Full Text
Introduction
Breast carcinoma poses an escalating global health challenge, with developing nations bearing a disproportionate and rising burden. In India, breast cancer rates have reached alarming proportions, surpassing cervical cancer as the leading cause of cancer-related mortality among women [1]. This shift represents a critical public health concern. Early diagnosis is essential for reducing mortality and morbidity in breast cancer [2].
Multiple preoperative diagnostic modalities are utilized for timely and accurate breast cancer detection. As a key component of triple assessment, fine-needle aspiration cytology (FNAC) offers rapid results with high sensitivity, aiding in categorizing breast lesions into benign and malignant, while remaining cost-effective and minimally invasive [3,4,5]. While histopathology remains the gold standard for diagnosis, cyto-histopathological correlation further enhances diagnostic accuracy and clinical decision-making. However, considerable cytomorphological overlap between benign and malignant lesions poses interpretative challenges, making absolute differentiation difficult in certain cases.
To address these inherent diagnostic limitations, the International Academy of Cytology (IAC) introduced the Yokohama System for Reporting Breast Cytopathology in 2016, a standardized five-tier classification for breast FNAC reporting [6]. Each category carries recommended management options and predetermined risk of malignancy (ROM) values [4]. By standardizing reporting terminology, this system improves interpretative consistency, enhances communication between clinicians and pathologists, and increases diagnostic reproducibility [4].
The five categories of the IAC Yokohama System for Reporting Breast Cytopathology [4] include: Category 1: Insufficient; Category 2: Benign; Category 3: Atypical; Category 4: Suspicious for malignancy; Category 5: Malignant.
In this study, we aim to categorize breast lesions using the IAC Yokohama System on FNAC samples and correlate cytological findings with histopathological outcomes, thereby assessing the diagnostic accuracy and clinical relevance of this reporting system in our setting.
Materials and methods
A hospital-based retrospective study was conducted in the Department of Pathology, Diphu Medical College and Hospital, Karbi Anglong, Assam, Northeast India after obtaining the Ethical Committee approval. The study included patients referred for routine FNAC of palpable breast lumps over a period of 5.5 years, from January 2020 to June 2025. A total of 443 FNAC procedures corresponding to 441 patients were analyzed. In cases of bilateral lesions, each side was evaluated independently if classified into different Yokohama categories.
All male and female patients who underwent FNAC (including both blind and image-guided procedures) during the study period were included. Patients with a previous diagnosis of breast cancer or with a history of chemotherapy, radiotherapy, or hormonal therapy were excluded. Cases in which informed consent was denied were also excluded from the study.
Relevant clinical information, including age, involved quadrant, size of the lesion, duration, and associated symptoms, were recorded. FNAC was performed following standard aseptic precautions after obtaining informed consent. Palpation-guided aspiration was done initially; in cases where smears were inadequate, a repeat aspiration was performed under ultrasound guidance. Smears were air-dried and stained using May–Grünwald–Giemsa (MGG). Cytomorphological evaluation was independently performed by three cytopathologists, and a final diagnosis was assigned only after consensus. All cases were categorized according to the IAC Yokohama System for Reporting Breast Cytopathology.
Statistical analysis
Statistical analysis was performed using Microsoft Excel Office 2019. Histopathological findings served as the reference standard to calculate the Risk of Malignancy (ROM), sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and diagnostic accuracy. Fisher’s exact test was used to determine statistical significance (p-value). ROM , sensitivity, specificity, PPV, NPV, and diagnostic accuracy were calculated using standard formulas. For cyto-histopathological correlation, statistical analysis was performed using three analytical groups: Group A - only malignant cases were considered positive for malignancy; Group B- both suspicious and malignant cases were considered positive; and Group C- atypical, suspicious, and malignant cases were included as positive.
Results
A total of 443 FNAC procedures were evaluated in 441 patients. Two patients presented with bilateral breast lesions, and since each side belonged to a different Yokohama category, they were analyzed as separate lesions. All cases were reviewed independently by three cytopathologists before assigning a final consensus diagnosis according to the International Academy of Cytology (IAC) Yokohama System for Reporting Breast Cytology (Table 1).
Table 1: Number of cases with assigned Yokohama categories with available Histopathological follow up and diagnosis.
|
Yokohama category & cytology cases
|
Number
|
Histopathology diagnosis
|
|
(number) Benign
|
(number) Malignant
|
|
C1 (Insufficient)
|
|
Insufficient material
|
25
|
0
|
0
|
|
C2 (Benign)
|
|
Fibroadenoma
|
179
|
(92) Fibroadenoma
(3) Tubular Adenoma
(1) Myxoid Fibroadenoma
|
|
|
Fibrocystic changes with Adenosis
|
11
|
(1) Fibroadenosis
|
|
|
Benign Phyllodes
|
3
|
(2) Benign Phyllodes
|
|
|
Lactational changes
|
8
|
0
|
|
|
Usual Ductal Hyperplasia with Fibrocystic changes
|
15
|
(3) Cellular Fibroadenoma
(1) Fibroadenoma with Usual Ductal Hyperplasia
(1) Fibroadenoma with Fibrocystic changes
|
|
|
Fat necrosis
|
4
|
0
|
|
|
Epidermal cyst
|
2
|
0
|
|
|
Accessory breast tissue
|
1
|
0
|
|
|
Galactocele
|
3
|
0
|
|
|
Duct ectasia
|
2
|
0
|
|
|
Simple cyst
|
2
|
0
|
|
|
Lipoma
|
1
|
0
|
|
|
Normal breast with terminal duct lobular unit
|
4
|
0
|
|
|
Lactating adenoma
|
1
|
(1) Lactating Adenoma
|
|
|
Mastitis
|
Acute mastitis
|
9
|
0
|
|
|
Abscess
|
7
|
0
|
|
Lactation associated
|
4
|
0
|
|
Plasma cell mastitis
|
1
|
0
|
|
Non-specific chronic mastitis
|
12
|
0
|
|
Non-caseating granulomatous mastitis
|
1
|
0
|
|
Gynecomastia
|
11
|
(6) Gynecomastia
|
|
|
Benign proliferative lesion
|
25
|
(3) Fibroadenoma
(1) Cellular fibroadenoma
(2) Benign phyllodes
(1) Lobular carcinoma in situ
|
|
|
C3 (Atypical)
|
|
Atypical ductal hyperplasia (ADH)
|
3
|
(1) Atypical ductal hyperplasia (ADH)
|
(1) Ductal carcinoma in situ (DCIS)
|
|
Benign proliferative with atypia
|
8
|
(1) Usual ductal hyperplasia
(1) Fibroadenosis
|
|
|
Fibroadenoma with atypia
|
2
|
(1) Fibroadenoma
|
|
|
Fibroadenoma with Apocrine changes
|
2
|
(1) Fibroadenoma with Apocrine changes
|
|
|
Papillary Lesion
|
1
|
(1) Intraductal papilloma
|
|
|
C4 (Suspicious for malignancy)
|
|
Suspicious for malignancy
|
16
|
(1) Intraductal papilloma
|
(3) Ductal carcinoma in situ (DCIS)
(5) Invasive Ductal carcinoma
|
|
C5 (Malignant)
|
|
Invasive ductal carcinoma
|
78
|
(1) Sclerosing adenosis
|
(52) Invasive ductal carcinoma, no special type;
(6) Invasive ductal carcinoma with comedo-necrosis;
(2) Invasive ductal carcinoma with mucinous differentiation
|
|
Infiltrating lobular carcinoma
|
1
|
0
|
|
|
Malignant phyllodes
|
1
|
0
|
|
|
Total
|
443
|
126
|
69
|
The majority of cases were concentrated in the younger population (n = 237; 53.74%), with an overall median age of 24 years (range: 13–85 years). When only malignant cases were considered, the median age increased to 45 years, and the youngest patient with malignancy was 15 years old, diagnosed with invasive breast carcinoma. A clear female predominance was observed, with 97.73% female patients and 2.27% male patients (Table 2).
Patients presenting only with a vague lumpy sensation without a discrete mass were excluded. All well-defined swellings were individually assessed for size. 61.26% of the lesions measured between 2 and 5 cm (Table 2). Upper outer quadrant was the most commonly involved quadrant (Figure 1). Bilateral breast involvement was noted in 41 cases, among which 2 patients exhibited lesions falling into different Yokohama categories. For analytical consistency, multiple ipsilateral lesions that demonstrated identical cytomorphological features and the same Yokohama category were consolidated and counted as a single case (Table 2).
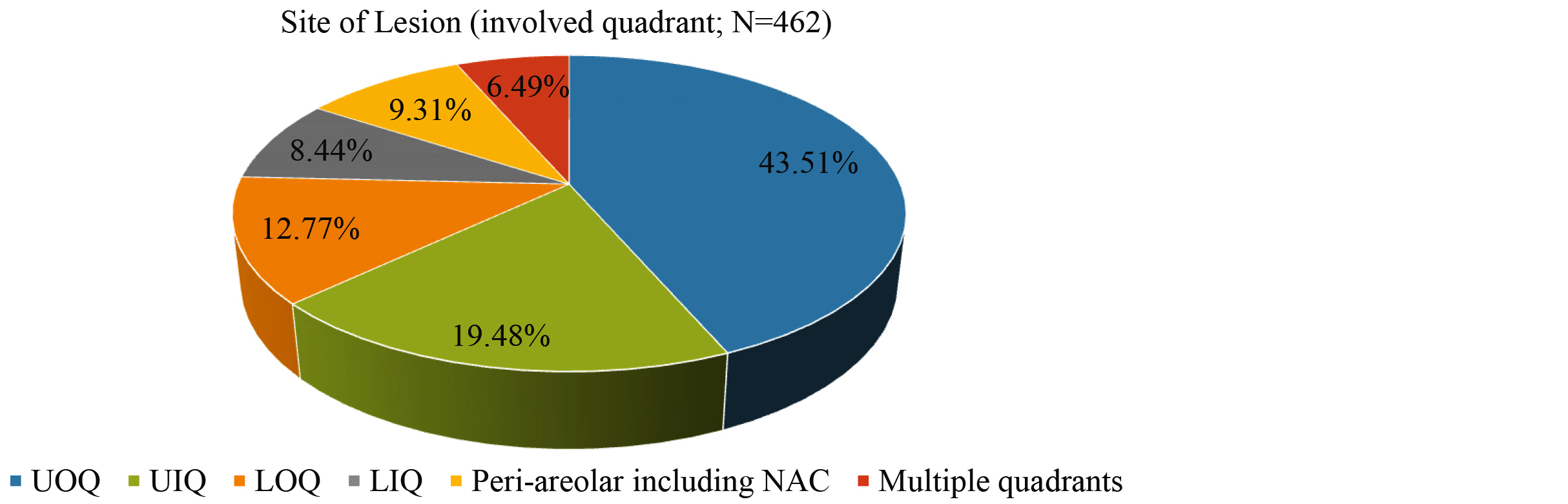
Figure 1: Site of Lesion (involved quadrant of breast; N=462; UOQ= upper outer quadrant, UIQ= upper inner quadrant, LOQ= lower outer quadrant, LIQ= lower inner quadrant, NAC= nipple areolar complex).
Table 2: Clinicopathological parameters of all of the patients in the study group.
|
Clinicopathological parameter
|
No.
|
Percentage (%)
|
|
Age (Years) N=441
|
|
<15
|
2
|
0.45
|
|
15-30
|
237
|
53.74
|
|
31-45
|
137
|
31.06
|
|
46-60
|
53
|
12.02
|
|
>60
|
12
|
2.72
|
|
Gender, N=441
|
|
male
|
10
|
2.27
|
|
female
|
431
|
97.73
|
|
Size of lesion (cms), N= 462
|
|
<2
|
132
|
28.57
|
|
2-5
|
283
|
61.26
|
|
>5
|
47
|
10.17
|
|
Site of lesion (Involved quadrant), N= 462
|
|
Upper Outer Quadrant (UOQ)
|
201
|
43.51
|
|
Upper Inner Quadrant (UIQ)
|
90
|
19.48
|
|
Lower Outer Quadrant (LOQ)
|
59
|
12.77
|
|
Lower Inner Quadrant (LIQ)
|
39
|
8.44
|
|
Central/ Peri-areolar including Nipple Areolar complex (NAC)
|
43
|
9.31
|
|
Multiple quadrants
|
30
|
6.49
|
The distribution of the 443 FNAC cases according to the IAC Yokohama categories is shown in Figure 2. Category 1 comprised 25 cases (5.65%), while Category 2 accounted for the largest proportion with 306 cases (69.07%). Category 3 to 5 included 16 cases (3.61%), 16 cases (3.61%), 80 cases (18.06%) respectively.
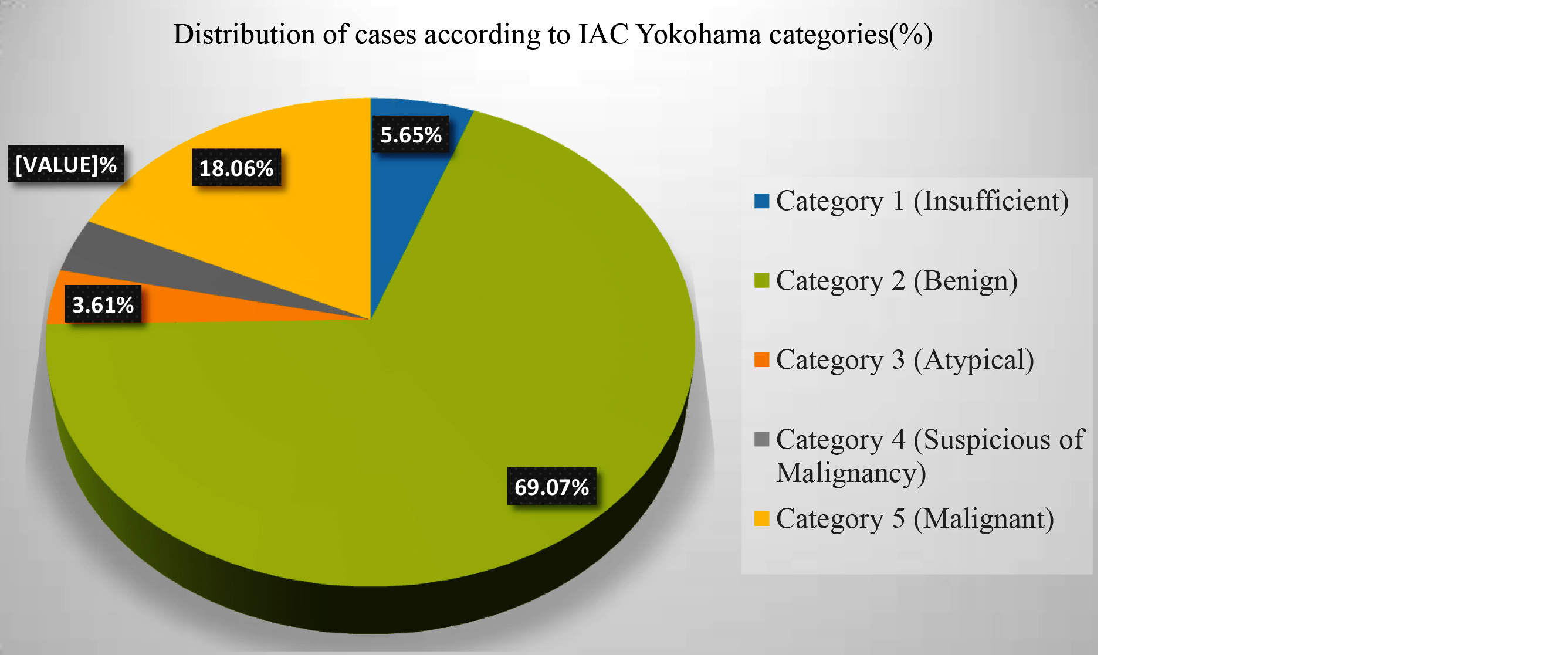
Figure 2: Cytological Categories (IAC Yokohama classification 2016; N=443; 2 bilateral cases with different Yokohama category).
Histopathological correlation was feasible in 195 of the 443 FNAC cases (44.02%). Cyto-histopathological concordance was observed in 193 cases, yielding an accuracy of 98.97% (Figure 3). Two discordant cases were noted—one each from Category 4 and Category 5—which were histopathologically confirmed as benign. The likely reasons for discordance included sampling error, challenging mimickers such as sclerosing adenosis, and inherent cytological limitations, particularly in subclassifying papillary lesions. Additionally, one case categorized as Category 3 was underdiagnosed as benign (ADH) but was subsequently confirmed as ductal carcinoma in situ (DCIS) on histopathology.
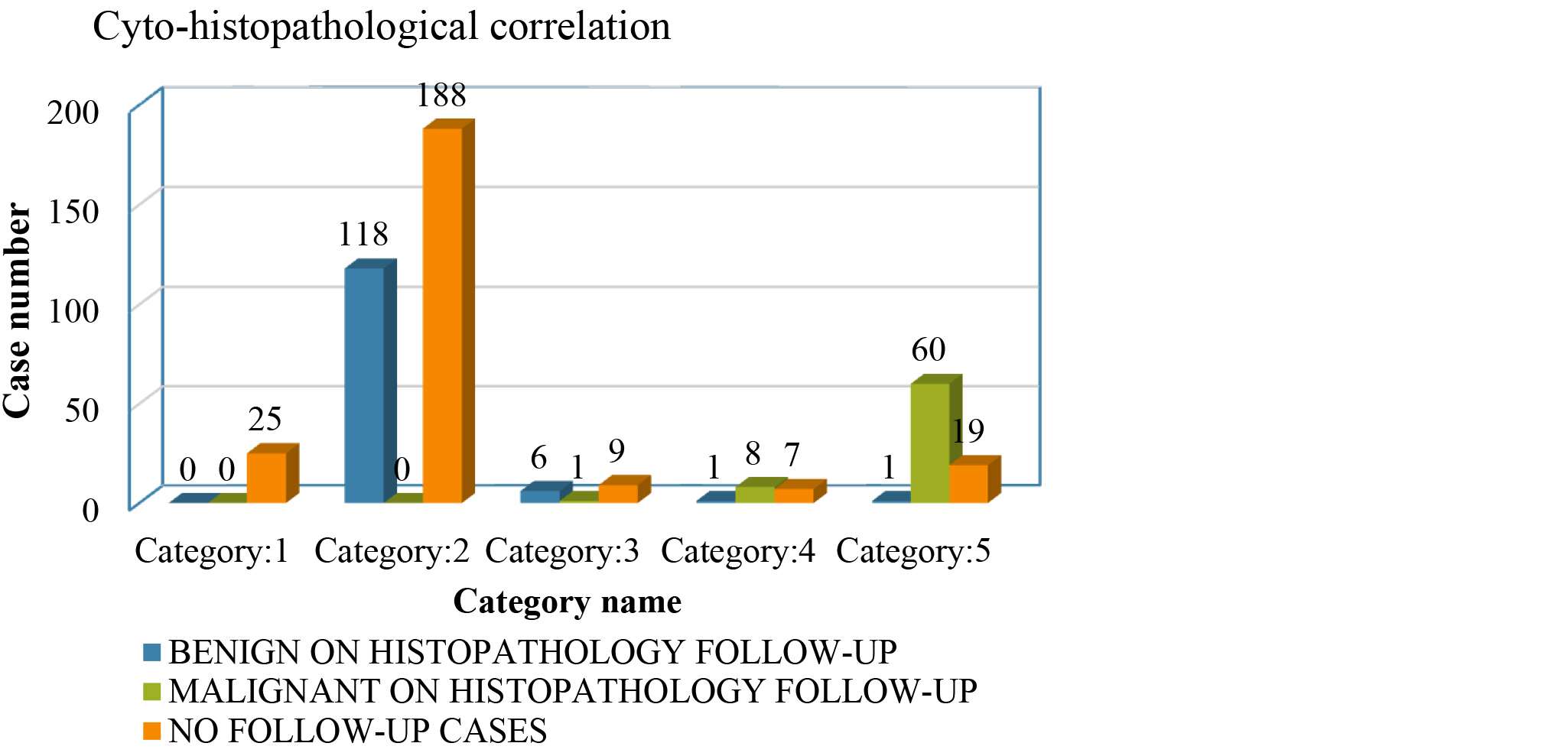
Figure 3: Cytology IAC Yokohama grades with histopathology correlation.
The risk of malignancy (ROM) values for different IAC Yokohama categories and statistical parameters for the three analytical groups are summarized in Table 3 and 4 respectively.
Table 3: ROM with respect to IAC Yokohama cytology categories.
|
IAC Yokohama cytology category
|
Risk of malignancy (ROM)
|
|
Insufficient
|
0%
|
|
Benign
|
0%
|
|
Atypical
|
14.28%
|
|
Suspicious for Malignancy
|
88.89%
|
|
Malignant
|
98.36%
|
Table 4: Sensitivity, specificity, PPV, NPV, diagnostic accuracy of IAC Yokohama system in different analytical groups.
|
Statistical parameters/ Analytical groups
|
Sensitivity
|
Specificity
|
PPV
|
NPV
|
Diagnostic Accuracy
|
|
Group A: Only malignant cases
|
86.96%
|
99.21%
|
98.36%
|
93.28%
|
94.87%
|
|
Group B: Both suspicious and malignant cases
|
98.55%
|
98.41%
|
97.14%
|
99.20%
|
98.46%
|
|
Group C: Atypical, suspicious, and malignant cases
|
100%
|
93.65%
|
89.61%
|
100%
|
95.90%
|
The highest diagnostic accuracy (98.46%) was achieved in analytical Group B, whereas specificity was maximum (99.21%) in analytical Group A. A statistically significant correlation (p < 0.001) was observed between cytological grading using the IAC Yokohama system and the corresponding histopathological outcomes, as determined by Fisher’s Exact test.
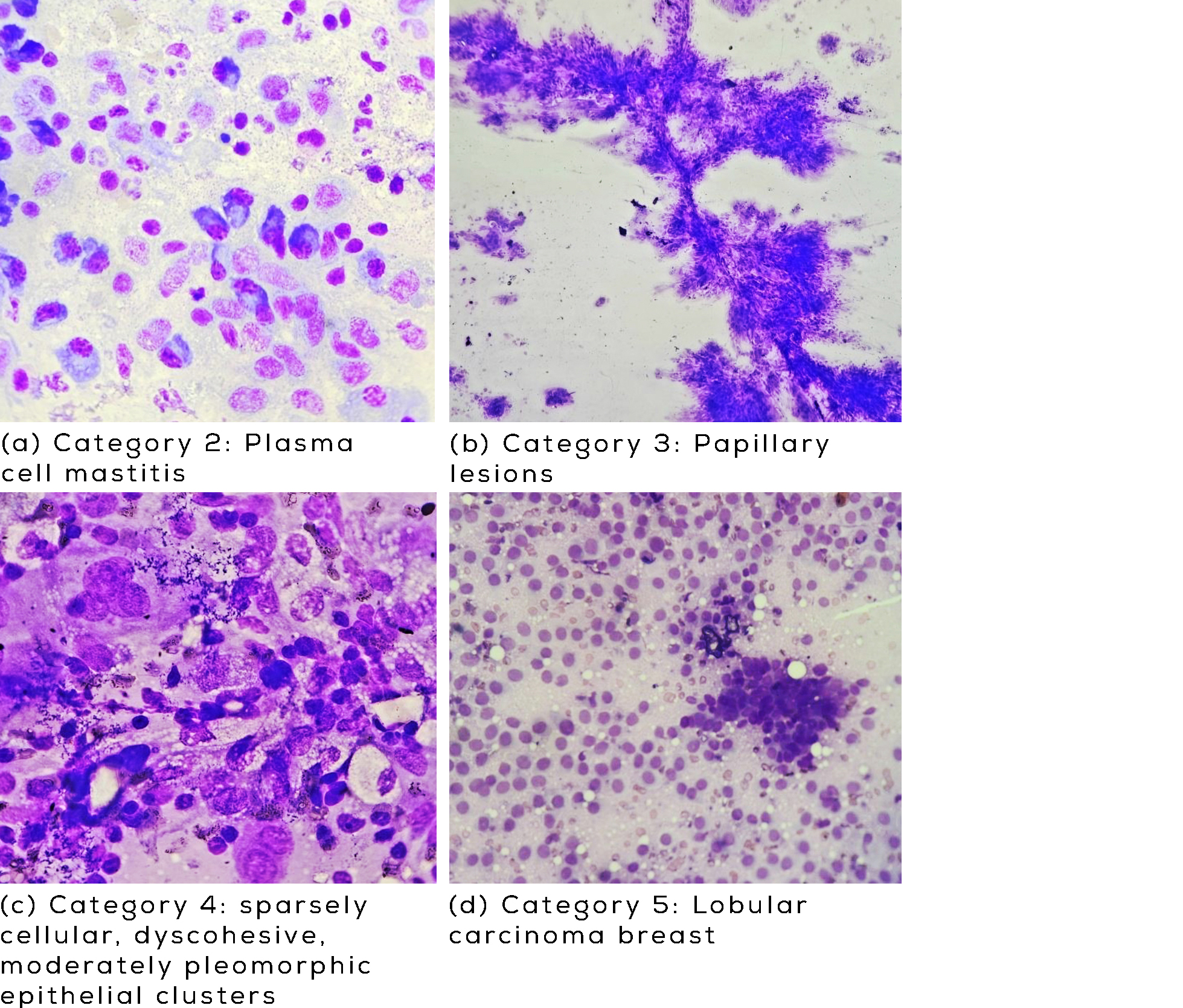
Figure 4a-d: Cytomorphological spectrum of benign, atypical, suspicious, and malignant breast lesion categorized using the IAC Yokohama reporting system.
Discussion
Breast cancer in India presents a distinct epidemiological pattern, with a significantly higher proportion of cases occurring in younger women with lower survival rates due to advanced stage at presentation, delays in treatment initiation, and fragmented or incomplete care compared to Western populations [16]. The World Cancer Report 2020 emphasizes that early detection and prompt treatment remain the most effective strategies to reduce breast cancer mortality [16].
Although, Core needle biopsy (CNB) is superior in certain grey-zone lesions such as atypical ductal hyperplasia (ADH) and papillary lesions, FNAC retains preference in resource-limited settings due to its rapid results, high patient compliance, cost-effectiveness, and its ability to sample both primary and metastatic sites [17].
Numerous studies evaluating IAC Yokohama System have been conducted worldwide, including across different regions of India; however, only a few originate from the Northeast, and even fewer from hilly geographical zones.
The present hospital-based retrospective study spanned 5.5 years (January 2020–June 2025), comprising 441 patients and 443 FNAC samples, with histopathological correlation available in 195 cases. All FNAC diagnoses were categorized according to the IAC Yokohama System, and comparative analysis with previously published work was performed.
In our study, the majority of cases occurred in younger individuals (53.74%), with an overall median age of 24 years and a median age of 45 years for malignant cases. Similar findings were reported by Verma D et al. [10], who observed a median age of 44 years (range: 15–85 years), with 34 years and 48 years as median ages for benign and malignant cases, respectively. Sigamani et al. [18] also noted a median age range of 30–40 years.
Female predominance was observed in our study (97.73%), with males accounting for only 2.27% of cases. This pattern aligns with findings reported by Kamatar PV et al. [19], Swain M et al. [11], Wong et al. [20], and Nargund et al. [21], all of whom documented similar female predominance.
In terms of laterality, the left breast was affected in 48.53% of cases and the right breast in 42.18%, while 9.29% showed bilateral involvement. The upper outer quadrant was the most frequently involved site. Comparable results were reported by Swain M et al. [11], who noted involvement of the left and right breast in 48.6% and 47.6% of cases, respectively, with only 3.8% bilateral cases.
Cyto-histopathological concordance in the present study was high (98.97%; 193/195), with only two discordant cases. Similar concordance rates were reported by Swain M et al. [11] (93.3%), Chauhan et al. [22] (98.4%), and Joshee et al. [23] (97.5%).
The distribution of FNAC cases across IAC Yokohama categories in our study was as follows: 5.65% insufficient, 69.07% benign, 3.61% atypical, 3.61% suspicious of malignancy, and 18.06% malignant. These findings correlate well with studies by Sunitha A et al. [7], Ahuja S et al. [8], Yadav R et al. [9], Verma D et al. [10], and Rohilla V et al. [12]. However, a different trend was observed by Cunha MD et al. [6] and Swain M et al. [11], where the majority of cases fell into the malignant category (Table 5).
Table 5: Comparison of IAC Yokohama distribution of cases with other studies.
|
IAC Yokohama category
|
Cunha MD, et al. [6]
|
Sunitha A et al [7]
|
Ahuja S et al [8]
|
Yadav R et al [9]
|
Verma D et al [10]
|
Swain M et al [11]
|
Rohilla V. et al [12]
|
Present study
|
|
Insufficient
|
0
|
6.89%
|
3.6%
|
3.8%
|
12.50%
|
3.8%
|
7.7%
|
5.65%
|
|
Benign
|
34.9%
|
72.46%
|
69.5%
|
71.6%
|
52.08%
|
19.1%
|
47.1%
|
69.07%
|
|
Atypical probably Benign
|
3.18%
|
3.28%
|
6.3%
|
6.3%
|
6.25%
|
15.2%
|
26.9%
|
3.61%
|
|
Suspicious for Malignancy
|
7.9%
|
3.28%
|
2.3%
|
3.1%
|
6.25%
|
4.8%
|
2.9%
|
3.61%
|
|
Malignant
|
53.9%
|
14.09%
|
18.2%
|
15.2%
|
22.92%
|
57.1%
|
15.4%
|
18.06%
|
The risk of malignancy (ROM) in the present study was 0% for both the insufficient and benign categories, 14.28% for the atypical category, 88.89% for suspicious cases, and 98.36% for malignant cases. In the insufficient category, our ROM was comparable to findings by Cunha MD et al. [6] (0%), Sunitha A et al. [7] (0%), Rohilla V et al. [12] (0%), and Ahuja S et al. [8] (5%), but differed significantly from Swain M et al. [11] (75%), Verma D et al. [10] (33.3%), and Yadav R et al. [9] (16.6%). This suggests that the insufficient samples in our study were either truly benign or were successfully repeated with adequate sampling.
For the benign category, our ROM matched those reported by Verma D et al. [10] and Swain M et al. [11], while slightly higher values were observed in studies conducted [6-9, 12]. In the atypical category, our ROM of 14.28% falls within the broad range reported in literature (0–50%). It was lower than that reported [6, 8-12]. The suspicious category demonstrated an ROM of 88.89%, comparable to studies by Swain M et al. [11] (100%), Rohilla V et al. [12] (100%), Ahuja S et al. [8] (81.8%), and Cunha MD et al. [6] (100%), but higher than values reported by Sunitha A et al. [7] (71.43%), Yadav R et al. [9] (75%), and Verma D et al. [10] (66.7%). The malignant category showed an ROM of 98.36%, closely matching the findings of all comparative studies (Table 6).
Table 6: Comparison of ROM with other studies.
|
IAC Yokohama category
|
Cunha MD, et al [6]
|
Sunitha A et al [7]
|
Ahuja S et al [8]
|
Yadav R et al [9]
|
Verma D et al [10]
|
Swain M et al [11]
|
Rohilla V. et al [12]
|
Present study
|
|
Insufficient
|
0%
|
0%
|
5%
|
16.6%
|
33.3%
|
75%
|
0%
|
0%
|
|
Benign
|
4.5%
|
2%
|
1.5%
|
1.02%
|
0%
|
0%
|
3.2%
|
0%
|
|
Atypical probably Benign
|
50%
|
0%
|
17.4%
|
19.04%
|
33.3%
|
25%
|
41.1%
|
14.28%
|
|
Suspicious for Malignancy
|
100%
|
71.43%
|
81.8%
|
75%
|
66.7%
|
100%
|
100%
|
88.89%
|
|
Malignant
|
100%
|
100%
|
100%
|
100%
|
100%
|
100%
|
100%
|
98.36%
|
In the present study, maximum sensitivity (100%) was achieved when atypical, suspicious, and malignant cases were collectively considered positive for malignancy. Even when only suspicious and malignant cases or exclusively malignant cases were regarded as positive, sensitivity remained high (98.55% and 86.96%, respectively), exceeding the values reported [8, 9, 12-15]. Specificity demonstrated a modest decline from analytical Group A (99.21%) to Group C (93.65%). This trend was comparable to findings reported by Ahuja et al. [8], Yadav et al. [9], and Rohilla et al. [12]. When atypical, suspicious, and malignant lesions were included as positive, the specificity of 93.65% in our study was substantially higher than those observed by Montezuma et al. [13] (54.8%), De Rosa et al. [14] (46.3%), and Agarwal et al. [15] (59.5%).
Diagnostic accuracy was highest when only suspicious and malignant cases were considered positive, yielding 98.46%. Including atypical cases resulted in a slight decline in diagnostic accuracy (95.90%), a finding consistent with the observations of Montezuma D et al. [13], De Rosa et al. [14], Agarwal et al. [15], Ahuja et al. [8], Yadav et al. [9], and Rohilla et al. [12].
A gradual decline in PPV was noted across the analytical groups, with the lowest PPV in Group C (89.61%) due to the inclusion of atypical cases. Conversely, NPV progressively increased across groups, reaching the highest value in Group C (100%). These trends were consistent with the comparative studies (Table 7).
Table 7: Comparison of statistical parameters with other studies.
|
Groups and statistical parameters
|
Montezuma D et al [13]
|
De Rosa et al [14]
|
Agarwal A et al [15]
|
Ahuja S et al [8]
|
Yadav R et al [9]
|
Rohilla V. et al [12]
|
Present study
|
|
Group A: Only Malignant cases
|
|
Sensitivity
|
68.7%
|
82.2%
|
86.7%
|
79.2%
|
82.1%
|
47.37%
|
86.96%
|
|
Specificity
|
100%
|
97.8%
|
100%
|
100%
|
100%
|
97.56%
|
99.21%
|
|
PPV
|
100%
|
98.8%
|
100%
|
100%
|
100%
|
90.0%
|
98.36%
|
|
NPV
|
87.7%
|
71.0%
|
71.2%
|
90.9%
|
93%
|
80.0%
|
93.28%
|
|
Diagnostic accuracy
|
90.3%
|
87.0%
|
90.0%
|
93.2%
|
94.7%
|
81.67%
|
94.87%
|
|
Group B: Both suspicious and malignant cases
|
|
Sensitivity
|
83.3%
|
93.7%
|
96.0%
|
91.7%
|
91.5%
|
52.63%
|
98.55%
|
|
Specificity
|
99.8%
|
90.8%
|
91.9%
|
98.7%
|
98.7%
|
97.56%
|
98.41%
|
|
PPV
|
99.5%
|
95.8%
|
97.3%
|
97.1%
|
96.6%
|
90.91%
|
97.14%
|
|
NPV
|
93.0%
|
86.6%
|
88.3%
|
96.1%
|
96.5%
|
81.63%
|
99.20%
|
|
Diagnostic accuracy
|
94.7%
|
92.8%
|
95.0%
|
96.4%
|
96.5%
|
83.33%
|
98.46%
|
|
Group C: Atypical, suspicious, and malignant cases
|
|
Sensitivity
|
98.3%
|
98.9%
|
98.2%
|
97.2%
|
95.7%
|
94.74%
|
100%
|
|
Specificity
|
54.8%
|
46.3%
|
59.5%
|
86%
|
90.2%
|
75.61%
|
93.65%
|
|
PPV
|
49.2%
|
80.5%
|
88.0%
|
77%
|
81.9%
|
64.29%
|
89.61%
|
|
NPV
|
98.6%
|
95.1%
|
91.7%
|
98.5%
|
97.9%
|
96.87%
|
100%
|
|
Diagnostic accuracy
|
68.2%
|
82.7%
|
88.6%
|
89.6%
|
86.3%
|
81.67%
|
95.90%
|
Limitations: This study was limited by a relatively small sample size due to the hospital’s hilly, sparsely populated location and COVID-19 disruptions. Loss to follow-up reduced histopathological correlation. Larger multicentric studies with improved patient compliance and follow-up systems are recommended.
Conclusion
Breast cancer is highly prevalent and represents a growing burden in Northeast India, reflecting national trends. FNAC, when interpreted using the standardized IAC Yokohama system, serves as a valuable and cost-effective diagnostic tool for evaluating breast lesions. The Yokohama system’s clear categorization enhances clinician acceptance and improves decision-making, particularly in resource-limited settings where access to advanced investigations is restricted.
Conflicts of interest
Authors declare no conflict of interest.
References
[1] Monica, Mishra R. An epidemiological study of cervical and breast screening in India: a district-level analysis. BMC Womens Health. 2020; 20:225.
[2] Mehrotra R, Yadav K. Breast cancer in India: present scenario and the challenges ahead. World J Clin Oncol. 2022; 13:209–218.
[3] Dogra A, Pant P, Joshi U, Pandey HS. Cytological evaluation of fine-needle aspiration of breast lesions using IAC Yokohama Classification System for Reporting Breast Cytopathology. J Med Sci Health. 2023; 9:245–250.
[4] Field AS, Raymond WA, Rickard M, Arnold L, Brachtel EF, et al. The International Academy of Cytology Yokohama System for reporting breast fine-needle aspiration biopsy cytopathology. Acta Cytol. 2019; 63:257–273.
[5] Chauhan V, Pujani M, Agarwal C, Chandoke R, Raychaudhuri S, et al. IAC standardized reporting of breast fine-needle aspiration cytology, Yokohama 2016: a critical appraisal over a 2-year period. Breast Dis. 2019; 38:109–115.
[6] Cunha MD, Kini RG. Breast fine needle aspiration biopsy cytology reporting using International Academy of Cytology Yokohama System: a single institution experience. Int J Clin Diagn Pathol. 2020; 3(3):303–306.
[7] Sunitha A, Chandra S, Rambabu, Kanth GVRNK. Application of IAC Yokohama System for breast cytology – the experience at a tertiary care hospital. IP Arch Cytol Histopathol Res. 2021; 6:237–242.
[8] Ahuja S, Malviya A. Categorization of breast fine-needle aspirates using the International Academy of Cytology Yokohama System along with assessment of risk of malignancy and diagnostic accuracy in a tertiary care centre. J Cytol. 2021; 38:158–163.
[9] Yadav R, Singh N, Bakna M, Bhadkariya A, Singh A. Cytomorphological spectrum of breast fine-needle aspiration cytology using the International Academy of Cytology Yokohama System and evaluation of associated risk of malignancy: a retrospective study in a tertiary care center. Surg Exp Pathol. 2024; 7:6.
[10] Verma D, Newaskar V, Balani S, Malik R, Khan A. Role of International Academy of Cytology Yokohama reporting system in breast lesions at a tertiary care centre in Central India. Surg Exp Pathol. 2024; 7:29.
[11] Swain M, Nedunuri S. Utility of Yokohama System for classification of breast FNA. Indian J Pathol Oncol. 2024; 11:20–26.
[12] Rohilla V, Kundu P, Gathwal M, Malik P, Gathwal C, et al. Categorization of breast fine-needle aspirates using Yokohama classification and its correlation with histopathological findings. Eur J Breast Health. 2025; 21:237–245.
[13] Montezuma D, Malheiros D, Schmitt FC. Breast fine-needle aspiration biopsy cytology using the newly proposed IAC Yokohama System for reporting breast cytopathology: the experience of a single institution. Acta Cytol. 2019; 1–6.
[14] De Rosa F, Migliatico I, Vigliar E, Salatiello M, Pisapia P, et al. The continuing role of breast fine-needle aspiration biopsy after the introduction of the IAC Yokohama System for reporting breast fine-needle aspiration biopsy cytopathology. Diagn Cytopathol. 2020; 48:1244–1253.
[15] Agarwal A, Singh D, Mehan A, Paul P, Puri N, et al. Accuracy of the International Academy of Cytology Yokohama System of breast cytology reporting for fine-needle aspiration biopsy of the breast in a dedicated breast care setting. Diagn Cytopathol. 2021; 49:195–202.
[16] Mehrotra R, Yadav K. Breast cancer in India: present scenario and the challenges ahead. World J Clin Oncol. 2022; 13:209–218.
[17] Mitra S, Dey P. Fine-needle aspiration and core biopsy in the diagnosis of breast lesions: a comparison and review of the literature. Cytojournal. 2016; 13:18.
[18] Sigamani K, Chandran N. Application of International Academy of Cytology Yokohama System for reporting breast fine-needle aspiration cytology: a retrospective study. Natl J Lab Med. 2022; 11:63–66.
[19] Kamatar PV, Athanikar VS, Dinesh US. Breast fine-needle aspiration biopsy cytology reporting using International Academy of Cytology Yokohama System: two-year retrospective study in a tertiary care centre in Southern India. Natl J Lab Med. 2019; 8:1–3.
[20] Wong YP, James EPV, Azhar M, Krishnamoorthy Y, Zainudin NA, et al. Implementation of the International Academy of Cytology Yokohama standardized reporting for breast cytopathology: an 8-year retrospective study. Diagn Cytopathol. 2021; 49:718–726.
[21] Nargund A, Mohan RH, Pai MM, Sadasivan B. Demystifying breast FNACs based on the International Academy of Cytology Yokohama Breast Cytopathology System: a retrospective study. J Clin Diagn Res. 2021; 15:1–5.
[22] Chauhan V, Pujani M, Agarwal C, Chandoke RK, Raychaudhuri S, et al. IAC standardized reporting of breast fine-needle aspiration cytology, Yokohama 2016: a critical appraisal over a 2-year period. Breast Dis. 2016; 38:109–115.
[23] Joshee A, Joshee R. Breast FNA cytology reporting using new proposed IAC Yokohama reporting system: a single-institution retrospective study. Int J Adv Res Med. 2021; 3:267–271.