Full Text
Introduction
Laparoscopic partial nephrectomy (LPN) for kidney tumors is gradually replacing classic open surgery and becoming a standard procedure performed routinely at major surgical centers. It is challenging, as it requires surgeons with more experience and advanced laparoscopic surgical skills and requires medical facilities equipped with appropriate equipment and supplies. LPN is often indicated for renal tumors ≤7 cm in size (pT1), with oncological results equivalent to laparoscopic radical nephrectomy (LRN). According to data sources, in 2010–2011, the 5-year cancer-free survival rate after LPN was 86% [1-3].
In Vietnam, major surgical centers, such as Viet Duc Hospital, Hue Central Hospital, and Binh Dan Hospital, commenced LPN very early. Thanh Hoa General Hospital commenced this procedure in late 2016 and has indicated and regularly performed LPN for cases of pT1 stage kidney tumors.
Therefore, we investigated LPN for the treatment of large kidney tumors to evaluate the characteristics and outcomes of retroperitoneal LPN (rLPN) in the treatment of large kidney tumors at provincial general hospitals.
Participants and research methods
This study included 25 patients (14 men, 11 women) diagnosed with stage pT1b renal tumor (tumor size >4 cm and ≤7 cm) on computed tomography (CT) scan images and who underwent rLPN at Thanh Hoa General Hospital between January 2017 and May 2023. The criteria for selecting indications for rLPN to treat large renal tumors were tumors at stage pT1b (AJCC) [4], absence of regional lymph nodes or distant metastases, absence of contraindications to retroperitoneal laparoscopic surgery, and no retroperitoneal surgery on the same side.
The study was conducted using a cross-sectional, prospective and retrospective data method. All patients were diagnosed and operated on using an Olympus 4K endoscopy system using Ethicon Endo-Surgery Hamonic Gen 11 with an endoscopic bulldog and Hem-o-lock clips of sizes 5 mm and 10 mm.
The complexity of the tumor according to the R.E.N.A.L system was assessed based on five criteria: radius, exophytic/endophytic type, nearness to collecting system or sinus, anterior/posterior, and location relative to polar lines. The scores were categorized as low (4–6 points), medium (7–9 points), and high (10–12 points) [5].
Patient preparation and surgical procedure
All patients were anesthetized via endotracheal intubation with a gastric tube and a urethral catheter and the bowel was cleaned before surgery. The research was approved according to Decision No. 830/BVĐKT-HĐKH. Thanh Hoa General Hospital allows the use and publication of data.
The renal artery and the tumor were dissected and exposed, and the area surrounding the tumor was marked with a monopolar electrosurgical scalpel. The renal artery was clamped using the endoscopic bulldog (or a rubber band). The endoscissors was used to cut the tumor as enucleation to the margin marked. The inner and outer layers of the renal parenchyma were sutured (using 2-0 or 3-0 Vicryl or V-lock with Hem-o-lock to ensure clamping at the end). Bolsters may be placed. The bulldog clamp is released and checked for hemostasis. The drainage is placed next to the kidney, and the trocars are closed.
Patient care and indicators
The patients’ pulse, arterial blood pressure, respiratory rate, temperature, and drainage status were monitored after surgery. Antibiotics used include those in the β-lactamine group and the quinolone group. Non-steroidal analgesics and morphine are also administered. The patient is re-examined after 3–6 months and 3 years (clinical examination, laboratory tests, X-ray, and ultrasonography).
Results
Pathological characteristics
The rate of incidental detection of kidney tumors was 68%, mainly detected by ultrasound through physical examination. Common symptoms were lumbar pain, hematuria, and weight loss; however, no patient had a palpable lumbar mass. For all patients, CT scans were used to assess tumor staging and complexity.
Table 1: Pathological characteristics.
|
Pathological characteristics
|
Average (range) or number (%)
|
|
Age (years)
|
56.4 (25–71)
|
|
Sex (male/female)
|
14/11
|
|
Body mass index
|
23.4 (18.2–26.6)
|
|
Kidney tumor side (right/left)
|
13/12
|
|
Tumor size (cm)
|
5.2 (4.1–6.5)
|
|
Tumor location
|
Upper pole
|
7 (28%)
|
|
Middle section
|
10 (40%)
|
|
Lower pole
|
8 (32%)
|
|
Nephrometry score*
|
Low
|
5 (20%)
|
|
Medium
|
13 (52%)
|
|
High
|
7 (28%)
|
|
Staging**
|
pT1b
|
25 (100%)
|
|
Histopathology
|
Clear cell tumor (01 cystic type)
|
16 (64%)
|
|
Papilloma
|
5 (20%)
|
|
Chromophobe
|
4 (16%)
|
Abbreviations: *. According to the R.E.N.A.L system; **. According to AJCC 2017.
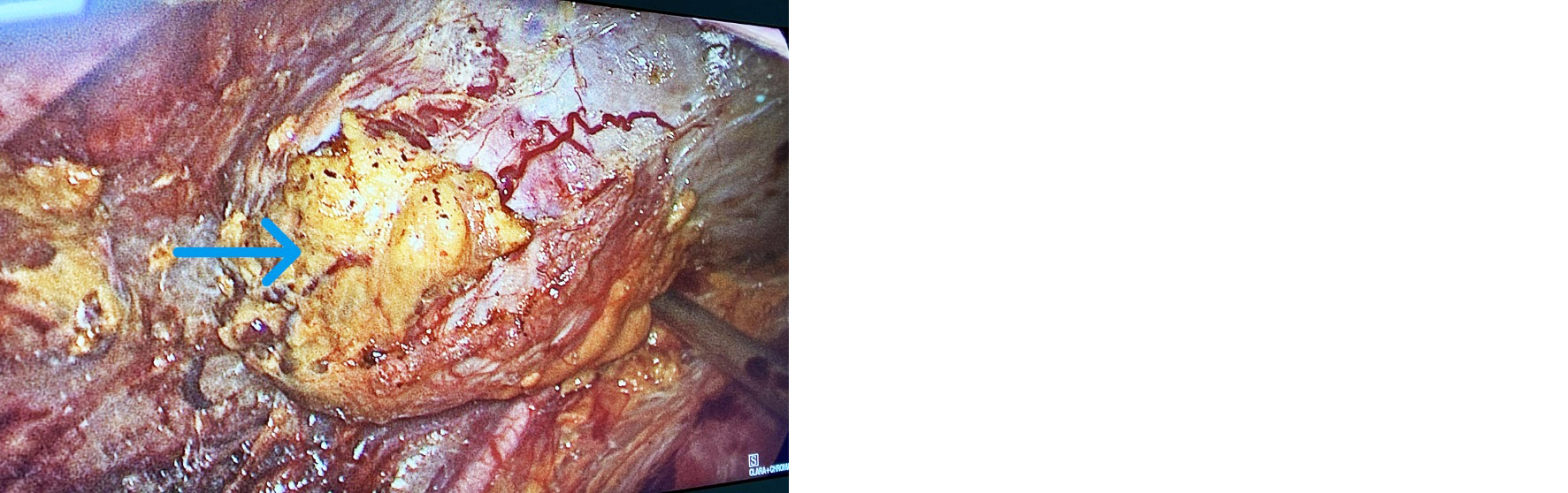
Figure 1: A tumor in the lower pole of the right kidney (arrow).
Table 2: Surgical characteristics.
|
Surgical characteristics
|
Average (range) or number (%)
|
|
Number of trocars
|
3
|
21 (84%)
|
|
4
|
4 (16%)
|
|
Operative time (minutes)
(One case with method change was not included)
|
87.9 (55–120)
|
|
Warm ischemia time* (minutes)
|
25.6 (20–40)
|
|
Renal artery control
|
Rubber band
|
6 (24%)
|
|
Bulldog
|
19 (76%)
|
|
Tumor margin
|
Positive
|
0 (0%)
|
|
Negative
|
10 (100%)
|
|
Blood loss (ml)
|
86.8 (50–250)
|
|
Drain removal time (days)
|
5 (4–6)
|
|
Hospital stay (days)
|
6 (5–7)
|
|
Surgical complications**
|
1 (4%)
|
|
Postoperative complications***
|
2 (8%)
|
|
Postoperative check-up****
|
3-6 months
|
20 (80%)
|
|
3 years
|
6 (24%)
|
Abbreviations: *. The WIT is calculated from the time control until the release of the renal artery; **. Intraoperative bleeding complications due to the tumor in the middle part of the kidney, deeply invading the renal sinus, later had to convert to laparoscopic radical nephrectomy; ***. Complications: one case of retroperitoneal fluid accumulation due to obstruction of surgical drainage, later performed ultrasound-guided aspiration and flushing of the fluid accumulation; one case of bleeding 5 days after discharge, The patient returned home with bleeding, with symptoms of mild hematuria and perirenal hematoma. The patient was brought back to the hospital (twice) for blood transfusion, embolization, and surgery to remove the retroperitoneal hematoma; ****. Patients were examined postoperatively by ultrasound, renal function tests, chest x-ray and clinical examination, no patient had urinary leakage, trocar site infection, local recurrence or distant metastasis (cases operated in early 2017 were evaluated after 3 years).
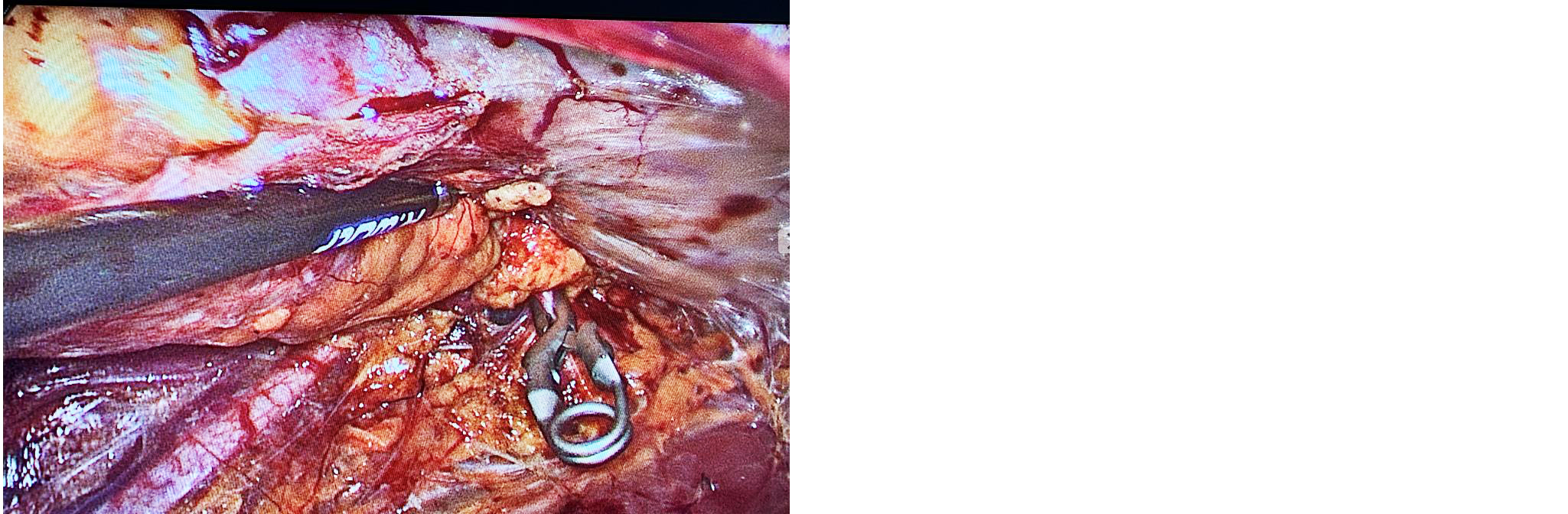
Figure 2: The endoscopic bulldog clamped on the right renal artery.
Table 3: Relationship between operative time and type of suture used.
|
Time (minutes)
|
≤60
|
>60 & ≤90
|
>90 & ≤120
|
>120
|
Total
|
|
Using V-lock (17 cases)
|
1
|
9
|
7
|
0
|
17
|
|
Using Vicryl (7 cases)
|
0
|
3
|
4
|
0
|
7
|
|
Total
|
1
|
12
|
11
|
0
|
24*
|
Note: *: One case in which LRN was eventually performed due to bleeding was excluded.
Later, radical laparoscopic nephrectomy had to be performed due to bleeding after releasing the renal artery control, with a surgical time of 130 minutes; Using the chi-square test with Yates correction, the chi-square result was 0.24. Hence, there was no statistically significant relationship between the operative time and the type of suture used for renorrhaphy; Using Vicryl sutures usually requires more Hem-o-lock and has a longer renorrhaphy time than using V-lock sutures.
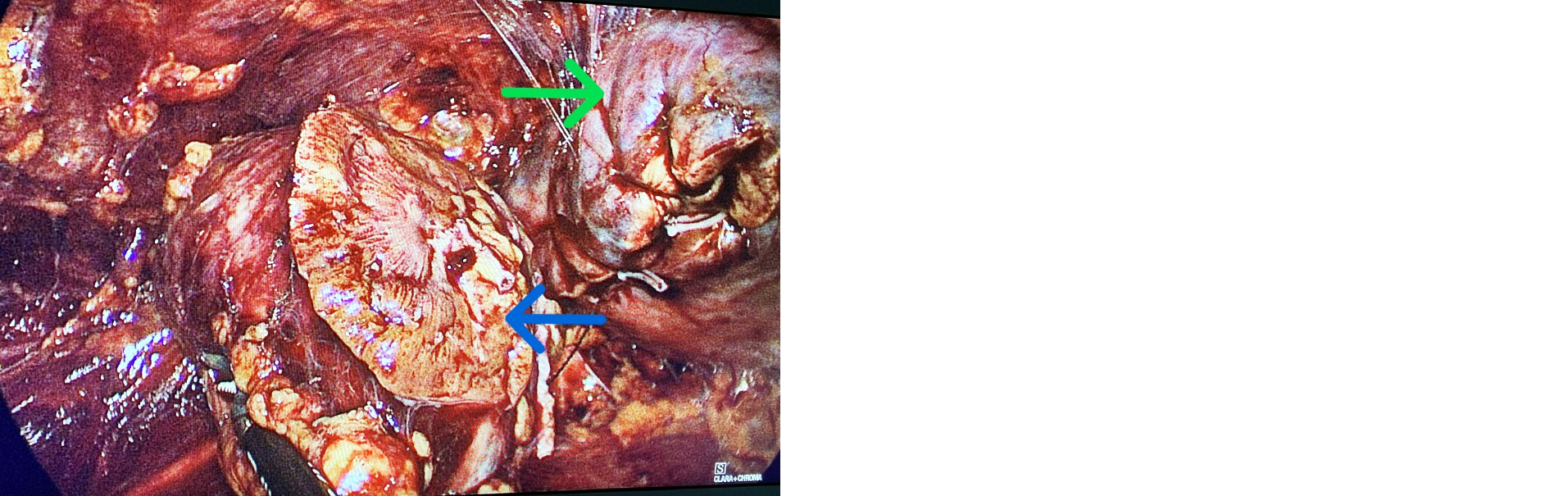
Figure 3: The tumor occupying half of the right kidney was removed. The remaining right kidney underwent renorrhaphy (the blue arrow indicates the tumor; the green arrow indicates the remaining right kidney).
Table 4: Association between blood loss and the used bolster.
|
Blood loss
|
≤60 ml
|
>60 & ≤80 ml
|
>80 & ≤100 ml
|
>100 ml
|
Total
|
|
Use of bolster
|
1
|
2
|
3
|
2
|
8
|
|
Non-use of bolster
|
4
|
7
|
5
|
1
|
17
|
|
Total
|
5
|
9
|
8
|
3
|
25
|
Note: Using the chi-square test with Yates correction, the chi-square result was 0.61. Hence, there was no statistically significant association between blood loss and the use or non-use of bolsters; Using a bolster is often more suitable for cases of tumors in the middle part of the kidney and tumors on the lateral border of the kidney.
Discussion
Partial nephrectomy has been performed frequently in the last two decades to treat renal tumors, with oncological treatment results equivalent to radical nephrectomy. In the long term, partial nephrectomy can preserve functional kidney units. LPN is a minimally invasive surgery with results similar to those of open partial nephrectomy. Therefore, LPN has been gradually accepted and is the preferred choice in the treatment of benign renal tumors and renal cancer at stage pT1 or lower [1, 6].
According to the guidelines of the Canadian Cancer Society, the preferred treatment options for renal tumors at stage pT1a (tumor size ≤4 cm) are partial nephrectomy, radiofrequency ablation, and cryoablation. For renal tumors at stage pT1b (tumor size >4 cm and ≤7 cm), only partial nephrectomy or radical nephrectomy should be performed. The choice of laparoscopic or open surgery for partial nephrectomy or radical nephrectomy depends on the surgeon's ability and the equipment available in the facility [2].
According to the new EAU Guidelines (2022), partial nephrectomy is still better than radical nephrectomy for renal tumors at stage pT1 (evidence: 1b). Laparoscopic or open surgery for partial nephrectomy gives equivalent results; the choice depends on the surgeon's experience and skills and the surgical facility's equipment capabilities [4].
Many authors have relied on the tumor complexity scoring system (usually calculated according to the R.E.N.A.L. system of Kutikov and Uzzo in 2009) to predict the difficulty of partial nephrectomy, especially using laparoscopic surgery. This system is based on five criteria for scoring; each criterion is graded 1–3 points, and the two criteria given the most attention are tumor size and the nearest distance from the tumor to the renal pelvis system (nearness to the collecting system). For partial nephrectomy by laparoscopic surgery, patients with renal tumors with low complexity (4–6 points) and medium complexity (7–9 points) should be selected, whereas those with high complexity (10–12 points) should be considered [5, 6].
Nadu et al. found that in the first 30 cases, although the average tumor size was 2.6 cm, the rate of surgical method conversion was up to 10%; in the next 110 cases, the rate of conversion was only 2.7% with an average tumor size of 3.9 cm [7]. The patients in our study had an average tumor size of 5.2 cm; the R.E.N.A.L. score was at medium and high levels, accounting for 80%, with successful surgical results in 24/25 cases (96%). A patient underwent conversion to LRN; this was a case of a renal tumor >5 cm in size, endophytic, located in the middle, and crossing the polar lines.
Access route and number of trocars: According to the guidelines in Laparoscopic Techniques in Uro-Oncology, the transabdominal route should be used for cases of tumors located on the anterior or anterolateral side or large tumors at the renal pole that require at least 30% of the kidney to be removed, whereas the retroperitoneal access should be used for tumors located on the posterior or posterolateral side [1].
Many authors believe that the choice of access route for LPN is often based on the surgeon's habits and the surgical facility. The EAU Guidelines (2022) show that laparoscopic surgery using the retroperitoneal or transabdominal route for LPN has similar intraoperative and postoperative results [4].
In this study, we performed all cases of rLPN, which has the advantage of directly accessing the renal artery for control; however, the cavity creation must be done to the maximum extent to facilitate the dissection process. The patients in our study had 28% of tumors located in the upper pole, 40% of tumors located in the middle part, and 32% of tumors located in the lower pole; LPN through the retroperitoneal route with 3–4 trocars working was performed in all cases.
Control of renal blood vessels and warm ischemia time (WIT): Control of renal pedicle vessels can cause renal ischemia. The maximum WIT of the kidney can be up to 30 minutes, which will limit the surgical time, especially for large and complex renal tumors [1,8]. Some authors suggest that renal hypothermia should be performed when necessary, at which time the cold ischemia time can be extended to 60 minutes [3]. An early release of renal artery control after completion of the inner layer suture can reduce the WIT by more than 50%. Some authors have recently reported successful cases of super-selective control [2]. In 2010, Wolf reported the following for LPN: for endophytic renal tumors (tumor depth) >5 mm, renal pedicle clamping should be performed; renal pedicle clamping is not necessary when tumors are ≤5 mm [3].
In our study, most cases were controlled by a laparoscopic bulldog (76%); a rubber band was used in only 24% of cases and usually only in the first cases. The average WIT is 25.6 minutes; the case with a WIT of 40 minutes was that of a large tumor deep in the renal parenchyma. Releasing the renal artery control caused excessive bleeding, which led to a conversion to LRN. The results of our WIT are not significantly different from those of Sinh et al. (10–20 minutes) and Nadu (30 minutes) [7,9].
Tumor resection and renorrhaphy: Before performing tumor resection during LPN, the perirenal fat tissue should be completely removed from the kidney surface (with care taken not to lose the renal capsule), and the fat layer adjacent and attached to the tumor should be left to facilitate tumor lifting during resection and help clarify the histopathological staging process later become clearer. The tumor is resected in a nuclear shape to the normal kidney (note that the surgical margin must be negative). To do this well, many authors recommend using non-disposable laparoscopic cold scissors. During the resection, attention should always be paid to controlling the points of violation of the renal pelvis or renal blood vessels for accurate handling. We often use clips to clamp the points of violation of the renal pelvis or blood vessels to reduce bleeding and avoid future leakage.
In our study group, we biopsied the surgical margin in 10 cases (40%), with the pathological results showing a negative rate of 100%. According to the EAU's Guideline (2022), the positive surgical margin rate is 2–8%. Patients with positive surgical margins do not need to be re-intervened immediately because only a few of these patients will have tumor recurrence, in which case closer monitoring and re-excision surgery may be necessary [4]. In this study, a bolster was not used in 17/25 (68%) of the cases; bolsters are often more suitable for cases of tumors in the middle part and tumors on the outer edge of the kidney. The V-lock suture is often more convenient than the Vicryl suture; the V-lock suture also requires less Hem-o-lock and has a faster renorrhaphy time than the Vicryl suture [10].
Surgical results: The mean operative time was 87.9 minutes. The operative time was often influenced by the dissection before and after renal artery clamping (the time between tumor resection and renorrhaphy suture was less [approximately 25.6 minutes]). There was no correlation between the type of renal parenchymal suture and the operative time.
The average blood loss was 86.8 ml; this estimated blood loss depended on the tumor resection and renorrhaphy suture. There was no statistically significant relationship between blood loss and the use or non-use of bolster.
Pathological results: Clear cell carcinoma accounted for 64% of the cases, whereas papillary carcinoma accounted for 20%; 16% of the cases were stain-resistant carcinoma. Regarding postoperative examination, at 3–6 months and 3 years, 80% and 24% of cases, respectively, had no urinary leakage, infection, or trocar hernia. No patient had signs of metastasis. Surgical complications: One case of intraoperative bleeding required conversion to LRN; approximately 250 ml of blood was lost in this case, and it required a long surgical time (120 minutes). Wolf advised surgeons to assess whether the tumor is exophytic/endophytic based on imaging diagnosis and assess the nearness to the collecting system or sinus and the location of the tumor before surgery; during surgery, endoscopic ultrasound should be used to assess the relationship of the tumor. They must also know how to use combined hemostatic means and techniques [3].
There were two postoperative complications: one case still had retroperitoneal fluid due to obstruction of surgical drainage. After, aspiration was performed under ultrasound guidance. The second case was that of bleeding five days after discharge from the hospital, with signs of hematuria and hematoma around the kidney. The patient returned for blood transfusion, embolization, and laparoscopy.
The limitations of this study are that the sample size was not large enough and the oncological follow-up was not long. For better results, it is necessary to use more advanced research designs.
Conclusion
Our study shows that rLPN in the treatment of large renal tumors (pT1b) is a minimally invasive, safe, effective surgery with few major complications during surgery, it can be performed at provincial hospitals and hospitals equivalent to provincial or regional hospitals. To perform this surgery well, the treatment facility needs to have adequate equipment and surgeons with experience in laparoscopic surgery and specialized training. Studies with a larger number of cases are warranted for an overview.
Conflicts of interest
Authors declare no conflicts of interest.
References
[1] Guillonneau B, Gill IS, Janetschek G, Tuerk IA. Partial nephrectomy. In: Laparoscopic Techniques in Uro-Oncology. London: Springer-Verlag; 2009. p.71–83.
[2] Dominguez-Escrig JL, Vasdev N, O'Riordon A, Soomro N. Laparoscopic partial nephrectomy: technical considerations and an update. J Minim Access Surg. 2011; 7:205–221.
[3] Wolf JS. Laparoscopic partial nephrectomy. In: Nakada SY, Hedican SP, editors. Essential Urologic Laparoscopy. Totowa, NJ: Humana; 2010. p.165–84.
[4] Ljungberg B, Albiges L, Abu-Ghanem Y. European Association of Urology Guidelines on renal cell carcinoma: The 2022 update. Eur Urol. 2022; 82:399–410.
[5] Kutikov A, Uzzo RG. The R.E.N.A.L. nephrometry score: a comprehensive standardized system for quantitating renal tumor size, location, and depth. J Urol. 2009; 182:844–853.
[6] Rendon RA, Kapoor A, Breau R, Leveridge M, Feifer A, et al. Surgical management of renal cell carcinoma: Canadian Kidney Cancer Forum Consensus. Can Urol Assoc J. 2014; 8:E398–E412.
[7] Nadu A, Mor Y, Laufer M, Winkler H, Kleinmann N, et al. Laparoscopic partial nephrectomy: single center experience with 140 patients-evolution of the surgical technique and its impact on patient outcomes. J Urol. 2007; 178:435–439.
[8] Shikanov S, Lifshitz D, Chan AA. Impact of ischemia on renal function after laparoscopic partial nephrectomy: A multicenter study. J Urol. 2010; 183:1714–1718.
[9] Sinh TN, Thuan CQ, Vu DQ. Surgery retroperitoneal laparoscopic partial nephrectomy for renal tumors: A harmonious solution in oncology. Ho Chi Minh City J Med. 2010; 14:275.
[10] Yin X, Jiang S, Shao Z. Kidney ventrally rotation technique in retroperitoneal robot-assisted partial nephrectomy for posterior hilar tumor: technical considerations and preliminary results. World J Surg Oncol. 2020; 18:1–7.