Full Text
Introduction
Chronic kidney disease (CKD), also known as kidney failure, refers to the progressive deterioration of kidney function [1]. The kidneys play a crucial role in filtering waste products and metabolites from the bloodstream, excreting them through urine. When kidney function declines, toxic metabolites accumulate, leading to electrolyte imbalances and various systemic complications [2]. CKD is clinically defined by a persistent glomerular filtration rate (GFR) of less than 60 ml/min/1.73 m² for at least three months, often accompanied by albuminuria. It represents the end stage of various chronic renal parenchymal diseases [1, 2].
The prevalence of CKD varies globally, ranging from less than 1% to 13%, with recent data from the International Society of Nephrology’s Kidney Disease Data Center Study reporting a prevalence of 17% [1, 2]. In India, the etiology of CKD differs across regions. Certain areas, including Andhra Pradesh, Odisha, and Goa, have reported high cases of CKD of unknown etiology (CKDu), a chronic interstitial nephropathy characterized by an insidious onset and slow progression [1, 2].
Renal osteodystrophy is a common skeletal complication in CKD patients, primarily resulting from impaired calcitriol production, which leads to hypocalcemia. This condition triggers excessive secretion of parathyroid hormone (PTH), promoting bone resorption, weakening bone structure, and increasing the risk of fractures. Additionally, CKD patients, particularly those with comorbid conditions such as diabetes mellitus and hypertension, exhibit a higher incidence of bone fractures and delayed healing following orthopedic surgeries [2].
This study aims to evaluate the relationship between CKD and clinical outcomes in patients with hip and femur fractures.
Methodology
A retrospective cohort study was carried out at PSG Institute of Medical Sciences & Research, where patient records were reviewed to collect blood analysis values and clinical history after obtaining approval from the Institutional Ethics Committee. Patient health information was not recorded or stored, in compliance with the Institutional Health Insurance Portability and Accountability Act (HIPAA) policy.
Blood parameters, including creatinine, urea, blood urea nitrogen (BUN), Radiographic Union Score for Hip (RUSH) Score, and Bone Mineral Density (BMD) Score, along with the age of patients who underwent interlocking nailing of the femur between January 2017, and September 2022, were analyzed. Data from 21 male and female patients aged 50 years and above were included in the study. A bivariate analysis was performed, and statistical measures such as Pearson correlation and confidence intervals were calculated to assess potential correlations between the variables. Subtrochanteric fracture of the femur reduced with an intramedullary nail, as shown in Figures 1 to 4.
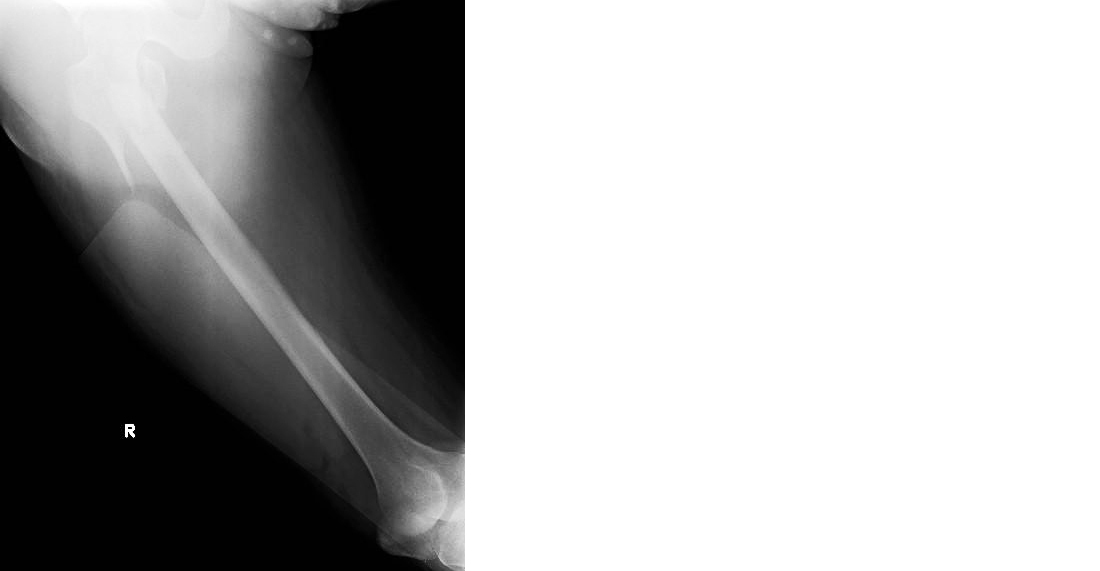
Figure 1: Preoperative radiograph showing subtrochanteric fracture right femur anteroposterior view.
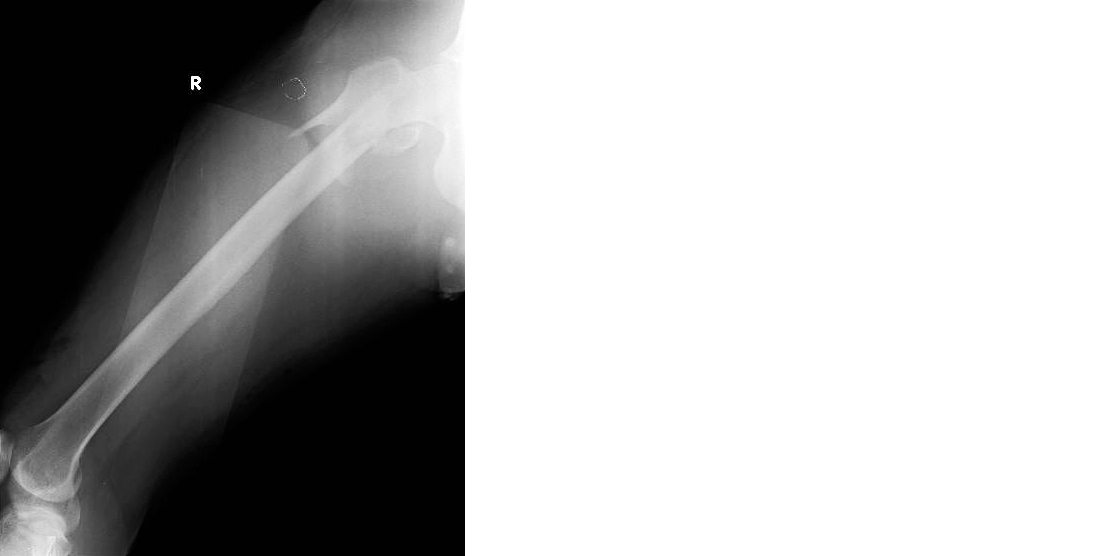
Figure 2: Preoperative radiograph showing subtrochanteric fracture right femur lateral view.
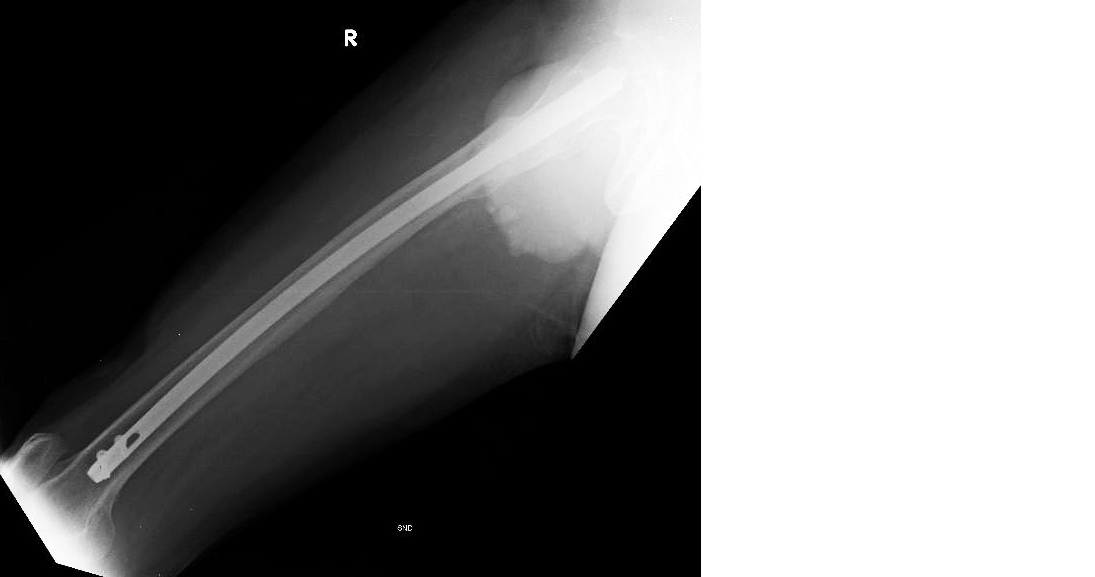
Figure 3: Post-operative radiograph showing subtrochanteric fracture right femur reduced with intramedullary nail lateral view.
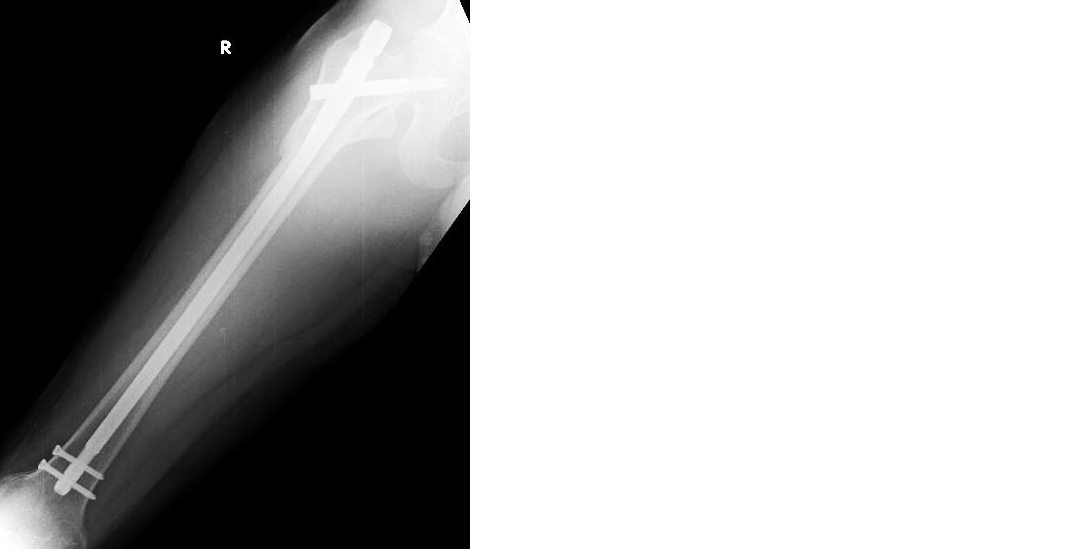
Figure 4: Post-operative radiograph showing subtrochanteric fracture right femur anteroposterior view.
To evaluate kidney function, serum creatinine, urea, and BUN levels were analyzed. These metabolites are produced during normal biochemical processes, and their elevated levels indicate a decreased ability of the kidneys to excrete waste products [3]. While these markers are generally reliable indicators of kidney function, they may be influenced by other factors which were considered during analysis.
For bone health and post-surgical healing assessment, BMD and RUSH scores were utilized.
BMD (Bone Mineral Density) score: This measures the severity of osteoporosis in individuals. Since vitamin D undergoes hydroxylation in the kidneys, impaired renal function results in reduced vitamin D activation, leading to decreased calcium absorption and an increased risk of osteoporosis. The BMD score is compared to the bone density of a healthy young adult [4]: (i) 1 – 2.5: Low bone density; (ii) 2.5 – 4: Onset of osteoporosis; (iii) > 4: Osteoporosis.
RUSH (Radiographic Union Score for Hip) score: This checklist-based system assesses fracture healing based on four parameters: cortical bridging, cortical fracture disappearance, trabecular consolidation, and trabecular disappearance. The RUSH score ranges from 10 to 30, with higher scores indicating better healing. This tool helps identify patients requiring additional treatment, prolonged care, or surgical intervention for nonunion.
Results
A RUSH score of 15.45 indicates some cortical bridging and trabecular consolidation, although visible fracture lines may still be present on imaging.
Interpretation of mean RUSH scores: A mean RUSH score of 15.45 suggests that while some degree of fracture healing has occurred, further assessment is necessary to evaluate complete bone union.
Clinical implication: Healthcare providers may recommend additional follow-up imaging to monitor fracture healing. Depending on the progress, alternative treatments or surgical interventions may be required. This finding suggests that most patients require post-surgical monitoring for further evaluation and management.
RUSH score: A Pearson correlation of -0.230 indicates a slight negative correlation between age and RUSH score. As age increases, the RUSH score tends to decrease, supporting the hypothesis that bone healing may decline with advancing age (Table 1).
Table 1: Age vs. RUSH score.
| |
N
|
Mean
|
SD
|
Pearson correlation
|
P value
|
95% confidence intervals
|
|
Age
|
20
|
65.2
|
9.418
|
-0.230
|
0.329
|
-0.611 -0.236
|
|
Rush score
|
20
|
15.45
|
1.669
|
|
|
|
BMD score: A Pearson correlation of 0.182 indicates a slight positive correlation between age and BMD scores. This suggests that as age increases, BMD scores also tend to rise, reflecting a worsening severity of osteoporosis. However, the weak correlation suggests that additional factors may influence bone mineral density beyond age alone (Table 2).
Table 2: Age vs. BMD score.
| |
N
|
Mean
|
SD
|
Pearson Correlation
|
P value
|
95% Confidence Intervals
|
|
Age
|
20
|
65.2
|
9.418
|
0.182
|
0.443
|
-0.284 -0.578
|
|
BMD score
|
20
|
4.239
|
0.61743
|
|
|
|
Serum creatinine: Creatinine is a waste metabolite produced by muscle metabolism and is excreted by the kidneys. Its levels generally remain stable despite variations in age, sex, and acute illnesses. However, elevated serum creatinine levels indicate declining kidney function.
A positive Pearson correlation (0.332) suggests that higher serum creatinine levels are associated with higher BMD scores. Since both elevated creatinine levels and increased BMD scores indicate worsening renal function and declining bone health, this correlation supports the link between impaired kidney function and osteoporosis progression (Table 3).
Table 3: BMD score vs. Creatinine.
| |
N
|
Mean
|
SD
|
Pearson Correlation
|
P value
|
95% Confidence Intervals
|
|
BMD Score
|
20
|
4.2395
|
0.61743
|
0.332
|
0.152
|
-0.129 -0.676
|
|
Creatinine
|
20
|
2.9375
|
2.6363
|
|
|
|
Serum urea and bone healing: Similar to serum creatinine, serum urea (blood urea nitrogen, or BUN) is a waste product formed during protein metabolism and serves as a key marker of kidney function. Elevated BUN levels can indicate kidney disease or failure, though other conditions such as liver disease, dehydration, and high-protein diets may also contribute to increased levels.
A negative Pearson correlation (-0.250) suggests that as urea levels increase, the RUSH score decreases, indicating slower bone healing with reduced cortical and trabecular consolidation. This finding is consistent with existing literature, which highlights the detrimental impact of impaired kidney function on bone healing post-surgery, often leading to delayed recovery and increased complications (Table 4).
Table 4: RUSH score vs. Urea.
| |
N
|
Mean
|
SD
|
Pearson Correlation
|
P value
|
95% Confidence Intervals
|
|
Rush Score
|
20
|
15.45
|
1.669
|
-0.250
|
0.288
|
-0.623 -0.217
|
|
Urea
|
20
|
73.5
|
35.769
|
|
|
|
A Pearson correlation of 0.193 indicates a mild positive correlation between BUN levels and BMD scores. This suggests a weak association between declining kidney function and worsening osteoporosis, supporting the hypothesis that kidney dysfunction may play a role in bone health deterioration. However, the weak correlation implies that additional factors may influence osteoporosis progression in patients with impaired renal function (Table 5).
Table 5: BMD score vs. BUN.
| |
N
|
Mean
|
SD
|
Pearson correlation
|
P value
|
95% Confidence intervals
|
|
BMD score
|
20
|
4.2395
|
0.61743
|
0.193
|
0.416
|
-0.273 -0.585
|
|
BUN
|
20
|
31.202
|
12.0893
|
|
|
|
Discussion
Existing literature indicates that individuals with chronic kidney disease (CKD) are at significant risk of developing renal osteodystrophy (ROD)—a condition that can lead to severe skeletal complications [1, 5]. Studies report that only 50% of hip fracture patients regain their pre-injury mobility and independence. In our study, we observed a slight correlation (Pearson correlation: 0.200–0.300) suggesting that declining kidney function may contribute to the development of ROD. However, this relationship is complex and influenced by multiple factors beyond the scope of this article.
Renal osteodystrophy is characterized by both osteoporosis and impaired bone healing after fractures. These factors were analyzed in our study using BMD scores and RUSH scores. While our findings align with existing literature linking CKD to ROD (as indicated by correlations between BMD, RUSH scores, and kidney function markers), the observed correlation was weaker than anticipated. This suggests that bone healing impairment involves more complex mechanisms beyond CKD alone [6, 7]. Thus, a key takeaway from this study is the importance of a multifaceted approach to bone disorders, rather than attributing them solely to kidney dysfunction.
In this study, creatinine and urea were used as biomarkers for kidney function [8]. While these markers are effective in assessing renal excretory function and providing a general measure of kidney health, they do not directly reflect the kidney’s role in specific metabolic pathways. A more targeted approach could involve measuring serum levels of Vitamin D and parathyroid hormone (PTH) to evaluate the kidney’s role in calcium metabolism [9]. This would offer a more precise understanding of how CKD impacts bone metabolism and healing, rather than serving as a generalized indicator of kidney function [10].
For bone health assessment, BMD and RUSH scores were utilized to evaluate postoperative recovery. However, incorporating additional biomarkers could improve patient monitoring and help determine the need for further interventions or surgeries.
Furthermore, larger prospective studies with extended follow-up periods would provide valuable insights into the long-term bone health trajectories of CKD patients following orthopedic procedures. In our study, CKD patients were analyzed without classifying the severity of their condition (e.g., mild, moderate, or severe CKD). Future studies should stratify patients based on CKD stages to investigate how different levels of kidney dysfunction impact bone healing rates. This would enhance our understanding of the extent to which CKD affects postoperative bone healing and help refine treatment strategies for these patients.
While blood markers such as creatinine and urea are commonly used to assess kidney function, their ability to predict bone healing and recovery in fracture patients remains limited. Although there is some evidence suggesting that decreased kidney function may contribute to delayed bone healing, our study found that the correlation between these markers and fracture recovery was not strong enough to establish a definitive link.
Monitoring kidney function remains essential in inpatient settings, particularly for CKD patients, as early identification of declining renal function can help manage associated comorbidities. However, our findings highlight the need for a more comprehensive approach when evaluating fracture healing in these patients. Future research should incorporate additional biomarkers, such as serum vitamin D and parathyroid hormone, to better understand the direct impact of CKD on bone metabolism and healing.
Beyond kidney function, other critical factors such as nutritional status, age, comorbidities, and the type of surgical intervention must be considered when assessing fracture recovery. A holistic approach to studying bone health in CKD patients will provide more meaningful insights into optimizing their management. Expanding research in this area and adopting more refined monitoring strategies will ultimately help improve treatment outcomes and enhance the recovery of CKD patients undergoing orthopedic procedures.
Conclusion
This study suggests that while kidney function markers such as creatinine and urea may indicate a potential link to fracture recovery outcomes, their correlation with bone healing remains weak. Although decreased kidney function might contribute to delayed healing, these markers alone do not strongly predict recovery. Monitoring renal function is essential, but additional biomarkers like vitamin D and parathyroid hormone should be explored for better assessment. Future research should consider factors like age, nutrition, and comorbidities to improve patient management. A comprehensive approach will help develop more effective treatment strategies, ultimately enhancing recovery outcomes in CKD patients with fractures.
Conflicts of interest
Authors declare no conflicts of interest.
References
[1] David V, Salusky IB, Malluche H, Nickolas TL. Renal osteodystrophy: something old, something new, something needed. Curr Opin Nephrol Hypertens. 2023; 32:559–564.
[2] Frank T, Osterhoff G, Sprague S, Garibaldi A, Bhandari M, et al. The Radiographic Union Score for Hip (RUSH) identifies radiographic nonunion of femoral neck fractures. Clin Orthop Relat Res. 2016; 474:1396–1404.
[3] Hosten AO. BUN and creatinine. In: Walker HK, Hall WD, Hurst JW, editors. Clinical Methods: The history, physical, and laboratory examinations. 3rd ed. Boston: Butterworths; 1990. Chapter 193. Available from: https://www.ncbi.nlm.nih.gov/books/NBK305/.
[4] Sheu A, Diamond T. Bone mineral density: testing for osteoporosis. Aust Prescr. 2016; 39:35–39.
[5] Jha V, Garcia-Garcia G, Iseki K. Chronic kidney disease: global dimension and perspectives. Lancet. 2013; 382:260–272. [6] Varma PP. Prevalence of chronic kidney disease in India—where are we heading? Indian J Nephrol. 2015; 25:133–135.
[7] Vaidya SR, Aeddula NR. Chronic kidney disease. [Updated 2024 Jul 31]. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2024. Available from: https://www.ncbi.nlm.nih.gov/books/NBK535404/.
[8] Gounden V, Bhatt H, Jialal I. Renal function tests. [Updated 2024 Jul 27]. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2024. Available from: https://www.ncbi.nlm.nih.gov/books/NBK507821/.
[9] Khundmiri SJ, Murray RD, Lederer E. PTH and vitamin D. Compr Physiol. 2016; 6:561–601.
[10] Cunningham J, Nitsch D, Stengel B. The epidemiology of chronic kidney disease in Europe. Nephrol Dial Transplant. 2015; 30:1–7.