Further research comparing GMS with established DNA-specific stains is necessary to validate its utility in micronuclear evaluation. Additionally, monitoring MN frequency during chemotherapy or radiotherapy could provide insights into genotoxic effects, supporting more personalized treatment strategies that minimize damage to healthy cells.
Table 4: Comparing average micronucleus score in categories of Yokohoma classification with other studies. Average micronucleus score/ 1000 cells (100X).
|
Cytological category
|
Present study
|
Saranyabai et al. [1]
|
Sylvia et al. [2]
|
|
Benign breast disease
|
0.3
|
-
|
-
|
|
Fibroadenoma
|
0.8
|
1
|
<1
|
|
Fibroadenosis
|
1.3
|
2
|
2
|
|
Usual ductal hyperplasia
|
1.2
|
4
|
2.9
|
|
Phyllodes
|
2.0
|
-
|
-
|
|
Atypical ductal hyperplasia
|
3.4
|
6
|
6.6
|
|
Suspicious of malignancy
|
5.2
|
-
|
-
|
|
Carcinoma
|
13.5
|
20
|
19.2
|
Table 5: Comparing average micronucleus score in malignant cases with other studies. Average micronucleus score/ 1000 cells (100X) for malignant cases.
|
Robinsons cytological
scoring system
|
Present study
|
Saranyabaiet al. [1]
|
Sylvia et al. [2]
|
|
Grade 1
|
8.9
|
8
|
13.2
|
|
Grade 2
|
13.8
|
18.5
|
20.35
|
|
Grade 3
|
17.4
|
27.5
|
27.5
|
Conclusion
Micronucleus scores show a clear correlation with malignancy severity, reinforcing their value as reliable biomarkers for genetic damage and chromosomal aberrations. This technique offers an objective and reproducible approach, improving diagnostic precision, especially in borderline cases where conventional FNAC may provide inconclusive results. As indicators of genomic instability, micronuclei significantly enhance diagnostic accuracy. With its superior sensitivity and contrast, GMS staining enables more precise pre-operative diagnosis of malignant lesions. This method marks a groundbreaking step in molecular diagnostics, being the first of its kind. Its affordability and practicality make it a strong alternative to other molecular diagnostic tools, ensuring availability at skilled health centre conveniently accessible to patients.
Conflicts of interest
Authors declare no conflicts of interest.
References
[1] Saranyabai S, Sangeetha N, Pradeep P, Nidhya E, Venkataraman B. Study of micronuclei as a potent biomarker in breast cytology aspirates. Panacea J Med Sci. 2022; 12:651–656.
[2] Sylvia MT, Baskaran L, Bhat RV. Micronucleus study on breast cytology aspirate smears and its diagnostic utility. J Cytol. 2018; 35:22–26.
[3] Katta R, Rao KN, Murthy BK, Kalam SR. Micronucleus scoring in fine needle aspiration cytology of breast lesions—a retrospective analytical study. J Clin Diagn Res. 2022; 16:EC52–EC55.
[4] Bancroft JD, Gamble M. Theory and practice of histological techniques. 6th ed. Philadelphia: Churchill Livingstone Elsevier; 2008. p.433–435.
[5] Fenech M, Holland N, Zeiger E, Chang WP, Burgaz S, et al. The HUMN and HUMNxL international collaboration projects on human micronucleus assays in lymphocytes and buccal cells—past, present and future. Mutagenesis. 2011; 26:239–245.
[6] Field AS, Raymond WA, Rickard M, Arnold L, Brachtel EF, et al. the international academy of cytology Yokohama system for reporting breast fine-needle aspiration biopsy cytopathology. Acta Cytol. 2019; 63:1–17.
[7] Yu W, Gan Q, Gong Y. The Yokohama system for reporting breast cytopathology. J Clin Transl Pathol. 2023; 3:99–105.
[8] Samanta S, Dey P. Micronucleus and its applications. Diagn Cytopathol. 2012; 40:84–90.
[9] Heddle JA, Fenech M, Hayashi M, MacGregor JT. Reflections on the development of micronucleus assays. Mutagenesis. 2011; 26:3–10.
[10] Sneige N, White VA, Katz RL, Troncoso P, Libshitz HI, et al. Ductal carcinoma-in-situ of the breast: fine-needle aspiration cytology of 12 cases. Diagn Cytopathol. 1989; 5:371–377.
[11] Hemalatha A, Suresh TN, Kumar MLH. Micronuclei in breast aspirates: is scoring them helpful? J Cancer Res Ther. 2014; 10:309–311.
[12] Goel S, Bhatia A, Dey P. Spontaneously occurring micronuclei in infiltrating ductal carcinoma of breast: a potential biomarker for aggressive phenotype detection. Diagnostic Cytopathology. 2013; 41:296–302.
[13] Basher ES, Hassan AM, Awad MH. Micronuclei and Ag NOR as biomarkers in fine needle aspiration cytology of breast tumor. Am J Res Commun. 2016; 4:136–143.
[14] Mangam SN, Deshmukh AV, Shivkumar VB. Role of micronucleus assay as an indicator of chromosomal instability in aspirates of breast carcinoma. J Cancer Res Pract. 2021; 8:20–25.
[15] Cardinale F, Bocci G, Prisco MG. Role of micronucleus test in predicting breast cancer susceptibility: a systematic review and meta-analysis. Br J Cancer. 2012; 106:780–790.
[16] Bonassi S, El-Zein R, Bolognesi C, Fenech M. Micronuclei frequency in peripheral blood lymphocytes and cancer risk: evidence from human studies. Mutagenesis. 2011; 26:93–100.