Full Text
Introduction
Breast lesions encompass a wide spectrum of pathological entities with significant clinical and morphological heterogeneity [1]. They are among the most frequently encountered conditions in women, often requiring prompt histopathological evaluation supported by immunohistochemistry (IHC) [2]. Globally, breast cancer accounts for approximately 22% of all female malignancies, making it the most common cancer among women [2,3]. Ductal carcinomas, believed to originate from the terminal duct–lobular units, represent the predominant histological subtype [4].
The glandular architecture of the breast consists of an inner layer of luminal epithelial cells surrounded by an outer layer of myoepithelial cells (MECs). These MECs form a crucial component of the terminal duct–lobular unit, providing both structural and functional support. Although they can sometimes be identified on routine hematoxylin and eosin (H&E) sections, immunohistochemical staining is often required for their reliable visualization [5,6]. Recognition of an intact myoepithelial cell layer is diagnostically essential to differentiate benign and proliferative lesions, as well as in situ carcinomas, from invasive breast malignancies [7].
Among various myoepithelial markers, p63 a nuclear transcription factor belonging to the p53 gene family is highly specific for the myoepithelial cells of the normal breast [6]. It is also expressed in the basal and epithelial cells of several other tissues, including the skin, esophagus, ectocervix, urinary bladder, salivary glands, prostate, and bronchi [5,6]. Unlike other markers such as smooth muscle actin (SMA), calponin, caldesmon, smooth muscle myosin heavy chain (SMMHC), cytokeratin 5/6, and CD10, p63 shows exclusive nuclear staining and does not cross-react with stromal myofibroblasts, vascular smooth muscle, or adipocytes [7,8]. Because of this high specificity, p63 serves as a superior marker for identifying myoepithelial cells and can be incorporated into immunohistochemical panels to resolve diagnostically challenging breast lesions [9].
In normal breast tissue, p63 expression appears as continuous, intense nuclear staining [10]. Benign non-proliferative lesions retain continuous positivity, proliferative lesions exhibit discontinuous or patchy positivity, and in situ carcinomas demonstrate only focal expression [11,12]. Invasive carcinomas, characterized by the absence of the myoepithelial cell layer, show complete loss of p63 staining [13,14]. Therefore, p63 expression provides a valuable diagnostic clue in distinguishing benign, proliferative, in situ, and invasive breast lesions [15,16].
This study aims to evaluate the pattern of p63 immunostaining in benign, in situ, and invasive breast lesions, and to assess its diagnostic significance in differentiating non-invasive from invasive breast carcinomas.
Materials and methods
This study was conducted on 53 breast tissue specimens received in the Histopathology Unit, Department of Pathology, Belagavi Institute of Medical Sciences and Hospital, between November 2020 and May 2022, irrespective of the patient’s age and gender. Relevant clinical data, including patient history and examination findings, were obtained for all cases. The study was approved by the Institutional Ethics Committee.
All specimens were fixed in 10% neutral buffered formalin, routinely processed, and stained with H&E for detailed histopathological evaluation. Unstained paraffin sections were subsequently subjected to immunohistochemical staining using the p63 antibody, employing the standard non-biotin polymerized horseradish peroxidase (HRP) method. Normal breast tissue was used as a positive control to validate the staining results. The study included patients of all age groups and both sexes, and the sample types comprised core needle biopsies, trucut biopsies, lumpectomy, and mastectomy specimens.
Immunohistochemical analysis for p63 was performed on formalin-fixed, paraffin-embedded tissue sections using the avidin–biotin peroxidase complex (ABC) method. Appropriate positive and negative controls were included in each staining run to ensure consistency and reproducibility. Sections were incubated with monoclonal anti-p63 antibody, followed by visualization using a diaminobenzidine (DAB) chromogen. Hematoxylin was applied as a counterstain. Only distinct nuclear staining was considered positive for p63 expression, while any cytoplasmic staining was disregarded.
Evaluation of p63 immunoreactivity was performed by assessing the nuclear staining pattern of myoepithelial cells at the periphery of ducts and lobules in benign and premalignant lesions, and around tumor nests in malignant lesions. The staining pattern was classified according to continuity and intensity, following the scoring criteria described by Verma et al. [10]. Cases demonstrating continuous strong nuclear staining (score 3) indicated a complete circumferential myoepithelial layer, whereas less continuous or patchy nuclear staining (score 2) reflected partial preservation. Discontinuous or focal nuclear staining (score 1) represented focally disrupted myoepithelial layers, and negative staining (score 0) indicated a complete absence of myoepithelial cells.
Each case was independently evaluated by two pathologists to minimize observer bias. The p63 expression pattern and corresponding scores were then correlated with the histopathological diagnosis to determine the diagnostic significance of p63 in differentiating benign, in situ, and invasive breast lesions.
Results
A total of 53 breast lesion cases were analyzed in the present study. Among these, 30 cases (56.6%) were benign, 5 cases (9.4%) were premalignant, and 18 cases (34.0%) were malignant.
The benign category included 17 cases of fibroadenoma (Figure 1a), 6 cases of fibrocystic disease, 3 cases of usual ductal hyperplasia, 2 cases of benign phyllodes tumor, 1 case of tubular adenoma, and 1 case of benign papilloma.
The premalignant group comprised 5 cases of ductal carcinoma in situ (DCIS), whereas the malignant group consisted of 15 cases of invasive ductal carcinoma – not otherwise specified (IDC-NOS) (Figure 1b), and 3 cases of papillary carcinoma. The distribution of various breast lesions is summarized in Table 1.
Table 1: Distribution of different breast lesions.
|
Diagnosis
|
No of cases
|
Percentage (%)
|
|
Benign
|
|
Fibroadenoma
|
17
|
32.07%
|
|
Fibrocystic disease
|
06
|
11.32%
|
|
Benign phyllodes tumor
|
02
|
3.77%
|
|
Tubular adenoma
|
01
|
1.88%
|
|
Usual ductal hyperplasia
|
03
|
5.66%
|
|
Benign papilloma
|
01
|
1.88%
|
|
Premalignant
|
|
Ductal carcinoma insitu
|
05
|
9.44%
|
|
Malignant
|
|
Infiltrating ductal carcinoma, not otherwise specified
|
15
|
28.30%
|
|
Papillary carcinoma
|
03
|
5.66%
|
|
Total
|
53
|
100%
|
Age- and size-wise distribution
The age of patients in the present study ranged from 14 to 75 years. The majority of cases, 21 (39.6%), were observed in the 31–50-year age group, indicating that breast lesions are most prevalent during the reproductive and perimenopausal periods.
With respect to tumor size, most lesions measured between 2 and 5 cm, accounting for 38 cases (71.7%). The mean size of benign tumors was 3.5 cm, whereas malignant tumors demonstrated a larger mean size of 7.5 cm (Table 2).
Table 2: Distribution of cases based on the size of the lump (n = 53).
|
Size (cm)
|
Benign
|
Malignant
|
Total
|
|
No
|
%
|
No
|
%
|
No
|
%
|
|
< 2cm
|
01
|
1.89%
|
-
|
-
|
01
|
1.89%
|
|
2-5 cm
|
30
|
56.60%
|
08
|
15.09%
|
38
|
71.69 %
|
|
> 5 cm
|
02
|
3.77%
|
12
|
22.65%
|
14
|
26.42%
|
|
Total
|
33
|
62.26%
|
20
|
37.74%
|
53
|
100%
|
p63 expression
In the present study, a total of 53 breast lesions were evaluated for p63 immunohistochemical expression. Among these, 30 cases (56.6%) were benign, 5 cases (9.4%) were premalignant, and 18 cases (33.9%) were malignant (Table 3).
All benign lesions demonstrated nuclear positivity for p63, whereas all malignant cases were completely negative. The premalignant lesions, represented by ductal carcinoma in situ (DCIS), exhibited focal or discontinuous nuclear staining, corresponding to the lowest level of p63 expression among the positive categories.
Within the benign group, fibroadenoma was the most frequent lesion and showed continuous, intense nuclear staining with a score of 3 (Figure 2A). Benign papilloma also demonstrated a similar pattern of continuous p63 nuclear positivity (score 3). Other benign lesions, including fibrocystic disease, usual ductal hyperplasia, and tubular adenoma (Figure 2B), exhibited less continuous nuclear staining with a score of 2.
Premalignant lesions such as ductal carcinoma in situ displayed focal or discontinuous nuclear staining with a score of 1 (Figure 3). In contrast, all malignant lesions, including invasive ductal carcinoma (Figure 4) and papillary carcinoma, showed a complete absence of p63 expression.
Table 3: p63 scoring in different spectrum of breast lesions.
|
Type of breast lesions
|
No of cases
|
p63 scoring
|
|
Score-0
|
Score-1
|
Score-2
|
Score-3
|
|
Benign
|
|
Fibroadenoma
|
17
|
|
|
|
✓
|
|
Fibrocystic disease
|
06
|
|
|
✓
|
|
|
Benign phyllodes tumor
|
02
|
|
✓
|
|
|
|
Tubular adenoma
|
01
|
|
|
✓
|
|
|
Usual ductal hyperplasia
|
03
|
|
|
✓
|
|
|
Benign papilloma
|
01
|
|
|
|
✓
|
|
Premalignant
|
|
Ductal carcinoma in situ
|
05
|
|
✓
|
|
|
|
Malignant
|
|
Invasive ductal carcinoma, not otherwise specified
|
15
|
✓
|
|
|
|
|
Papillary carcinoma of breast
|
03
|
✓
|
|
|
|
|
Total
|
53
|
|
|
|
|
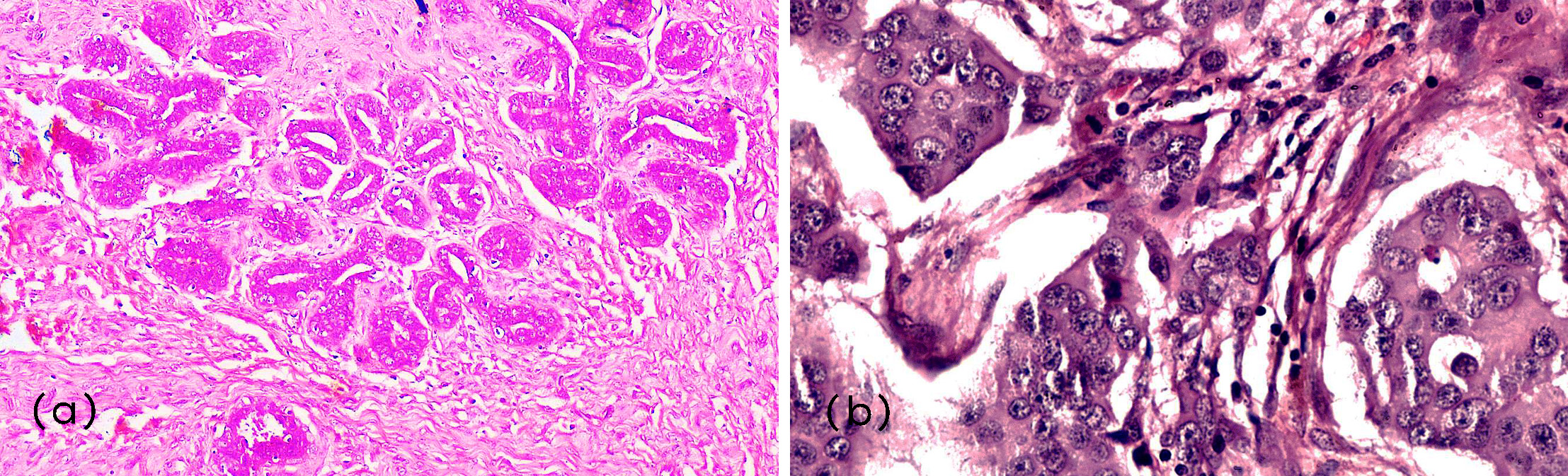
Figure 1: Microphotograph of: (a) Fibroadenoma (H&E, 200x); (b) Invasive papillary carcinoma (H&E, 400x).
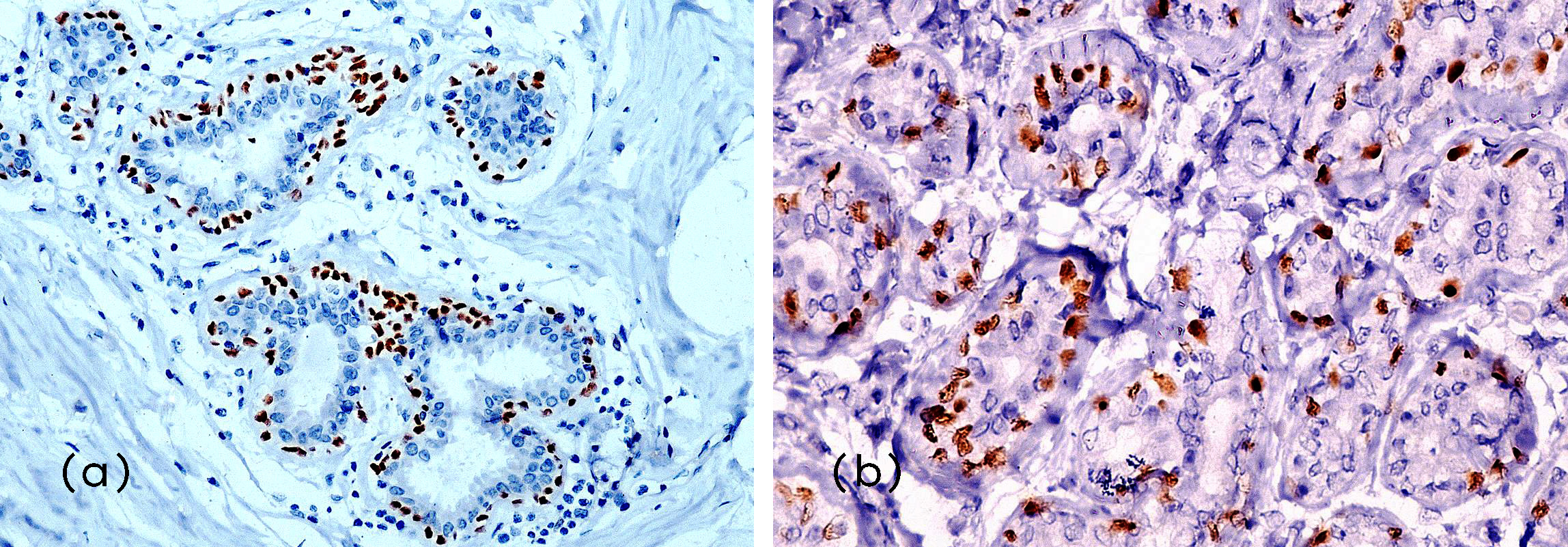
Figure 2: Microphotograph of: IHC: (a) p63 expression in Fibroadenoma (Score-3) (200x), (b) p63 expression in Tubular adenoma (Score-2) (400x).
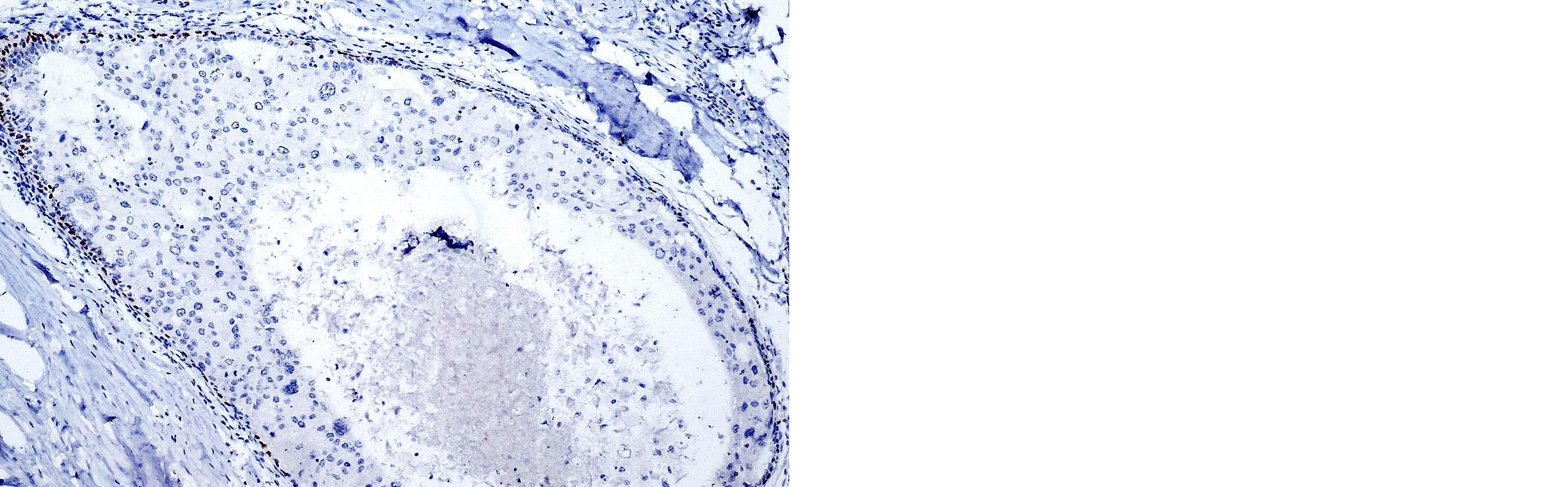
Figure 3: Microphotograph of IHC - p63 expression in Ductal carcinoma in situ (Score – 1) (200x).
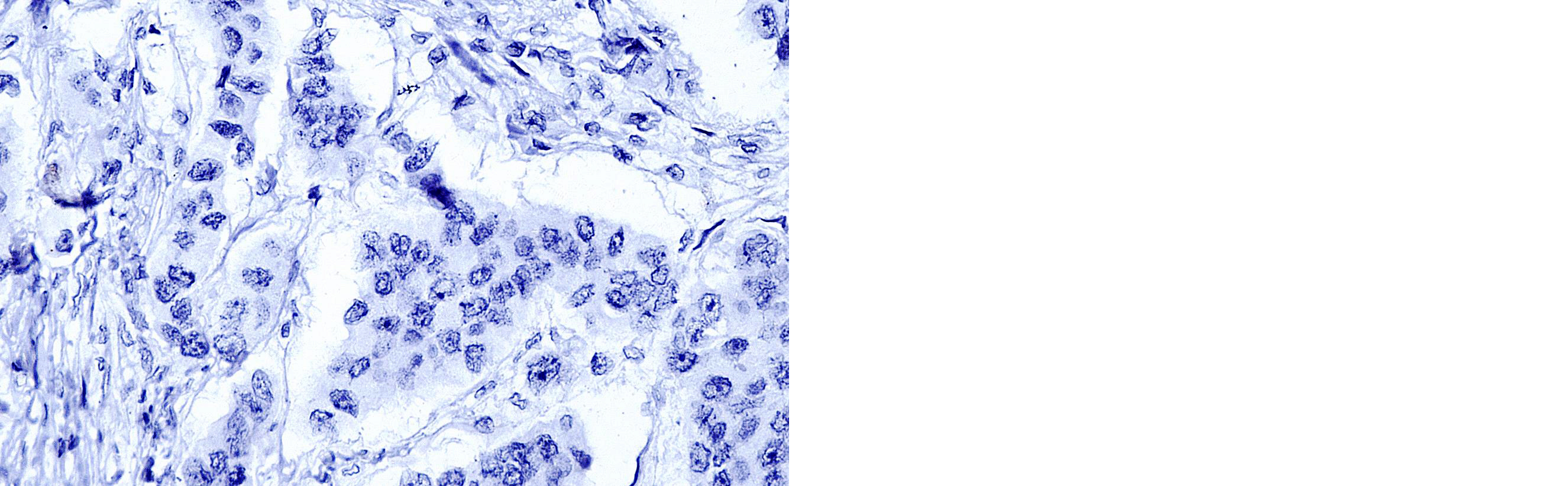
Figure 4: Microphotograph of IHC - p63 expression in Invasive ductal carcinoma (NOS) (Score – 0) (400x).
Discussion
In the present study comprising 53 breast lesions, benign cases formed the majority (56.6%), followed by malignant (33.9%) and premalignant lesions. This distribution closely resembles the findings of Verma et al. [1], who reported 67.6% benign and 32.4% malignant lesions. Similarly, Stefanou et al. [7] observed 52.63% benign and 36.09% malignant cases, whereas Werling et al. [10] reported a lower proportion of benign lesions (12.8%) and a higher incidence of malignant cases (41.17%). These inter-study variations may be attributed to differences in sample size, study population, and inclusion criteria.
Among the benign lesions, fibroadenoma was the most frequent (49.3%), which is consistent with the majority of published series, where fibroadenoma accounts for approximately 46–68% of benign breast lumps [1,7]. Other benign lesions encountered included fibrocystic disease, usual ductal hyperplasia, benign phyllodes tumor, and papilloma. The predominance of fibroadenoma in younger women reflects the hormonal responsiveness of breast tissue during the reproductive years, particularly to estrogen and progesterone.
In the malignant category, infiltrating ductal carcinoma–not otherwise specified (IDC-NOS) was the most common histological subtype (28.3%), consistent with the findings of Verma et al. [1] (27.5%) and Stefanou et al. [7] (23.3%). IDC-NOS represents the most frequent invasive carcinoma worldwide, demonstrating a broad spectrum of morphologic appearances and prognostic behaviors. Papillary carcinoma was less frequent (5.6%), similar to the low prevalence reported by Werling et al. [10] and Moraes Schenka et al. [15], who described papillary carcinoma as an uncommon histologic variant.
The age of patients ranged from 14 to 75 years, with the highest incidence (39.6%) in the 31–50-year age group. This observation corresponds with the studies of Verma et al. [1], Stefanou et al. [7], and Werling et al. [10], who also documented peak occurrence in the fourth and fifth decades. The increased prevalence of breast lesions during this period can be attributed to cyclical hormonal influences, lactational changes, and cumulative genetic mutations.
Tumor size in the present study varied from 0.5 cm to 8 cm, with most lesions (71.7%) measuring between 2–5 cm. Only 3.7% of benign lesions exceeded 5 cm, while 22.6% of malignant cases were larger than 5 cm. These findings align with Verma et al. [1], who reported that lesions larger than 2 cm were more likely to be malignant. Tumor size remains an important prognostic indicator in breast carcinoma and often correlates with advanced stage and lymph node involvement.
p63 expression
Immunohistochemical analysis revealed that all benign and premalignant lesions were positive for p63 expression, whereas all malignant lesions were negative. This distinct difference underscores the diagnostic value of p63 as a specific marker of myoepithelial cells. In benign lesions such as fibroadenoma and papilloma, p63 showed continuous, strong nuclear positivity (score 3), representing an intact myoepithelial layer. Lesions like fibrocystic disease, usual ductal hyperplasia, and tubular adenoma displayed less continuous nuclear staining (score 2). Premalignant lesions, particularly ductal carcinoma in situ (DCIS), demonstrated weak or discontinuous nuclear staining (score 1), consistent with partial myoepithelial attenuation. In contrast, all invasive carcinomas, including IDC-NOS and papillary carcinoma, completely lacked p63 expression.
Our findings are comparable to those of Barbareschi et al. [5], who studied 384 breast samples and reported consistent p63 positivity in benign lesions and complete absence in invasive carcinomas. Similarly, Wang et al. [6] demonstrated that p63 expression was confined to myoepithelial nuclei in normal ducts, reduced in ductal hyperplasia and carcinoma in situ, and entirely lost in invasive carcinoma. Stefanou et al. [7] and Werling et al. [10] also confirmed that p63 is a reliable nuclear marker useful for distinguishing in situ from invasive lesions.
The diagnostic role of p63 is supported by its nuclear localization and absence of cross-reactivity with stromal myofibroblasts, vascular smooth muscle cells, or adipocytes [8,9]. Other markers such as SMA, calponin, or CD10 may stain non-myoepithelial components, potentially leading to diagnostic ambiguity. Hence, p63 is often incorporated into immunohistochemical panels for difficult or borderline cases [3,4]. Loss of p63 expression in invasive carcinomas corresponds to the disruption of the myoepithelial layer and basement membrane, key morphological features of invasion [2,19,20]. Koker and Kleer [11] also emphasized the high specificity of p63 for identifying metaplastic carcinomas, further underscoring its diagnostic relevance.
Recent literature continues to substantiate the diagnostic and biological importance of p63 in breast pathology. Gatti et al. [26] emphasized that p63 plays a pivotal role “at the crossroads between stemness and metastasis,” linking its expression to tumor differentiation and progression. Saini et al. [28] and Aden et al. [27] demonstrated consistent nuclear p63 positivity in benign and in situ lesions, with complete loss in invasive carcinomas, reaffirming its reliability in confirming myoepithelial integrity. Rajitha et al. [30] observed a significant association between p63 loss and higher tumor grade, suggesting potential prognostic relevance. Similarly, Velamala et al. [29] reported strong p63 expression in benign and borderline lesions among South Indian patients, underscoring its diagnostic reproducibility across populations.
Collectively, these findings corroborate the present study’s results and highlight p63 as a sensitive, specific, and reproducible immunomarker, useful not only for differentiating invasive from non-invasive lesions but also as a possible indicator of tumor behavior.
Overall, the present study reinforces the role of p63 as a highly specific nuclear myoepithelial marker that aids in distinguishing benign and preinvasive lesions from invasive carcinomas. Incorporation of p63 immunostaining into the histopathological evaluation enhances diagnostic confidence, particularly in cases with overlapping morphology or limited biopsy material.
Conclusion
The present study underscores the diagnostic value of p63 immunostaining in differentiating the full spectrum of breast lesions. All benign and premalignant lesions demonstrated nuclear p63 positivity, whereas all malignant lesions were completely negative, reaffirming that the loss of the myoepithelial layer is a hallmark of invasion. A clear correlation was observed between histomorphological patterns and p63 expression intensity—continuous staining in benign lesions, discontinuous staining in premalignant lesions, and complete absence in invasive carcinomas. No significant association was found between p63 expression and clinicopathological parameters such as patient age, tumor size, histologic grade, or lymph node status. Overall, p63 proves to be a sensitive and specific nuclear myoepithelial marker that enhances diagnostic confidence in distinguishing benign, in situ, and invasive breast lesions. Its routine inclusion in immunohistochemical evaluation aids accurate diagnosis and supports appropriate clinical management.
Conflicts of interest
There are no conflict of interest.
References
[1] Verma N, Sharma B, Singh P, Sharma SP, Rathi M, et al. Role of p63 expression in non-proliferative and proliferative lesions of breast. Int J Res Med Sci. 2018; 6:2705–2710.
[2] Silva AR, Ramalho LNZ, Garcia SB, Zucoloto S. Is p63 reliable in detecting microinvasion in ductal carcinoma in situ of the breast? Pathol Oncol Res. 2003; 9:20–23.
[3] Khazai L, Rosa M. Use of immunohistochemical stains in epithelial lesions of the breast. Cancer Control. 2015; 22:220–225.
[4] Corben AD, Lerwill MF. Use of myoepithelial cell markers in the differential diagnosis of benign, in situ and invasive lesions of the breast. Int J Surg Pathol. 2009; 17:351–373.
[5] Barbareschi M, Pecciarini L, Cangi MG. p63, a p53 homologue, is a selective nuclear marker of myoepithelial cells of the human breast. Am J Surg Pathol. 2001; 25:1054–1060.
[6] Wang T, Zhao W, Li J, Chen Y, Zhang H. p63 expression in normal, hyperplastic and malignant breast tissue. Breast Cancer. 2002; 9:216–219.
[7] Stefanou D, Batistatou A, Nonni A, Arkoumani E, Agnantis NJ. p63 expression in benign and malignant breast lesions. Histol Histopathol. 2004;19:465–471.
[8] Zaha DC. Significance of immunohistochemistry in breast cancer. World J Clin Oncol. 2014; 5(3):382–392.
[9] Abdallah DM, El Deeb NMF. Comparative immunohistochemical study of p63, SMA, CD10 and calponin in distinguishing in situ from invasive breast carcinoma. J Mol Biomark Diagn. 2017; 8:2155–9929.
[10] Werling RW, Hwang H, Yaziji H, Gown AM. Immunohistochemical distinction of invasive from non-invasive breast lesions: a comparative study of p63 versus calponin and smooth muscle myosin heavy chain. Am J Surg Pathol. 2003; 27:82–89.
[11] Koker MM, Kleer CG. p63 expression in breast cancer – a highly sensitive and specific marker of metaplastic carcinoma. Am J Surg Pathol. 2004; 28:1506–1512.
[12] Ribeiro A, Ramalho LN, Garcia SB, Brandao DF, Chahud F, et al. p63 correlates with both BRCA1 and cytokeratin 5 in invasive breast carcinomas: further evidence for the pathogenesis of the basal phenotype of breast cancer. Histopathology. 2005; 47:458–466.
[13] Silva AR, Ramalho LNZ, Garcia SB, Zucoloto S. The relationship between p63 and p53 expression in normal and neoplastic breast tissue. Arch Pathol Lab Med. 2003; 127:336–340.
[14] Reisenbichler ES, Ross JR, Hameed O. The clinical use of a p63/cytokeratin7/18/cytokeratin5/14 antibody cocktail in diagnostic breast pathology. Ann Diagn Pathol. 2014; 18:313–318.
[15] Moraes Schenka NG, Schenka AA, De Souza Queiroz L, De Almeida Matsura M, Vassallo J, et al. Use of p63 and CD10 in the differential diagnosis of papillary neoplasms of the breast. Breast J. 2008; 14:68–75.
[16] Di Como CJ, Urist MJ, Babayan I. p63 expression in normal and tumor tissues. Clin Cancer Res. 2002; 8:494–501.
[17] Kaufmann O, Fietze E, Mengs J. Value of p63 and cytokeratin 5/6 as immunohistochemical markers for the differential diagnosis of poorly differentiated and undifferentiated carcinomas. Am J Clin Pathol. 2001;116:823–30.
[18] Russell A, Singh N, Roberts T. Myoepithelial cell differentiation markers in ductal carcinoma in situ progression. Am J Pathol. 2015; 185:3076–3089.
[19] Moriya T, Kozuka Y, Kanomata N, Tse GM, Tan PH. The role of immunohistochemistry in the differential diagnosis of breast lesions. Pathology. 2009; 41:68–71.
[20] Gottlieb C, Raju U, Greenwald KA. Myoepithelial cells in the differential diagnosis of complex benign and malignant breast lesions: an immunohistochemical study. Mod Pathol. 1990; 3:135–140.
[21] Liu H. Application of immunohistochemistry in breast pathology: a review and update. Am J Clin Pathol. 2014; 138:1629–1642.
[22] Zhao L, Yang X, Khan A. Diagnostic role of immunohistochemistry in the evaluation of breast pathology specimens. Arch Pathol Lab Med. 2014;138:16–24.
[23] Shekhar MP, Kato I, Nangia-Makker P, Tait L. Comedo-DCIS is a precursor lesion for basal-like breast carcinoma: identification of a novel p63/Her2/neu expressing subgroup. Oncotarget. 2013; 4:231–241.
[24] Gusterson BA, Warburton MJ, Mitchell D. Distribution of myoepithelial cells and basement membrane proteins in the normal breast and in benign and malignant breast diseases. Cancer Res. 1982; 42:4763–4770.
[25] Shamloula MM, Shorbagy SH, Saied EME. p63 and cytokeratin 8/18 expression in breast, atypical ductal hyperplasia, ductal carcinoma in situ and invasive duct carcinoma. J Egypt Natl Cancer Inst. 2007; 19:202–210.
[26] Gatti V, Bongiorno-Borbone L, Fierro C, Annicchiarico-Petruzzelli M, Melino G, Peschiaroli A. p63 at the crossroads between stemness and metastasis in breast cancer. Int J Mol Sci. 2019; 20:2683.
[27] Aden D, Afroze N, Sufian Z, Khan AI, Khan T, et al. The role of p63 myoepithelial cell marker in papillary lesions of the breast. Int J Clin Diagn Pathol. 2022; 5:72–76.
[28] Saini A, Saluja SK, Garg MK, Agarwal D, Kulhria A, et al. Immunohistochemical expression of p63 in benign and malignant breast lesions. J Clin Diagn Res. 2021; 15:PC01–PC06.
[29] Velamala P, Dogga SK, Tandyala N, Dhavala MMR, Pappala P, et al. A study of p63 immunohistochemical expression in benign and malignant breast lesions at a tertiary hospital in South India. Cureus. 2024; 17:235.
[30] Rajitha J, Gangadhar R, Sekhar PI, Kasturi S. Role of p63 in benign and malignant lesions of breast. Indian J Public Health Res Dev. 2024; 15:181–186.